Impact Factor ISSN: 1837-9664
J Cancer 2023; 14(11):2085-2092. doi:10.7150/jca.84854 This issue Cite
Research Paper
Prognostic Value of Lymph Node Necrosis at Different N Stages in Patients with Nasopharyngeal Carcinoma
1. Department of Laboratory Medicine, The Third Affiliated Hospital of Sun Yat-Sen University, Guangzhou 510630, China.
2. Department of Radiation Oncology, The Third Affiliated Hospital of Sun Yat-Sen University, Guangzhou 510630, China.
3. Department of Radiology, Sun Yat-sen University Cancer Center, State Key Laboratory of Oncology in South China, Collaborative Innovation Center for Cancer Medicine, Guangdong Key Laboratory of Nasopharyngeal Carcinoma Diagnosis and Therapy, Guangzhou 510060, China.
4. Radiotherapy Department of Nasopharyngeal Carcinoma, Cancer Center, The First People's Hospital of Foshan Affiliated to Sun Yat-sen University, Foshan 528000, China.
5. Department of Molecular Radiation Oncology, German Cancer Research Center, Heidelberg 69120, Germany.
6. State Key Laboratory of Oncology in South China, Sun Yat-sen University Cancer Center, Collaborative Innovation Center for Cancer Medicine, Guangdong Key Laboratory of Nasopharyngeal Carcinoma Diagnosis and Therapy, Guangzhou 510060, China.
7. Biotherapy Centre, The Third Affiliated Hospital of Sun Yat-Sen University, Guangzhou 510630, China.
*These authors contributed equally to this work.
Abstract
Background: Lymph node necrosis (LNN), including retropharyngeal nodal necrosis and cervical nodal necrosis, which is related to radiotherapy/ chemotherapy resistance, is a common phenomenon in nasopharyngeal carcinoma (NPC). This study was to assess the prognostic value of LNN at different N stages in NPC patients.
Materials and Methods: In total, 1,665 newly diagnosed NPC patients at stage TxN1-3M0 from two centers were enrolled. Univariate and multivariate models were constructed to assess the association between LNN and long-term survival outcomes. The propensity score matching method was performed to balance treatment groups for baseline characteristics.
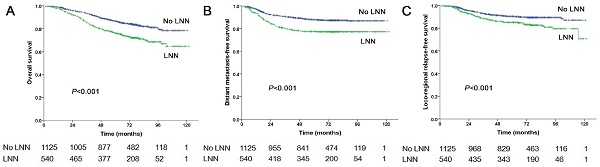
Results: Of the 1,665, 540 patients (540/1665, 32.4%) were diagnosed with LNN, of which 54.1% (292/540) patients were at stage N1, 31.3% (169/540) at stage N2, and 14.6% (79/540) at stage N3. Univariate and multivariate analyses indicated LNN as an independent predictor for progression‑free survival (PFS), overall survival (OS), distant metastasis-free survival (DMFS), and locoregional relapse-free survival (LRRFS) in stage N1-3 patients (all P<0.001). When patients were analyzed according to stage, similar findings were observed for N1 patients (all P<0.001); for N2 patients, LNN independently predicted PFS (P=0.003), OS (P=0.011), and DMFS (P=0.004), and for stage N3, LNN only independently predicted LRRFS (P=0.019). 123 pairs of patients who received induction chemotherapy plus concurrent chemoradiotherapy or only concurrent chemoradiotherapy were matched, adding induction chemotherapy improved 5-year OS, PFS and LRFFS, but the results were not statistically significant.
Conclusions: In NPC patients, LNN could independently predict poor prognosis at all N1-3 stages and at each N stage (N1 to N3). The value of adding induction chemotherapy to concurrent chemoradiotherapy in patients with LNN still requires further prospective studies.
Keywords: nasopharyngeal carcinoma, stage N1-3, lymph node necrosis, induction chemotherapy, prognosis

 Global reach, higher impact
Global reach, higher impact