Impact Factor
ISSN: 1837-9664
J Cancer 2022; 13(8):2440-2446. doi:10.7150/jca.71141 This issue Cite
Research Paper
Association between Timing and Duration of Adjuvant Chemotherapy and Colorectal Cancer Survival in Korea, 2011-2014: A Nationwide Study based on the Health Insurance Review and Assessment Service Database
1. Department of Radiation Oncology, Chung-Ang University Hospital, Seoul, South Korea.
2. Clinical Research Center, Asan Medical Center, Seoul, South Korea.
3. Kyung Hee University Medical Center, Seoul, South Korea.
4. Department of Oncology/Hematology, Kyungpook National University Medical Center, Daegu, South Korea.
5. Department of Oncology, Asan Medical Center, University, Seoul, South Korea.
6. Health Insurance Review and Assessment Service, Wonju, South Korea.
7. Quality Assessment Management Division, Health Insurance Review and Assessment Service, Wonju, South Korea.
8. Soonchunhyang University Cheonan Hospital, Cheonan, South Korea.
9. Department of Internal Medicine, Chung-Ang University College of Medicine, Seoul, South Korea.
Abstract
Background: Population-based analyses of the treatment outcomes of colorectal cancer (CRC) in Asian countries are limited. Therefore, we conducted a nationwide study to assess the relationship between the timing and duration of adjuvant chemotherapy (AC) and survival in patients with CRC in South Korea.
Methods: Data on AC from the Health Insurance Review and Assessment Service Database (HIRA) were analyzed, and the survival of patients who underwent curative-intent surgical resection for CRC between 2011 and 2014 was investigated.
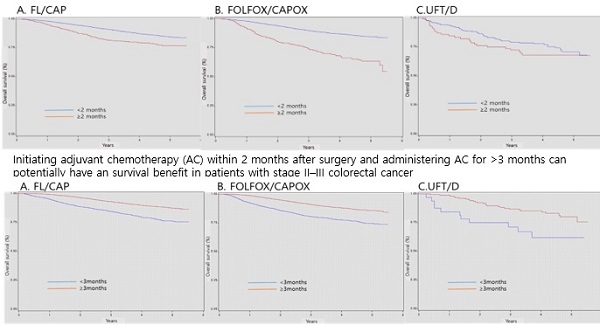
Results: From the HIRA data, 45,992 patients with stage II-III CRC were identified. Chemotherapy regimens were administered as follows: 10,640 (23.3%) received 5-fluorouracil and leucovorin/capecitabine (FL/CAP), 13,083 (28.7%) received FL/CAP plus oxaliplatin (FOLFOX/CAPOX), 299 (0.7%) received uracil and tegafur/doxifluridine (UFT/D), and 21,570 (47.3%) underwent surgery alone. Patients who did not receive AC had worse survival than those who received AC in both the colon and rectum groups (HR, 1.96, 95% CI, 1.85-2.07 and HR, 2.18, 95% CI, 2.01-2.37, respectively). Regarding patients with stage II-III CRC, AC initiation ≥ 2 months after surgery was associated with a significant decrease in overall survival (OS) (FL/CAP: HR, 1.82; 95% CI, 1.53-2.17 and FOLFOX/CAPOX: HR, 2.92; 95% CI, 2.47-3.45); however, the effects of UFT/D regimens were not statistically significant. For patients with stage II-III colon cancer, AC <3 months had lower OS (FL/CAP: HR, 3.72, 95% CI, 2.80-4.94; FOLFOX/CAPOX: HR, 2.15, 95% CI, 1.87-2.47; and UFT/D: HR, 1.74, 95% CI, 0.56-5.41). In terms of patients with stage II-III rectal cancer, AC <3 months, regardless of chemotherapy regimens, had a significant lower survival (FL/CAP: HR, 1.91, 95% CI, 1.66-2.20; FOLFOX/CAPOX: HR, 2.20, 95% CI, 1.75-2.77; and UFT/D: HR, 3.71, 95% CI, 1.45-9.44).
Conclusions: Postoperative time to initiation and duration of AC were associated with survival. Based on our results, initiating AC within 2 months after surgery and administering AC for >3 months can potentially have an OS benefit in patients with stage II-III CRC.
Keywords: colorectal cancer, adjuvant chemotherapy, timing, duration

 Global reach, higher impact
Global reach, higher impact