Impact Factor ISSN: 1837-9664
J Cancer 2026; 17(3):679-687. doi:10.7150/jca.129779 This issue Cite
Research Paper
A CRM-Integrated ypT Staging System Improves Prognostic Stratification Following Neoadjuvant Therapy in Rectal Cancer
1. Department of Radiation Oncology, Chi Mei Medical Center, Tainan, Taiwan.
2. Department of Medical Research, Chi Mei Medical Center, Tainan, Taiwan.
3. Department of Information Management, Southern Taiwan University of Science and Technology, Tainan, Taiwan.
4. Division of Colorectal Surgery, Department of Surgery, Chi Mei Medical Center, Tainan, Taiwan.
5. Center of General Education, Chia Nan University of Pharmacy and Science, Tainan, Taiwan.
6. Department of Pharmacy, Chia-Nan University of Pharmacy and Science, Tainan, Taiwan.
7. School of Medicine, College of Medicine, National Sun Yat-sen University, Kaohsiung, Taiwan.
* Hsuan-Yi Huang and Ching-Chieh Yang contributed equally as corresponding author to this work.
Received 2025-12-11; Accepted 2026-3-3; Published 2026-3-17
Abstract
Introduction: The ypT staging system has limited prognostic value after neoadjuvant therapy, as it primarily reflects only tumor characteristics alone. This study proposes a novel staging system that integrates circumferential resection margin (CRM) status with the ypT category to enhance prognostic accuracy following neoadjuvant chemoradiotherapy (nCRT) for rectal cancer.
Methods: We analyzed data from 4,308 rectal adenocarcinoma patients treated with nCRT followed by surgery, using the Taiwan Cancer Registry and National Health Insurance Research Database (2011-2021). CRM involvement was defined as a margin ≤1 mm. Overall survival was assessed using multivariable Cox regression, and prognostic performance of the proposed CRM-integrated ypT staging system was compared with the American Joint Committee on Cancer (AJCC) TNM system using Harrell's c-statistic.
Results: CRM involvement (≤1 mm) was significantly associated with worse 5-year survival (adjusted odds ratio, 0.44; 95% CI, 0.31-0.61). Due to the low rate of CRM positivity in ypT0-2 patients, a modified ypT classification was established: new ypT3 (ypT3 and CRM-), new ypT4A (ypT4A and CRM-), new ypT4B (ypT3 and CRM+ or ypT4B and CRM-), and new ypT4C (ypT4A and CRM+ or ypT4B and CRM+). This system demonstrated better prognostic discrimination than the current AJCC classification (Harrell's c-statistic: 0.756 vs. 0.752, P = 0.034).
Conclusions: Incorporating CRM into the ypT stage offers survival stratification and may guide more individualized postoperative treatment strategies for rectal cancer patients after nCRT.
Keywords: rectal cancer, neoadjuvant chemoradiotherapy, CRM, ypT, survival.
1. Introduction
Surgical resection is a cornerstone of curative therapy for rectal cancer. Introduced in the early 1980s, total mesorectal excision (TME) has become the surgical standard, leading to marked improvements in local control and long-term survival [1-3]. However, surgery alone is often insufficient for optimal tumor control, especially in cases involving bulky tumors or lesions that extend toward the mesorectal fascia, where achieving a clear circumferential resection margin (CRM) can be technically challenging. This limitation has driven the widespread adoption of neoadjuvant chemoradiotherapy (nCRT), which enhances local control and reduces perioperative morbidity [4-8]. Consequently, this treatment paradigm has not only improved surgical outcomes but also shifted clinical emphasis toward pathological features, including post-treatment tumor and nodal status (ypT/ypN), tumor regression grade (TRG), and CRM.
Among these, CRM status is one of the most extensively validated predictors of both local recurrence and long-term survival [9-12]. Liu et al., analyzing a large national cohort of rectal cancer patients undergoing surgery, demonstrated that a CRM ≤ 1 mm was independently associated with nearly double the risk of cancer-specific mortality compared with margins of 1.1-2.0 mm (HR = 1.99, 95% CI = 1.61-2.45, P < 0.001), underscoring the importance of achieving a clear CRM at resection [9]. Similarly, Park et al. reported that among various CRM thresholds, ≤ 1 mm provided the strongest prognostic discrimination for disease-free survival in both chemoradiotherapy and non-chemoradiotherapy groups [10]. In a meta-analysis of 75 studies including over 85,000 patients published between 2006 and 2019, Detering et al. found that CRM involvement was significantly associated with inferior 5-year outcomes: local recurrence (OR = 4.67), disease-free survival (OR = 3.63), distant metastasis (OR = 2.95), and overall survival (OR = 3.21), even in the era of modern surgical and multidisciplinary treatment [11]. More recently, in a propensity-matched cohort of stage II/III rectal cancer patients, CRM involvement was again associated with markedly poorer outcomes, with 5-year recurrence-free survival of 15.7% versus 57.9% (HR = 2.72, P = 0.004) and cancer-specific survival of 35.3% versus 71.3% (HR = 3.52, P = 0.002) [12].
