Impact Factor ISSN: 1837-9664
J Cancer 2022; 13(4):1313-1323. doi:10.7150/jca.65444 This issue Cite
Research Paper
PLHPP2 inhibits the stemness of colorectal cancer by inactivating the Nrf2 signaling pathway
1. Department of Hepatobiliary Surgery, Affiliated Hospital of North Sichuan Medical College, Nanchong 637007, China.
2. Department of Thoracic and Hepatobiliary Surgery, Wusheng County People's Hospital, Guangan 638400, China.
3. Institute of Hepato-Biliary-Pancreatic-Intestinal Disease, North Sichuan Medical College, Nanchong 637007, China.
4. Research Office of Hepato-Biliary-Pancreatic-Intestinal Disease, Affiliated Hospital of North Sichuan Medical College, Nanchong 637007, China.
5. Department of Gastrointestinal Surgery, The First Affiliated Hospital of Chongqing Medical University, Chongqing, China.
6. Department of Gastrointestinal Surgery, Affiliated Hospital of North Sichuan Medical College, Nanchong 637007, China.
7. Weikang technology, Affiliated Hospital of North Sichuan Medical College, Nanchong 637007, China.
#These authors contributed equally to this work.
Abstract
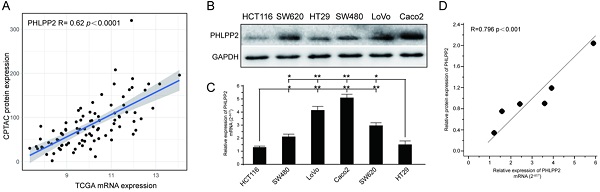
Pleckstrin homology (PH) domain leucine-rich repeat protein phosphatase 2 (PHLPP2) is a critical regulator of cellular homeostasis and acts as a tumor suppressor in multiple human cancers. However, its exact biological function in colorectal cancer (CRC) and the underlying molecular mechanism remain poorly understood. The correlation between the transcription and protein abundance of PHLPP2 was analyzed using proteomic and corresponding transcriptional data. Immunohistochemistry was used to validate the protein expression and the role of PHLPP2 in patient prognosis. In addition, a series of experiments in vitro and in vivo were performed to investigate the underlying molecular mechanism. Immunohistochemical staining of a CRC tissue microarray revealed that PHLPP2 protein expression was significantly downregulated compared to that in adjacent normal tissues. Low expression of PHLPP2 was an independent prognostic risk factor for poor survival. A nomogram established by integrating PHLPP2 expression and traditional clinicopathological factors achieved more reliable prognostic assessment in CRC patients. Additionally, PHLPP2 overexpression suppressed CRC cell migration, invasion and stemness in vitro as well as tumorigenesis in vivo. Further experiments revealed that upregulation of PHLPP2 increased ROS levels by suppressing the Nrf2-ARE signaling pathway, which inhibited the stemness of CRC cells. Moreover, incubation with sulforaphane, a selective chemical agonist of Nrf2, reversed this inhibitory effect in CRC. PHLPP2 acts as a tumor suppressor gene in CRC by restraining the Nrf2-ARE signaling pathway and increasing ROS levels, affecting the stemness of CRC cells. These anticancer molecular mechanisms indicate PHLLPP2's significant clinical value in prognosis prediction and targeted therapy.
Keywords: colorectal cancer, stemness, PHLPP2, Nrf2

 Global reach, higher impact
Global reach, higher impact