Impact Factor
ISSN: 1837-9664
J Cancer 2022; 13(4):1097-1102. doi:10.7150/jca.67050 This issue Cite
Research Paper
Safety and anti-tumor effects of vismodegib in patients with refractory advanced gastric cancer: A single-arm, phase-II trial
1. Division of Hematology-Oncology, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
2. Department of Internal Medicine, Samsung Changwon Hospital, Sungkyunkwan University School of Medicine, Changwon, Korea.
3. Division of Hematology-Oncology, Department of Internal Medicine, Gyeongsang National University Hospital, Jinju, Korea.
#Co-authors contributed equally
Abstract
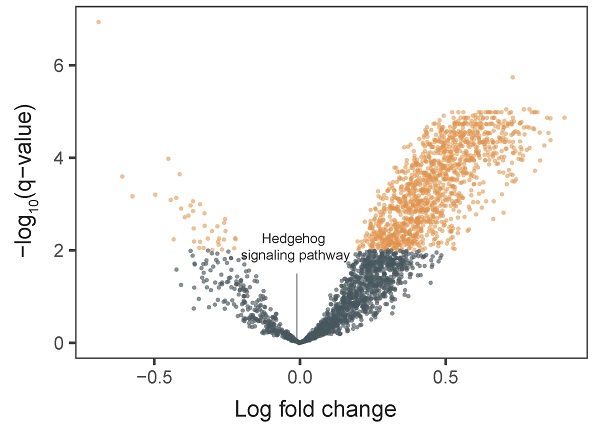
This phase-II study (ClinicalTrials.gov identifier: NCT03052478) aimed to evaluate the efficacy and safety of vismodegib, an inhibitor targeting the Hedgehog signaling pathway, in patients with refractory advanced gastric cancer. Patients with refractory advanced gastric cancer, whose disease had progressed after undergoing standard therapies, were enrolled in this phase-II trial of vismodegib. Vismodegib (150 mg) was administered orally once a day for a 21-day cycle. The primary endpoint was objective response rate, and the secondary endpoints were overall survival and safety profile. Tumor biopsies were obtained before vismodegib treatment. We conducted whole-exome and transcriptome sequencing to analyze biomarkers. Twenty-three patients were enrolled in this study. Among 19 patients who were eligible for response evaluation, only one showed stable disease, yielding a disease control rate of 5.3%. Median overall survival was 74 days (95% confidence interval, 74-151 days). Treatment-related adverse events of any grade were reported in seven patients (31.8%), and most were grade 1 or 2. Whole transcriptome data showed that the Hedgehog signaling pathway was not enriched in patient samples. This is the first clinical trial demonstrating the clinical activity and safety of vismodegib monotherapy in refractory advanced gastric cancer patients. Further well-designed clinical trials should be conducted to select advanced gastric cancer patients who are likely to benefit from vismodegib.
Keywords: Advanced gastric cancer, Hedgehog pathway, Phase II clinical trial, SMO, Vismodegib

 Global reach, higher impact
Global reach, higher impact