Impact Factor ISSN: 1837-9664
J Cancer 2022; 13(1):124-133. doi:10.7150/jca.63578 This issue Cite
Research Paper
Akkermansia muciniphila administration exacerbated the development of colitis-associated colorectal cancer in mice
1. Gannan Medical University, Ganzhou, Jiangxi, 341000, China.
2. The Fifth People's Hospital of Jinan, Jinan, Shandong, 250000, China.
3. School of Basic Medicine, Gannan Medical University, Ganzhou, Jiangxi 341000, China.
4. Center for Immunology, Key Laboratory of Prevention and Treatment of Cardiovascular and Cerebrovascular Diseases, Ministry of Education, Gannan Medical University, Ganzhou, Jiangxi 341000, China.
Abstract
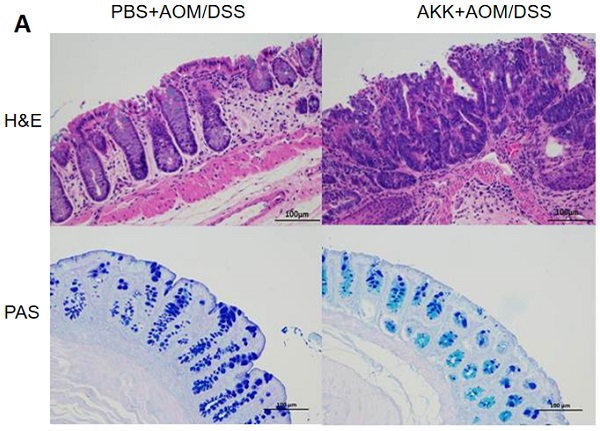
Colorectal cancer (CRC) is one of the most common digestive tract malignancies and inflammation and gut microbiota are well-known key factors to influence CRC development. Akkermansia mucinipila is an important gram-negative anaerobic bacterium that can degrade mucin in gut. Previous studies suggested that A. muciniphila may affect inflammation and cell proliferation, but the relationship between A. muciniphila and CRC is not clarified. Here C57BL/6 mice were administrated with A. muciniphila or PBS and then treated with azoxymethane (AOM)/dextran sodium sulphate (DSS) to induce CRC. The mice receiving A. muciniphila administration had more serious weight loss, shorter colon length and more intestinal tumors than those receiving PBS administration after AOM/DSS treatment. More colon damage and less goblet cells were also observed in A. muciniphila treated mice. Furthermore, A. muciniphila administration induced more Ki67+ proliferating cells, higher PCNA expression and elevated gene expression of proliferation-associated molecules including Snrpd1, Dbf4 or S100A9. At early stage of CRC development, in comparison with controls, the mice receiving A. muciniphila administration also had more body weight loss and shorter colon length, as well as higher gene expression of inflammatory cytokines. Furthermore, the in vitro experimental results showed that the co-culture of colon epithelial cells with A. muciniphila enhanced the cell proliferation and gene expression of proliferation-associated molecules. Therefore, A. mucinipila may promote the formation of CRC in mice through increasing the early level of inflammation and the proliferation of intestinal epithelial cells.
Keywords: Akkermansia mucinipila, Colorectal cancer, Cell proliferation, Inflammatory cytokines

 Global reach, higher impact
Global reach, higher impact