Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(14):4247-4256. doi:10.7150/jca.51657 This issue Cite
Research Paper
Downregulation of THRSP Promotes Hepatocellular Carcinoma Progression by Triggering ZEB1 Transcription in an ERK-dependent Manner
1. Department of Laboratory Medicine, Zhoushan Hospital of Zhejiang Province, Zhoushan, Zhejiang, 316021, China.
2. Department of Clinical Laboratory, Fudan University Shanghai Cancer Center; Department of Oncology, Shanghai Medical School, Fudan University, Shanghai, 200032, China.
Abstract
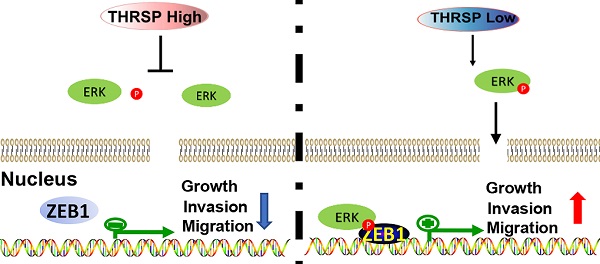
Background: Hepatocellular carcinoma (HCC) is a major leading cause of cancer mortality worldwide. Thyroid hormone responsive (THRSP) gene is primarily known for regulating responses to thyroid hormones, but its expression has been correlated with differential outcomes in some cancers. To date, however, its role in the progression of HCC remains unknown.
Methods: The mRNA and protein expression of THRSP was measured in HCC tissues and cell lines via qPCR and western blot assays. Lentiviral transfection was used to establish stable cell lines overexpressing THRSP and shRNA was used to silence THRSP. The effects of THRSP on cell growth were then determined in vivo and in vitro. Cell migration and invasion of HCC cells were investigated using transwell and wound healing assays.
Results: In tissue samples from patients, HCC tissues had decreased THRSP expression relative to adjacent healthy tissues. Further, patients with decreased THRSP protein and mRNA expression had worse outcomes. Knockdown of THRSP led to increased cell growth, migration, and invasion of HCC cells, and THRSP overexpression exerted an anti-tumor effect in vivo and in vitro. We found that increased expression of THRSP inhibited hepatocellular carcinogenesis by inhibiting the process of epithelial-to-mesenchymal transition through acting on the ERK/ZEB1 signaling pathway.
Conclusion: THRSP may act as a functional tumor suppressor and was frequently reduced in HCC tissue samples. We identified a novel pathway for the THRSP/ERK/ZEB1-regulated suppression of HCC tumorigenesis and invasion. Restoring THRSP expression may represent a promising approach for HCC therapies.
Keywords: Hepatocellular carcinoma, Thyroid hormone responsive gene, epithelial to mesenchymal phenotypes, HCC therapies

 Global reach, higher impact
Global reach, higher impact