Impact Factor ISSN: 1837-9664
J Cancer 2022; 13(15):3606-3614. doi:10.7150/jca.77643 This issue Cite
Research Paper
Expression of Programmed Death Ligand-2 is associated with Prognosis in Nasopharyngeal Carcinoma Microenviroment
1. Department of Radiotherapy, the First Affiliated Hospital, Hengyang Medical School, University of South China, Hengyang, Hunan, 421001, China.
2. Department of Anesthesia, the Second Affiliated Hospital, Hengyang Medical School, University of South China, Hengyang, Hunan, 421001, China.
3. State Key Laboratory of Oncology in South China, Cancer Center, Sun Yat-Sen University, Guangzhou, 510060, China.
*These authors contributed equally to the article.
Abstract
Background: Although immune checkpoint inhibitors have opened a new mode of treatment for solid tumors, their efficacy in nasopharyngeal carcinoma (NPC) needs to be further investigated. Inhibitors of the PD-1/PD-L1 immune checkpoint are one of the hot topics in tumor immunotherapy. Programmed death ligand-2 (PD-L2) is a less studied ligand of PD-1 and has not yet been fully explored, especially in NPC. Understanding the clinical significance of PD-L2 expression, together with immune cell infiltration, might provide clues for biomarker screening in NPC immunotherapy. This study aimed to evaluate the role of PD-L2 as a prognostic factor for NPC patients as well as its role in immune regulation.
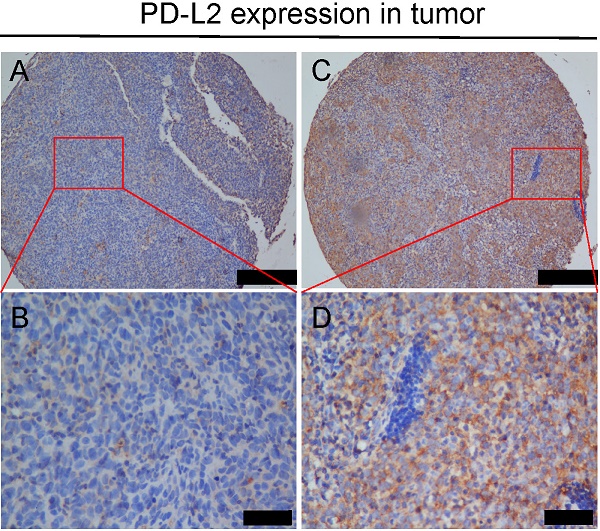
Methods: Immunohistochemistry (IHC) was performed on a tissue microarray including 557 NPC specimens using PD-L2 antibody. The immune cell markers CD4, FOXP3 and CD68 were also stained and quantified. The expression of PD-L2 exhibited different spatial patterns among NPC tumor and stromal tissues.
Results: A total of 90.8% of the cases showed membranous PD-L2 expression in tumors, and 80.8% showed membranous PD-L2 expression in stromal tissue. High stromal expression of PD-L2 predicted favorable overall and disease-free survival of NPC patients and was negatively correlated with tumor size, recurrence or metastasis and clinical stage. In contrast, high tumor abundance of PD-L2 correlated with poor disease-free survival, but had no obvious correlation with clinicopathological parameters. Multivariate analysis indicated that stromal PD-L2 was an independent and favorable prognostic factor. Furthermore, we found a positive correlation between stromal PD-L2 expression and the infiltration of CD68+ macrophages and CD4+Foxp3+ Treg cells in NPC stromal tissues (Pearson correlation=0.181 and 0.098, respectively).
Conclusions: Our results suggest that different PD-L2 expression patterns have distinct predictive values. PD-L2 expressed on stromal cells might play a role in the regulation of NPC progression, and involve in immune activation in the tissue microenvironment and have an independent good prognosis for NPC patients.

 Global reach, higher impact
Global reach, higher impact