The prognosis and treatment strategies for rectal cancer are largely guided by the American Joint Committee on Cancer (AJCC) TNM classification. However, the current system lacks sufficient precision in outcome prediction, as it primarily reflects tumor characteristics alone [13]. Patients within the same stage often experience markedly different outcomes, underscoring the need to refine the TNM staging system to better identify individuals at high risk of recurrence or death. Previous studies have established CRM as both a marker of surgical quality and an independent prognostic factor. Therefore, integrating CRM with the ypT classification may provide clinicians with more accurate prognostic information. This study investigates whether incorporating CRM into the ypT category can improve survival risk stratification in rectal cancer patients treated with nCRT.
2. Materials and Methods
2.1 Data source and study cohort
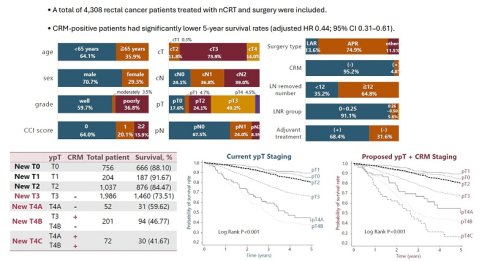
This study was designed to assess the effect of CRM on survival of rectal cancer patients. We retrieved data from the national TCR and NHIRD between January 2011 and December 2021, which captures almost all cancer patients in Taiwan. Because the NHI institute employed an accuracy program to verify the quality of coding, the TCR database has been approved as excellent and complete [14]. According to the International Classification of Disease for Oncology, third edition codes, the study cohort included patients diagnosed with topography codes for 'rectum' (C20.9) and histology codes for 'adenocarcinoma' (8140, 8210, 8263, and 8280) [15]. The staging for all cases was defined based from the 8th edition American Joint Committee on Cancer (AJCC) classification system [16]. Collected demographic and clinicopathological variables included age, sex, clinical and pathological tumor and nodal category, grade, surgery type, CRM, lymph node yield, lymph node ratio, comorbid status, and cancer-related treatment. Surgical procedures were categorized as low anterior resection, abdominoperineal resection, and others which included pull-through with sphincter preservation, proctectomy or proctocolectomy with en bloc resection of adjacent organs, or local tumor excision/total proctocolectomy. The CRM, recorded to the nearest tenth of a millimeter in the pathology report, represents the distance between the tumor's leading edge and the nearest surgically dissected margin, as defined by the AJCC Seventh Edition Cancer Staging Manual as the non-peritonealized surface of the specimen. A CRM ≤ 1 mm is defined as positive in our database and selected for analysis. The Charlson Comorbidity Index (CCI) score was employed to quantify the complexity of comorbid conditions [17]. Patients were excluded if they had (1) a prior history of cancer, including non-rectal malignancies, or metastatic disease identified intraoperatively and pathologically confirmed, or (2) incomplete or unclear coding of clinicopathological or treatment information. Finally, a total of 4308 rectal cancer patients received nCRT and surgery were included in our study participants (Figure 1).
Flow chart of study participant.
2.2 Statistical Analysis
2.2.1Patient information
Demographic and clinicopathological in-formation were presented with descriptive statistic. The primary endpoint was 5-year overall survival, calculated from the date of cancer diagnosis rather than the date of surgery. All statistical analyses were performed using SAS 9.4 for Windows (SAS Institute, Inc., Cary, NC, USA), and Stata version 15 (Stata Corp, College Station, Texas, USA). Statistical significance was set at P < 0.05.
2.2.2 Identification and handling of CRM-inclusive ypT staging system
By using a logistic regression model, the estimated effect between survival rate, ypT, CRM, and those prognostic factors, was presented as odd ratios (ORs) with 95% confidence intervals (95% CIs). Prognostic factors potentially related to the survival that were significant on univariable analysis were entered into the multivariable analysis. According to the study aim, we modified ypT category incorporating CRM status to assess the association between survival and achieving a clear margin in CRM-positive patients. Survival rate stratified by modified ypT category incorporating CRM status displayed a monotonically decreasing trend, and the Cochran-Armitage trend test was used to test the ordinal trend in the newly defined staging system [18]. To compare the prognostic discrimination of the CRM-inclusive ypT staging system versus the traditional ypT classification, we applied Harrell's concordance index (C-index) to compare the performance of the respective staging categories [19]. In addition, cumulative survival rates in the newly defined staging system were also used to evaluate the time-dependent survival predictive performance, by using the Kaplan-Meier method, and the differences between curves were derived from the Log-rank test. Heagerty's integrated area under the curve (iAUC) analysis estimates the time-dependent predictive performance using a rank-based method for the followed-up survival [20]. The C-index and iAUC, ranging from 0.5 (no discrimination) to 1.0 (perfect discrimination), is used to assess predictive accuracy in survival prediction.
