Impact Factor ISSN: 1837-9664
J Cancer 2021; 12(11):3335-3343. doi:10.7150/jca.50324 This issue Cite
Research Paper
Cytoreductive Surgery may be beneficial for highly selected patients with Metastatic Gastrointestinal Stromal Tumors receiving Regorafenib facing Local Progression: A Case Controlled Study
1. GIST Team, Department of Surgery, Chang Gung Memorial Hospital, Linkou; Chang Gung University, Taiwan.
2. GIST Team, Division of Hematology-Oncology, Department of Internal Medicine, Chang Gung Memorial Hospital, Linkou; Chang Gung University, Taiwan.
*These authors equally contributed to this paper.
Abstract
Background: Current evidence have shown surgery may provide progression-free survival (PFS) benefit for selected patients with metastatic gastrointestinal stromal tumor (GIST) who received first line imatinib and second line sunitinib. However, impact of cytoreductive surgery for GIST patients receiving third line regorafenib facing progression is not yet reported.
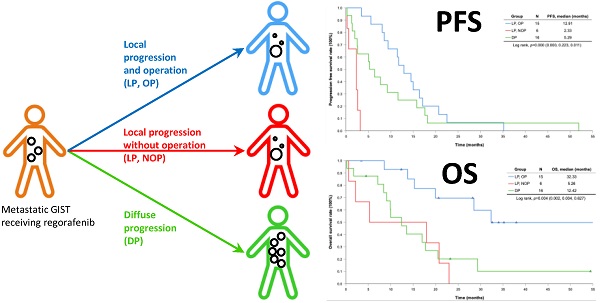
Methods: Between 2014 and 2019, 41 patients with metastatic GIST received regorafenib and 37 of them facing progression.
Results: 37 of 41 (90.2%) pre-treated GIST patients receiving regorafenib who experienced progression of disease after a median follow-up of 12.42 months after regorafenib use and 15 out of 37 (40.5%) patients with local progression underwent cytoreductive surgery (local progression and operation, LPOP). All the patients facing local progression (LP) were significantly younger with more exon 17 mutation than diffuse progression (DP). The complication rate for cytoreductive surgery was 33.3% (5/15). Cytoreductive surgery provided PFS prolongation of 5.52 months. Patients underwent cytoreductive surgery, compared with control group (local progression and no operation (LPNOP) and DP), may gain a significant PFS (12.91 versus 2.33 versus 5.29 months, p = 0.0001) and overall survival (OS) benefit (32.33 versus 5.26 versus 12.42 months, p = 0.004).
Conclusions: Cytoreductive surgery might be feasible in highly selected patients with pre-treated GIST who are being treated with regorafenib experiencing LP.
Keywords: cytoreductive Surgery, GIST, regorafenib, local progression

 Global reach, higher impact
Global reach, higher impact