Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(11):3315-3324. doi:10.7150/jca.56262 This issue Cite
Research Paper
RNA-binding protein YBX1 promotes cell proliferation and invasiveness of nasopharyngeal carcinoma cells via binding to AURKA mRNA
1. Hunan Key Laboratory of Cancer Metabolism, Hunan Cancer Hospital and the Affiliated Cancer Hospital of Xiangya School of Medicine, Central South University, Changsha 410013, Hunan, China.
2. The Key Laboratory of Carcinogenesis and Cancer Invasion of the Chinese Ministry of Education, Cancer Research Institute and School of Basic Medical Sciences, Central South University, Changsha 410078, Hunan, China.
3. The Key Laboratory of Carcinogenesis of the Chinese Ministry of Health, Xiangya Hospital, Central South University, Changsha 410008, Hunan, China.
4. Hunan Key Laboratory of Nonresolving Inflammation and Cancer, The Third Xiangya Hospital, Central South University, Changsha 410013, Hunan, China.
5. Department of Dermatology, The Second Xiangya Hospital, The Central South University, Changsha 410011, Hunan, China.
6. Department of Dermatology, Xiangya Hospital, Central South University, Changsha 410008, Hunan, China.
* These authors contributed equally.
# Present address: Department of pathology, the third affiliated hospital, Sun Yat-Sen University, Guangzhou, Guangdong 510630, China.
Abstract
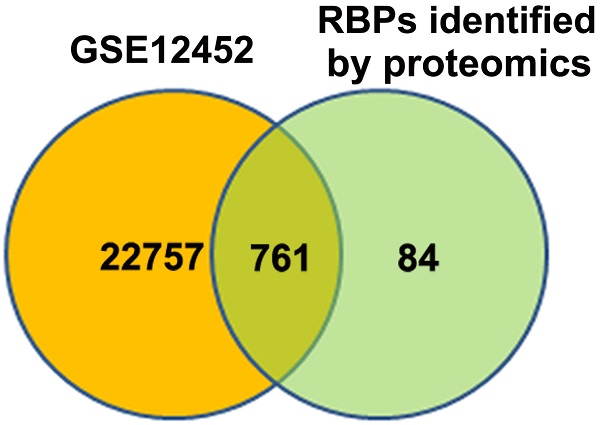
Background: RNA-binding proteins (RBPs) play essential roles in post-transcriptional control of gene expression. Dysregulation of RBPs is intensively implicated in development and progression of human diseases, including cancers. However, the roles of RBPs in nasopharyngeal carcinoma (NPC), which is a distinct subtype of head and neck cancer, remain elusive.
Methods: NPC-related RBPs were explored by analyzing GEO database and high-throughput proteomic data obtained from crosslinking immunoprecipitation. The expression levels of Y box binding protein 1 (YBX1) protein in NPC samples were measured by immunohistochemistry (IHC) staining. The association of YBX1 protein levels with prognosis of NPC patients was analyzed by Kaplan-Meier Plotter. The expression levels of YBX1 in NPC cells were inhibited by RNA interference. Cell growth was measured by CCK-8 assay. Cell mobility and invasiveness were measured by transwell assays. Tumorigenicity was measured by using a xenograft tumor assay. The expression levels of mRNAs or proteins were determined by qPCR or western blot assays, respectively. The mRNAs binding to YBX1 were determined by RNA immunoprecipitation (RIP) and qPCR. The effect of YBX1 on mRNA translation was measured by luciferase reporter assay.
Results: In the present study, we demonstrated a differentially expressed RBPs profile between NPC and its normal counterpart. Among these aberrantly expressed RBPs, YBX1 was overexpressed in NPC. We found that YBX1 is mainly localized in the cytoplasm of NPC cells. Loss of YBX1 led to reduced cell proliferation, migration and invasiveness in vitro, and reduced tumorigenicity in vivo. Overexpression of YBX1 associates with high expression of cell cycle G2/M checkpoint modulators. In addition, YBX1 promotes AURKA protein expression by directly binding to its mRNA. Loss of YBX1 leads to reduction of AURKA protein level. Forced expression of AURKA rescues cell proliferation and invasiveness in YBX1-silenced NPC cell.
Conclusions: The current study indicated that YBX1 promotes NPC cell proliferation and invasiveness through enhancing protein synthesis of AURKA.
Keywords: RNA binding protein, posttranscriptional regulation, YBX1, AURKA, translation.

 Global reach, higher impact
Global reach, higher impact