2.2.3 CRM-inclusive ypT staging system validation
Internal validation was performed using bootstrapping to assess potential optimism in the performance of the CRM-inclusive ypT staging system. A total of 500 bootstrap samples were generated from the original dataset, with stepwise variable selection repeated in each sample. This nonparametric approach estimates prediction error and provides stable, assuming the study population represents a random sample of rectal cancer patients [21]. External validation was conducted using an independent dataset to evaluate the consistency of staging system performance, including patient in different ypN and lymph node yield.
3. Results
3.1 Patient Characteristics
The baseline characteristics of the study cohort are summarized in Table 1. A total of 4,308 rectal cancer patients treated with neoadjuvant chemoradiotherapy followed by surgery were included, comprising 3,046 males (70.7%) and 1,262 females (29.3%), with a mean age of 60.4 ± 11.7 years and a mean follow-up of 3.5 ± 1.6 years. Clinical tumor status (cT) was cT1 in 13 (0.3%), cT2 in 508 (11.8%), cT3 in 3,169 (73.9%), and cT4 in 601 (14.0%) patients. Clinical nodal status (cN) was cN0 in 1,037 (24.1%), cN1 in 1,583 (36.8%), and cN2 in 1,677 (39.0%). After nCRT and surgery, pathological tumor status (ypT) was ypT0 in 756 (17.6%), ypT1 in 204 (4.7%), ypT2 in 1,037 (24.1%), ypT3 in 2,118 (49.2%), and ypT4 in 193 (4.5%). Pathological nodal status (ypN) was ypN0 in 2,908 (67.5%), ypN1 in 1,035 (24.0%), and ypN2 in 365 (8.5%). Most patients (74.9%) underwent low anterior resection, 64.8% had a lymph node yield ≥12, and 2,948 (68.4%) received adjuvant chemotherapy. CRM positivity (≤1 mm) was observed in 208 patients (4.8%). Survival outcomes by baseline characteristics are shown in Supplementary Table 1.
Baseline characteristic of rectal cancer patients treated with neoadjuvant chemoradiotherapy and surgery.
| N | % | |
|---|---|---|
| Overall | 4308 | 100.00 |
| Age, years | ||
| < 65 | 2761 | 64.09 |
| ≧ 65 | 1547 | 35.91 |
| Sex | ||
| Male | 3046 | 70.71 |
| Female | 1262 | 29.29 |
| Grade | ||
| Well | 2573 | 59.73 |
| Moderately | 152 | 3.53 |
| Poorly | 1583 | 36.75 |
| cT category, missing n=17 | ||
| 1 | 13 | 0.30 |
| 2 | 508 | 11.84 |
| 3 | 3169 | 73.85 |
| 4 | 601 | 14.01 |
| cN category, missing n=11 | ||
| 0 | 1037 | 24.13 |
| 1 | 1583 | 36.84 |
| 2 | 1677 | 39.03 |
| ypT category | ||
| 0 | 756 | 17.55 |
| 1 | 204 | 4.74 |
| 2 | 1037 | 24.07 |
| 3 | 2118 | 49.16 |
| 4 | 193 | 4.48 |
| ypN category | ||
| 0 | 2908 | 67.50 |
| 1 | 1035 | 24.03 |
| 2 | 365 | 8.47 |
| Surgery type | ||
| LAR | 3228 | 74.93 |
| APR | 584 | 13.56 |
| Others | 496 | 11.51 |
| CRM | ||
| negative | 4100 | 95.17 |
| positive | 208 | 4.83 |
| LN removed number | ||
| < 12 | 1518 | 35.24 |
| ≧ 12 | 2790 | 64.76 |
| LNR group | ||
| 0-0.25 | 3926 | 91.13 |
| 0.26-0.50 | 241 | 5.59 |
| 0.56-0.75 | 91 | 2.11 |
| 0.76-1.00 | 50 | 1.16 |
| Adjuvant treatment | 2948 | 68.43 |
| CCI score | ||
| 0 | 2756 | 63.97 |
| 1 | 866 | 20.10 |
| ≧ 2 | 686 | 15.92 |
Abbreviations: cT, clinical tumor; cN, clinical nodal; ypT, pathological tumor; ypN, pathological nodal; LAR, low anterior resection; APR, abdominoperineal resection; CRM, circumferential resection margin; LN, lymph node; LNR, lymph node ratio, CCI, Charlson Comorbidity Index.
