Impact Factor ISSN: 1837-9664
J Cancer 2022; 13(7):2159-2170. doi:10.7150/jca.70323 This issue Cite
Research Paper
Biliverdin reductase B impairs cholangiocarcinoma cell motility by inhibiting the Notch/Snail signaling pathway
1. Department of Nuclear Medicine, Zhongshan Hospital, Fudan University, 180 Fenglin Road, Shanghai 200032, China.
2. Department of General Surgery, Zhongshan Hospital, Fudan University, 180 Fenglin Road, Shanghai 200032, China.
3. Biliary Tract Disease Center of Zhongshan Hospital, Fudan University, 180 Fenglin Road, Shanghai 200032, China.
4. Cancer Center, Zhongshan Hospital, Fudan University, 180 Fenglin Road, Shanghai 200032, China.
5. Biliary Tract Disease Institute, Fudan University, Shanghai 200032, China.
6. Shanghai Biliary Tract Minimal Invasive Surgery and Materials Engineering Research Center, Shanghai 200032, China.
7. Basic Medical Institute; Key Laboratory of Cell Differentiation and Apoptosis of Chinese Ministry of Education, Shanghai Jiao Tong University School of Medicine, 280 South Chongqing Road, Shanghai 200025, China.
8. Fudan University Shanghai Cancer Center & Institutes of Biomedical Sciences; Shanghai Medical College, Fudan University, Shanghai 200032, China.
9. Department of General Surgery, Shanghai Xuhui Central Hospital, Zhongshan-Xuhui Hospital, Fudan University, Shanghai 200031, China.
*These authors contributed equally to this work.
Abstract
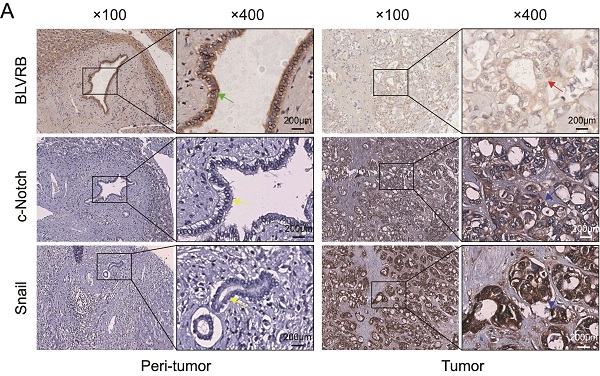
Cholangiocarcinoma (CCA) is one of the most lethal types of solid tumors worldwide. Lymph node metastasis is common in the early stage, which is associated with recurrence and reduced survival time after CCA resection. The molecular pathogenesis of CCA is complex and requires extensive investigation. It involves multiple genomic alterations and the dysregulation of signaling pathways. Biliverdin reductase B (BLVRB) is a non-redundant NAD(P)H-dependent biliverdin reductase that regulates cellular redox status by reducing biliverdin to bilirubin. This study aimed at describing the biological functions and molecular mechanisms of BLVRB in human CCA. Prognostic clinical data showed that low expression BLVRB was associated with poor prognosis and lymph node metastasis. BLVRB depletion accelerated epithelial-mesenchymal transition (EMT), cell migration and invasion. In contrast, BLVRB overexpression was associated with reduced EMT and cell migration and invasion in CCA. BLVRB suppression activated Notch signaling, and activated c-Notch enhanced EMT by upregulating Snail expression levels, thereby increasing cell migration and invasion in CCA. Our results identified an unexpected function of BLVRB in CCA migration and invasion through the regulation of Notch/Snail signaling.
Keywords: Cholangiocarcinoma, BLVRB, EMT, Snail, Notch

 Global reach, higher impact
Global reach, higher impact