Impact Factor ISSN: 1837-9664
J Cancer 2022; 13(2):669-680. doi:10.7150/jca.61622 This issue Cite
Research Paper
PERK Regulates the Sensitivity of Hepatocellular Carcinoma Cells to High-LET Carbon Ions via either Apoptosis or Ferroptosis
1. Institute of Modern Physics, Chinese Academy of Sciences, Lanzhou 730000, China.
2. Key Laboratory of Heavy Ion Radiation Biology and Medicine, Chinese Academy of Sciences, Lanzhou 730000, China.
3. Key Laboratory of Basic Research on Heavy Ion Radiation Application in Medicine, Gansu Province, Lanzhou 730000, China.
4. University of Chinese Academy of Sciences, Beijing 100049, China.
Abstract
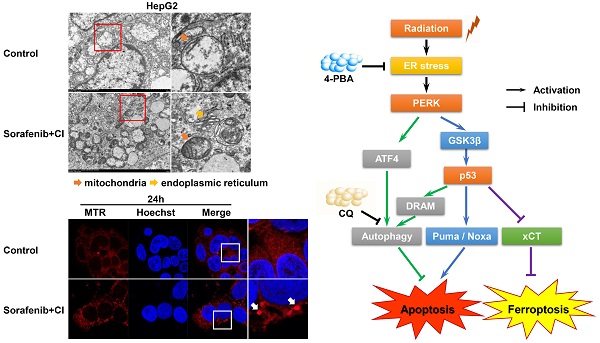
PERK is one of the transmembrane sensors of unfolded protein response (UPR) triggered by ER stress. In this study, we evaluated the role of PERK in the sensitivity of hepatocellular carcinoma (HCC) cells to high linear energy transfer (LET) carbon ions (CI). We found that CI irradiation could induce ER stress in HCC cells. On the one hand, PERK promoted autophagy via regulating ATF4 expression; on the other hand, PERK regulated p53 expression, and the latter either induced autophagy through up-regulating DRAM, or directly promoting apoptosis through the mitochondrial pathway or facilitating ferroptosis via down-regulating SLC7A11 (the extrinsic pathway), but independent of GPX4 (the intrinsic pathway). These factors jointly determined the sensitivity of HCC cells to high-LET CI radiation. Inhibiting TP53 directly increased cellular radioresistance definitely. Moreover, the death of HepG2 (TP53 wild type) cells induced by high-LET CI irradiation combined with sorafenib treatment might be caused by a mixed-type regulated cell death (RCD) including both apoptosis and ferroptosis, suggesting that apoptosis and ferroptosis are synergetic cell death modes regulated by TP53, which is one of the reasons why the sensitivity of HepG2 cells is higher than that of Hep3B (TP53 null type) and PLC/PRF5 (TP53 mutated type) cells. Therefore, our work might shed light on the potential therapeutic implication of CI radiotherapy combined with PERK targeted clinical drugs to implement personalized and precise treatment of HCCs.
Keywords: PERK, carbon ion, ER stress, autophagy, ferroptosis

 Global reach, higher impact
Global reach, higher impact