3.2 Modified ypT staging system in combination with CRM
In both univariable and multivariable analyses, CRM positivity was significantly associated with lower 5-year survival and remained an independent prognostic factor after adjustment for clinicopathologic variables (adjusted odds ratio [aOR] 0.44; 95% CI: 0.31-0.61; Table 2). Based on these findings, we developed a modified staging system combining ypT category and CRM (Table 3). The new prognostic staging system was defined as follows: new ypT3 (ypT3 and CRM-), new ypT4A (ypT4A and CRM-), new ypT4B (ypT3 and CRM+ or ypT4B and CRM-), and new ypT4C (ypT4A and CRM+ or ypT4B and CRM+). Patients with ypT0-2 were not reclassified in the modified system due to the small number with CRM positivity; only four patients (ypT0: n = 1; ypT1: n = 1; ypT2: n = 2) had CRM-positive status. The new system showed a stepwise decline in 5-year survival from 73.5% (new ypT3) to 59.6% (new ypT4A), 46.8% (new ypT4B), and 41.7% (new ypT4C), compared with 71.8% (ypT3) and 48.5% (ypT4) in the AJCC system. Overall, the new system demonstrated superior prognostic discrimination, with a higher C-index (0.756 vs. 0.752, P = 0.034; Table 4) and better curve separation (Supplementary Figure 1).
The risk factor of death in rectal cancer patients treated with neoadjuvant chemoradiotherapy and surgery.
| Variable | Crude OR (95% CIs) | P-value | Adjusted OR (95% CIs) | P-value |
|---|---|---|---|---|
| ypT category | ||||
| 0 | Ref. | Ref. | ||
| 1 | 1.45 (0.85-2.49) | 0.172 | 1.61 (0.92-2.80) | 0.095 |
| 2 | 0.74 (0.56-0.97) | 0.031 | 0.75 (0.56-1.00) | 0.052 |
| 3 | 0.35 (0.27-0.44) | < .001 | 0.42 (0.33-0.55) | < .001 |
| 4 | ||||
| T4A | 0.15 (0.09-0.25) | < .001 | 0.22 (0.12-0.38) | < .001 |
| T4B | 0.12 (0.08-0.18) | < .001 | 0.16 (0.10-0.26) | < .001 |
| CRM | ||||
| negative | Ref. | Ref. | ||
| positive | 0.22 (0.16-0.29) | < .001 | 0.44 (0.31-0.61) | < .001 |
| Age, years | ||||
| < 65 | Ref. | Ref. | ||
| ≧ 65 | 0.48 (0.41-0.55) | < .001 | 0.47 (0.40-0.55) | < .001 |
| Sex | ||||
| Male | 0.73 (0.62-0.86) | < .001 | 0.68 (0.57-0.82) | < .001 |
| Female | Ref. | Ref. | ||
| Grade | ||||
| Well | Ref. | Ref. | ||
| Moderately | 0.26 (0.19-0.37) | < .001 | 0.31 (0.22-0.46) | < .001 |
| Poorly | 0.98 (0.84-1.14) | 0.802 | 1.01 (0.86-1.19) | 0.909 |
| ypN category | ||||
| 0 | Ref. | Ref. | ||
| 1 | 0.45 (0.38-0.53) | < .001 | 0.51 (0.42-0.62) | < .001 |
| 2 | 0.23 (0.19-0.29) | < .001 | 0.40 (0.27-0.61) | < .001 |
| Surgery type | ||||
| LAR | Ref. | Ref. | ||
| APR | 0.49 (0.41-0.59) | < .001 | 0.59 (0.47-0.73) | < .001 |
| Others | 0.99 (0.79-1.25) | 0.944 | 0.89 (0.69-1.15) | 0.387 |
| LNR group | ||||
| 0-0.25 | Ref. | Ref. | ||
| 0.26-0.50 | 0.34 (0.26-0.45) | < .001 | 0.83 (0.56-1.24) | 0.363 |
| 0.56-0.75 | 0.28 (0.18-0.43) | < .001 | 0.75 (0.42-1.33) | 0.329 |
| 0.76-1.00 | 0.14 (0.08-0.26) | < .001 | 0.39 (0.19-0.82) | 0.013 |
| Adjuvant treatment | 1.43 (1.23-1.66) | < .001 | 1.98 (1.67-2.36) | < .001 |
| CCI score | ||||
| 0 | Ref. | Ref. | ||
| 1 | 0.82 (0.68-0.98) | < .001 | 0.91 (0.75-1.12) | 0.386 |
| ≧ 2 | 0.48 (0.40-0.58) | < .001 | 0.54 (0.44-0.66) | < .001 |
Abbreviations: ypT, pathological tumor; ypN, pathological nodal; LAR, low anterior resection; APR, abdominoperineal resection; CRM, circumferential resection margin; LNR, lymph node ratio, CCI, Charlson Comorbidity Index.
The survival rates of rectal cancer patients according to different ypT category plus CRM combinations.
| Category | ypT | CRM | Total patient | Survival, % | P-value |
|---|---|---|---|---|---|
| All patients | < .001 | ||||
| New T0 | T0 | - | 756 | 666 (88.10) | |
| New T1 | T1 | - | 204 | 187 (91.67) | |
| New T2 | T2 | - | 1,037 | 876 (84.47) | |
| New T3 | T3 | CRM (-) | 1,986 | 1,460 (73.51) | |
| New T4A | T4A | CRM (-) | 52 | 31 (59.62) | |
| New T4B | T3 | CRM (+) | 201 | 94 (46.77) | |
| T4B | CRM (-) | ||||
| New T4C | T4A | CRM (+) | 72 | 30 (41.67) | |
| T4B | CRM (+) |
P-value was derived from Cochran-Armitage Trend Test. Abbreviations: ypT, pathological tumor; CRM, circumferential resection margin.
