Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(6):1698-1707. doi:10.7150/jca.40500 This issue Cite
Research Paper
Expression of PD1/PDL1 in gastric cancer at different microsatellite status and its correlation with infiltrating immune cells in the tumor microenvironment
1. Tumor Etiology and Screening Department of Cancer Institute and General Surgery, the First Hospital of China Medical University, Shenyang 110001, China.
2. Key Laboratory of Cancer Etiology and Prevention in Liaoning Education Department, the First Hospital of China Medical University, Shenyang 110001, China.
3. Key Laboratory of GI Cancer Etiology and Prevention in Liaoning Province, the First Hospital of China Medical University, Shenyang 110001, China.
4. Department of Medical Oncology, the First Affiliated Hospital of Jinzhou Medical University, Jinzhou 121001, China.
Abstract
Objective: The microsatellite status and tumor immune microenvironment have a remarkable influence on tumor immunotherapy. This study was performed to investigate programmed cell death protein 1/programmed death ligand 1 (PD1/PDL1) expression and their correlations with CD8+ T cell/CD68+ macrophage (CD68+ M) densities in gastric cancer (GC) at different microsatellite statuses.
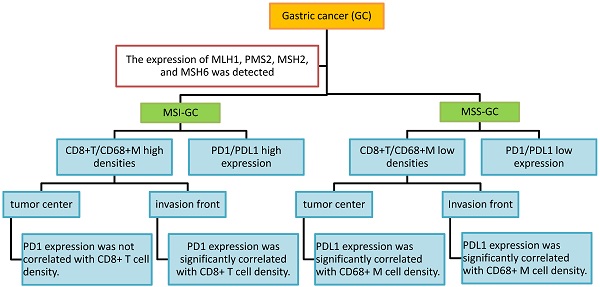
Methods: The expression of MLH1, PMS2, MSH2, and MSH6 was detected via immunohistochemistry (IHC) to determine the microsatellite status in 215 GC samples obtained from surgical resections. Furthermore, the expression of PD1, PDL1, CD8, and CD68 was detected in the samples via IHC, and the differences and correlations in GC at different microsatellite statuses were then analyzed. PDL1 expression in tumor cells was labeled as PDL1[T], while expression of PD1 and PDL1 in tumor-infiltrating immune cells was labeled as PD1 and PDL1, respectively. Kaplan-Meier analysis was used to evaluate the significance of PD1/PDL1 expression in determining overall survival. Multivariate Cox regression analysis was performed using SPSS software. P-values were determined using the log-rank test.
Results: Our results indicated that PD1, PDL1[T], and PDL1 positivity rates were 59%, 35%, and 57% in 46 microsatellite unstable (MSI) GCs and 45%, 22%, and 40% in 169 microsatellite stable (MSS) GCs, respectively. Compared with MSS GC, PD1, PDL1[T], and PDL1 expression was higher in MSI GC (P = 0.109, 0.090, and 0.044, respectively). Additionally, CD8+ T cell and CD68+ M densities were higher in MSI GC than in MSS GC (P = 0.537 and <0.001, respectively). Additionally, CD8+ T cell/CD68+ M densities were evaluated according to tumor center and invasion front. We found that PD1 expression was significantly correlated with CD8+ T cell density at the invasion front of the MSI GC (P = 0.031), whereas PDL1 expression was significantly correlated to high CD68+ M density in the tumor center and invasion front of MSS GC (P = 0.001 and 0.014, respectively). Survival analysis showed that patients with PD1-positive and PDL1[T]/PDL1-negative GC had better prognosis (P = 0.012, 0.005, and 0.022, respectively). Multivariate Cox survival analysis showed that PDL1[T] was an independent prognostic factor for GC.
Conclusion: The results suggested that PD1/PDL1 expression and immune response varied at different microsatellite statuses in GC. PD1/PDL1 expression was correlated with CD8+ T cell/CD68+ M densities in GC at different microsatellite statuses, especially at the invasion front. The patients exhibiting high PD1/PDL1 expression or high CD8+ T cell/CD68+ M densities MSI GC might be potential beneficiaries of PD1/PDL1 immunotherapy.
Keywords: gastric cancer, tumor immune microenvironment, PD1, PDL1, CD8, CD68

 Global reach, higher impact
Global reach, higher impact