Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(23):7069-7078. doi:10.7150/jca.63415 This issue Cite
Research Paper
EZH2-mediated epigenetic suppression of lncRNA PCAT18 predicts a poor prognosis and regulates the expression of p16 by interacting with miR-570a-3p in gastric cancer
1. Department of Oncology, First Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu, 210000, PR China.
2. Department of Radiation Oncology, First Affiliated Hospital of Soochow University, Suzhou, Jiangsu, 215006, PR China.
3. Department of Biochemistry and Molecular Biology, Nanjing Medical University, Nanjing, Jiangsu, 210000, PR China.
4. Clinical Research Center, The Second Hospital of Nanjing, Nanjing University of Chinese Medicine. Zhong Fu Road, Gulou District, Nanjing, Jiangsu 210003, PR China.
5. Department of Respiratory Medicine, First Affiliated Hospital of Soochow University, Suzhou, Jiangsu, 215006, PR China.
#Co-first authors with equal contributions to this work.
Abstract
It was recently demonstrated that long noncoding RNAs (lncRNAs) have key regulation functions in the biology of human cancer. The current study aimed to determine the expression, clinicopathological characteristics and functional roles of lncRNA PCAT18 in gastric cancer (GC).
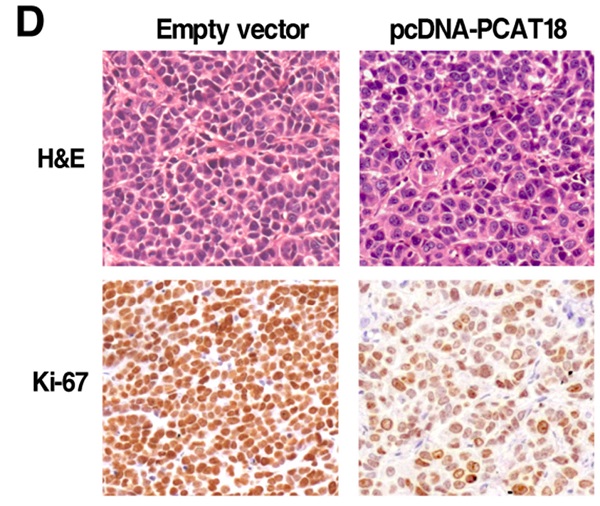
By analysis of (Gene Expression Omnibus) GEO and TCGA data, following experimental verification, we identified the function role and molecular mechanism of PCAT18 in tumorigenesis of GC. We discovered that PCAT18 is significantly decreased in paired GC tissues and correlates with a poor outcome. Mechanistic studies found that suppression of the expression of EZH2 could prevent its binding to the PCAT18's promoter region and decrease H3K27's trimethylation modification. In addition, PCAT18 could adjust cell proliferation of GC in vitro as well as in vivo. Further mechanism research revealed that PCAT18 could regulate the expression of p16 by interacting with miR-570a-3p, thus inhibiting cell proliferation of GC. Our results have shown that the histone modification-mediated epigenetic suppression of PCAT18 and its essential role of PCAT18 in GC oncogenesis, which could provide a theoretical basis for GC therapy.
Keywords: EZH2, PCAT18, long noncoding RNA, miRNA, p16, gastric cancer

 Global reach, higher impact
Global reach, higher impact