The performance between ypT and CRM-inclusive ypT staging system in rectal cancer patients treated with neoadjuvant chemoradiotherapy and surgery.
| Model 1 ypT based | Model 2 New category pT | ||||
|---|---|---|---|---|---|
| Variable | Adjusted OR (95% CIs) | Variable | Adjusted OR (95% CIs) | ||
| ypT | New ypT | ypT | CRM | ||
| 0 | Ref. | New T0 | 0 | Ref. | |
| 1 | 1.59 (0.91-2.77) | New T1 | 1 | 1.59 (0.91-2.76) | |
| 2 | 0.75 (0.56-1.01) | New T2 | 2 | 0.75 (0.56-1.00) | |
| 3 | 0.40 (0.31-0.53) | New T3 | 3 | CRM (-) | 0.43 (0.33-0.56) |
| 4A | 0.17 (0.10-0.30) | New T4A | 4A | CRM (-) | 0.20 (0.11-0.38) |
| 4B | 0.12 (0.08-0.19) | New T4B | 3 | CRM (+) | 0.15 (0.10-0.22) |
| 4B | CRM (-) | ||||
| New T4C | 4A | CRM (+) | 0.10 (0.06-0.18) | ||
| 4B | CRM (+) | ||||
| Harrell's C-Index (95%CI) | 0.752 (0.735-0.770) | 0.756 (0.738-0.773) | |||
| P-value | 0.034 |
Adjusted variable including age, sex, grade, pN, surgery type, LNR group, adjuvant treatment, and CCI score. Abbreviations: ypT, pathological tumor; CRM, circumferential resection margin.
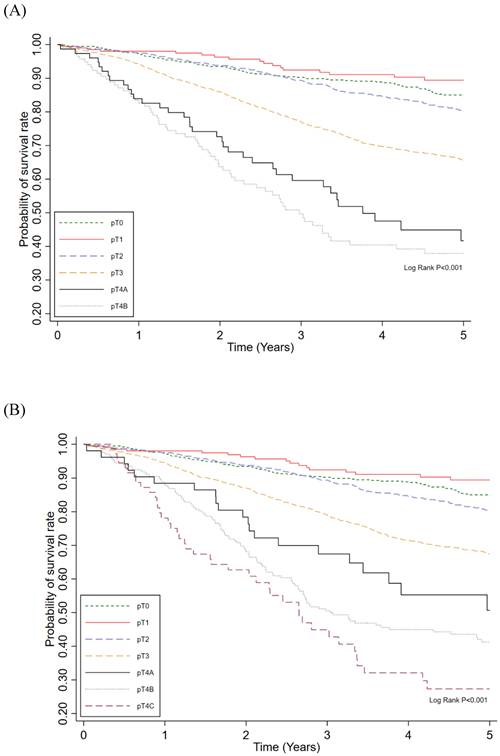
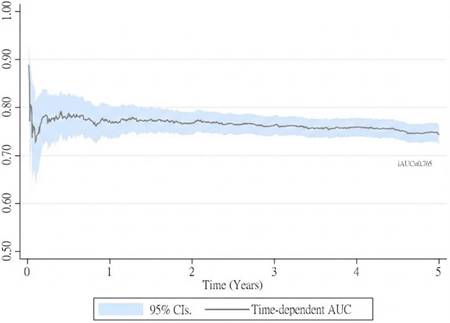
To assess the time-dependent predictive performance of the new prognostic staging system, Kaplan-Meier survival curves were plotted for 5-year survival according to the AJCC ypT staging system (Figure 2A) and the new system (Figure 2B). The new staging system demonstrated more distinct separation of survival curves, with differences between stages reaching statistical significance (P < 0.01). In addition, the cumulative predictive performance of the new system yielded an iAUC of 0.765 over the 5-year follow-up period (Figure 3).
Probability of survival rate among ypT category (A) and new category pT (B) in rectal cancer patients treated with neoadjuvant chemoradiotherapy and surgery.
The performance of new category pT derived from adjusted cox regression in rectal cancer patients treated with neoadjuvant chemoradiotherapy and surgery.
3.3CRM-inclusive ypT staging system validation
The performance of the CRM-inclusive ypT staging system during 500-sample bootstrap validation is presented in Table 5. Internal bootstrap validation yielded a C-index of 0.756 (95% CI, 0.720-0.791). We further evaluated the performance of this revised ypT category across different nodal statuses (ypN and lymph node yield), and it consistently demonstrated excellent predictive ability. Taken together, our findings indicate that incorporating CRM into the conventional AJCC ypT staging system improves the accuracy of 5-year survival prognostication in rectal cancer patients treated with nCRT and surgery.
The performance of new category pT in rectal cancer patients treated with neoadjuvant chemoradiotherapy and surgery.
| Total patient | Model 2: Harrell's C-Index | |
|---|---|---|
| Variable | Bootstrap Coef. (95% CIs) | |
| CRM-inclusive ypT staging system | 4308 | 0.756 (0.720-0.791) |
| External validation | ||
| ypN | ||
| - | 2908 | 0.722 (0.698-0.747) |
| + | 1400 | 0.750 (0.723-0.777) |
| LN removed number | ||
| < 12 | 1518 | 0.755 (0.721-0.789) |
| ≥ 12 | 2790 | 0.761 (0.726-0.795) |
4. Discussion
We propose a novel ypT staging system for rectal cancer that incorporates CRM status to improve survival stratification. Using a large, population-based cohort from Taiwan's National Cancer Registry and National Health Insurance Research Database, we analyzed patients treated with nCRT followed by surgery. To our knowledge, this is the first study to formally integrate CRM into the ypT framework. The revised system produced more clearly separated survival curves, indicating improved prognostic discrimination, which was supported by a significantly higher Harrell's C-index compared with the conventional AJCC staging system (0.756 vs. 0.752; P = 0.034). The modest absolute improvement in the C-index likely reflects that patients with ypT0-2 disease were not reclassified in the modified system, as CRM positivity was uncommon in this subgroup. Accordingly, the added prognostic value of the new staging system was mainly observed among patients with more advanced ypT categories, in whom CRM status provided further risk stratification.
Although widely adopted after neoadjuvant therapy, the ypT staging system offers only modest prognostic value, as shown by Cui et al., who found that ypT stage was not independently associated with disease-free survival in multivariate analysis [13]. A similar concern was raised by Min et al., who observed that patients with identical ypT stages could still experience differing survival outcomes depending on their tumor regression grade, particularly in the absence of nodal involvement [22]. This highlights how ypT staging fails to reflect the degree of histologic tumor response following nCRT, which has been shown to correlate with long-term survival. Given the limitations of the current ypT category, increasing efforts have been made to incorporate additional prognostic factors into the staging system. Relevant prognostic variables include TRG, CRM, lymphovascular invasion (LVI), nodal status, and tumor budding [9-12, 22-26]. KRAS mutations and radiologic evidence of EMVI have likewise been identified as prognostically relevant in recent studies [27-29]. Several studies have investigated the integration of TRG into post-treatment staging systems to enhance predictive accuracy. For example, Cui et al. analyzed 329 patients treated with nCRT and surgery at a single institution, proposing the M-TTRG system—a composite metric that combines ypT and TRG into five prognostic groups [13]. This model demonstrated superior stratification of 3-year disease-free survival compared to ypT or TRG alone. Similarly, Song et al. examined 331 patients with ypStage II/III disease and found that combining Dworak TRG (poor response: 1/2; good response: 3/4) with pathologic stage provided better prediction of 5-year disease-free survival, yielding a higher C-statistic than the conventional ypStage (0.784 vs. 0.757; P = 0.012) [30]. However, TRG assessment is inherently subjective and prone to interobserver variability, which may limit its consistency in clinical application. In contrast to these TRG-based models, our study is the first to integrate CRM status directly into the ypT staging system. By integrating an anatomically defined and consistently reported margin parameter, our CRM-integrated system enhances reproducibility and clinical applicability, making it a more practical tool for routine risk assessment and treatment planning.
CRM involvement is a well-established prognostic factor in rectal cancer, strongly associated with both recurrence and overall survival [9-12]. This association persists even in patients receiving nCRT. In a large cohort study, Park et al. reported that CRM ≤ 1 mm independently predicted worse outcomes in both nCRT and non-nCRT groups, with 5-year disease-free survival declining from 82.8% to 54.7% in non-nCRT patients and from 88.9% to 55.5% in those receiving nCRT (P < 0.001 and P = 0.001, respectively) [10]. Despite its consistent prognostic relevance, CRM status has yet to substantially influence adjuvant treatment decisions and is still primarily considered a marker of surgical quality and an adverse factor guiding postoperative management.
A similar concept has been demonstrated in head and neck squamous cell carcinoma (HNSCC), where the AJCC 8th edition incorporated extranodal extension (ENE) into the nodal category. ENE has been consistently validated as a strong adverse prognostic factor across multiple cohorts, leading to proposals for revised nodal stratification. Moreover, RTOG 9501 and EORTC 22931 showed that ENE-positive patients benefit more from concurrent chemoradiotherapy than radiotherapy alone, highlighting how key risk features can shape staging and treatment decisions [31]. Similarly, our results suggest that CRM involvement—a well-documented prognostic factor in rectal cancer—may warrant inclusion in future staging systems to enhance risk stratification and guide postoperative management.
This refined stratification enabled clearer identification of patients with unfavorable prognosis—those who might benefit from intensified adjuvant therapy or alternative systemic approaches. The conventional AJCC ypT classification, which separates ypT3 from ypT4 disease with estimated 5-year survival rates of 71.8% and 48.5%, respectively, offers only a broad risk distinction and may overlook meaningful differences among tumors with similar invasion depth. In contrast, our CRM-integrated model delineated four prognostically distinct subgroups, with 5-year survival ranging from 73.5% in new ypT3 to 41.7% in new ypT4C. This finer gradation in survival outcomes emphasizes the prognostic value of CRM and strengthens its potential role in guiding postoperative therapy selection and informing the use of novel treatment strategies, including consideration of more intensive adjuvant systemic therapy regimens or closer surveillance schedules for higher-risk subgroups.
This study has several limitations. First, CRM status was analyzed using a binary cutoff of ≤1 mm, and we did not explore alternative thresholds or continuous measurements, which may have further refined prognostic stratification. Second, in our cancer registry database, CRM assessment was limited to pathological findings instead of radiologist reading, and detailed information distinguishing positivity from tumor deposits versus direct tumor extension was not available. Future studies are warranted to address these limitations. Third, although we accounted for whether adjuvant therapy was administered, detailed information on chemotherapy regimens, dosing, treatment adherence, and molecular markers such as KRAS mutations was not available, which limited our ability to evaluate the impact of systemic treatment across subgroups. Finally, certain clinical details, such as surgical approach (open, laparoscopic, or robotic), were not available in the registry databases, and treatment strategies reflecting contemporary practice, including total neoadjuvant therapy (TNT), were not widely adopted during the study period, which may limit the generalizability of our findings to current clinical settings.
5. Conclusion
The findings from this study highlight the potential clinical utility of incorporating CRM status into ypT staging to improve risk stratification after neoadjuvant therapy. Recognizing its consistent association with survival outcomes, CRM holds potential for future incorporation of staging systems such as the AJCC, where its integration may offer a more clinically informative framework for guiding postoperative treatment decisions.
Abbreviations
LARC, locally advanced rectal cancer; NACRT, neoadjuvant chemoradiotherapy; TME, total mesorectal excision; TCR, Taiwan cancer registry; NHIRD, National Health Insurance Research Database; AJCC, American Joint Committee on Cancer; ICD-O-3, International Classification of Diseases for Oncology, 3rd Edition; CCI, Charlson comorbidity index; OR, Odds ratio; CI, confidence interval.
Supplementary Material
Supplementary figure and table.
Acknowledgements
We are grateful to Health Data Science Center, National Cheng Kung University Hospital for providing administrative and technical support.
Ethics approval and informed consent
The Institutional Review Board of the Chi Mei Medical Center approved current study (No: 11405-007), and the protocol followed the Declaration of Helsinki and its updates. Because this study has a retrospective design using the national cancer registry database with no personally identifiable information, written informed consent was not necessary.
Data availability statement
Clinicopathological datasets are available from the corresponding author upon reasonable request.
Author contributions
Study design: Chow WH, Ho CH, Chen YC, Huang HY, Yang CC. Data analysis: Ho CH, Chen YC, Yang CC. Manuscript writing and editing: Chow WH, C, Ho CH, Chen YC, Huang HY, Yang CC. All authors have read and agreed to the published version of the manuscript.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Enker WE. et al. Total mesorectal excision in the operative treatment of carcinoma of the rectum. J Am Coll Surg. 1995;181:335-46
2. MacFarlane JK. et al. Mesorectal excision for rectal cancer. The Lancet. 1993;341:457-60
3. Martling AL. et al. Effect of a surgical training programme on outcome of rectal cancer in the County of Stockholm. The Lancet. 2000;356:93-6
4. Sauer R. et al. Preoperative versus postoperative chemoradiotherapy for locally advanced rectal cancer: results of the German CAO/ARO/AIO-94 randomized phase III trial after a median follow-up of 11 years. J Clin Oncol. 2012;30:1926-33
5. Roh MS. et al. Preoperative multimodality therapy improves disease-free survival in patients with carcinoma of the rectum: NSABP R-03. J Clin Oncol. 2009;27:5124-30
6. Quirke P. et al. Effect of the plane of surgery achieved on local recurrence in patients with operable rectal cancer: a prospective study using data from the MRC CR07 and NCIC-CTG CO16 randomised clinical trial. Lancet. 2009;373:821-8
7. Kapiteijn E. et al. Preoperative Radiotherapy Combined with Total Mesorectal Excision for Resectable Rectal Cancer. New England Journal of Medicine. 2001;345:638-46
8. van Gijn W. et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer: 12-year follow-up of the multicentre, randomised controlled TME trial. Lancet Oncol. 2011;12:575-82
9. Liu Q. et al. Circumferential resection margin as a prognostic factor after rectal cancer surgery: A large population-based retrospective study. Cancer Med. 2018;7:3673-81
10. Park JS. et al. A circumferential resection margin of 1 mm is a negative prognostic factor in rectal cancer patients with and without neoadjuvant chemoradiotherapy. Dis Colon Rectum. 2014;57:933-40
11. Detering R. et al. Prognostic importance of circumferential resection margin in the era of evolving surgical and multidisciplinary treatment of rectal cancer: A systematic review and meta-analysis. Surgery. 2021;170:412-31
12. Sugimoto K. et al. Positive Circumferential Resection Margin in Rectal Cancer Is a Robust Predictor of Poor Long-term Prognosis With Clinicopathological Bias Between Groups Compensated by Propensity-score Matching Analysis. Anticancer Res. 2023;43:3623-30
13. Cui J. et al. The combination of early treatment response and ypT stage is a novel metric to stage rectal cancer patients treated with neoadjuvant chemoradiotherapy. Oncotarget. 2017;8:37845-54
14. Chiang CJ. et al. Incidence and survival of adult cancer patients in Taiwan, 2002-2012. J Formos Med Assoc. 2016;115:1076-88
15. Wang J. et al. The treatment model of stage II/III rectal cancer: A study based on the SEER database. Asian J Surg. 2023;46:2243-4
16. Chen VW. et al. Analysis of stage and clinical/prognostic factors for colon and rectal cancer from SEER registries: AJCC and collaborative stage data collection system. Cancer. 2014;120(Suppl 23):3793-806
17. Tian Y. et al. Age-adjusted charlson comorbidity index score as predictor of prolonged postoperative ileus in patients with colorectal cancer who underwent surgical resection. Oncotarget. 2017;8:20794-801
18. Hu ZD. et al. How to analyze tumor stage data in clinical research. J Thorac Dis. 2015;7:566-75
19. Harrell FE Jr. et al. Multivariable prognostic models: issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Stat Med. 1996;15:361-87
20. Heagerty PJ, Zheng Y. Survival model predictive accuracy and ROC curves. Biometrics. 2005;61:92-105
21. Steyerberg EW. et al. Internal validation of predictive models: efficiency of some procedures for logistic regression analysis. J Clin Epidemiol. 2001;54:774-81
22. Min BS. et al. Clinical Impact of Tumor Regression Grade after Preoperative Chemoradiation for Locally Advanced Rectal Cancer: Subset Analyses in Lymph Node Negative Patients. J Korean Soc Coloproctol. 2011;27:31-40
23. Fokas E. et al. Tumor Regression Grading After Preoperative Chemoradiotherapy for Locally Advanced Rectal Carcinoma Revisited: Updated Results of the CAO/ARO/AIO-94 Trial. Journal of Clinical Oncology. 2014;32:1554-62
24. CHO HJ. et al. Prognostic Significance of Clinicopathological and Molecular Features After Neoadjuvant Chemoradiotherapy in Rectal Cancer Patients. In Vivo. 2019;33:1959-65
25. Kim TH. et al. Pathologic Nodal Classification Is the Most Discriminating Prognostic Factor for Disease-Free Survival in Rectal Cancer Patients Treated With Preoperative Chemoradiotherapy and Curative Resection. International Journal of Radiation Oncology*Biology*Physics. 2010;77:1158-65
26. Trotsyuk I. et al. Tumor budding outperforms ypT and ypN classification in predicting outcome of rectal cancer after neoadjuvant chemoradiotherapy. BMC Cancer. 2019;19:1033
27. Grimminger PP. et al. Biomarkers for Cetuximab-Based Neoadjuvant Radiochemotherapy in Locally Advanced Rectal Cancer. Clinical Cancer Research. 2011;17:3469-77
28. Lord AC. et al. MRI-Diagnosed Tumor Deposits and EMVI Status Have Superior Prognostic Accuracy to Current Clinical TNM Staging in Rectal Cancer. Annals of Surgery. 2022;276:334-44
29. Chand M. et al. EMVI-positive stage II rectal cancer has similar clinical outcomes as stage III disease following pre-operative chemoradiotherapy. Annals of Oncology. 2014;25:858-63
30. Song C. et al. Impact of Tumor Regression Grade as a Major Prognostic Factor in Locally Advanced Rectal Cancer after Neoadjuvant Chemoradiotherapy: A Proposal for a Modified Staging System. Cancers (Basel). 2018 10
31. Lydiatt WM. et al. Head and Neck cancers-major changes in the American Joint Committee on cancer eighth edition cancer staging manual. CA Cancer J Clin. 2017;67:122-37
Author contact
Corresponding authors: Ching-Chieh Yang, MD, PhD, Department of Radiation Oncology, Chi Mei Medical Center, No. 901, Zhonghua Rd., Yungkang Dist., Tainan City 710, Taiwan. Tel: +88662812811 ext. 53501 E-mail: cleanclear0905com. Hsuan-Yi Huang, MD, Division of Colorectal Surgery, Department of Surgery, Chi Mei Medical Center, No. 901, Zhonghua Rd., Yungkang Dist., Tainan City 710, Taiwan. Tel: +88662812811 E-mail: crazydiamandcom.tw.

 Global reach, higher impact
Global reach, higher impact