Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(17):5355-5364. doi:10.7150/jca.60682 This issue Cite
Research Paper
Metronomic vinorelbine is an excellent and safe treatment for advanced breast cancer: a retrospective, observational study
1. Division of Hematology-Oncology, Department of Internal Medicine, Kaohsiung Chang Gung Memorial Hospital, Kaohsiung 833, Taiwan.
2. Chang Gung University, College of Medicine, Tao-Yuan 333, Taiwan
3. Department of Hematology-Oncology, E-Da Cancer Hospital, Kaohsiung 822, Taiwan.
4. College of Medicine, I-Shou University, Kaohsiung 822, Taiwan.
5. Department of Surgery, E-Da Cancer Hospital, Kaohsiung 822, Taiwan.
6. Department of Surgery, Kaohsiung Chang Gung Memorial Hospital, Kaohsiung 833, Taiwan.
7. Division of General Surgery, Department of Surgery, E-Da Hospital, Kaohsiung, 822 Taiwan.
* Equally contribution.
Abstract
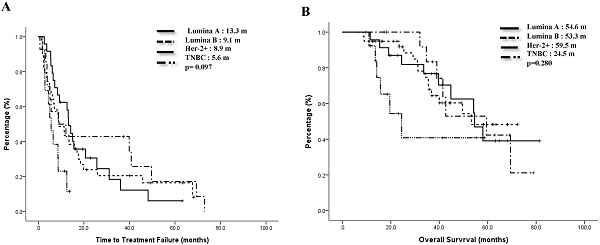
Advanced breast cancer (ABC) has become a chronic disease. In such a situation, an effective therapy with low toxicities and economically acceptable is needed. Metronomic vinorelbine (mVNR) has been proved to be effective on the control of MBC. The aim of this study is to evaluate the efficacy and safety of mVNR as the salvage therapy for patients with ABC. Oral vinorelbine (VNR) was administered at 70 mg/m2, fractionated on days 1, 3, and 5, for 3 weeks on and 1 week off. Once the mVNR was combined with trastuzumab, or was combined with bevacizumab, the schedule was changed to 2 weeks on and 1 week off. Clinical data of patients with ABC who had received treatment with mVNR and tumor characteristics were collected and analyzed. From Mar. 2013 to Dec, 2020, there were 90 patients with ABC received mVNR. The overall response rate was 53.3% and overall disease control rate (DCR) was 78.9% in this study, including 4 (4.4%) cases reached complete response, 44 (48.9%) cases reached partial response and 23 (25.6%) cases were table disease. The median time to treatment failure (TTF) of the Lumina A patients was 13.3 months, Lumina B patients was 9.1 months, Her-2 enrich patients was 8.9 months, and triple negative breast cancer (TNBC) patients was 5.6 months. Median overall survival time for Lumina A, Lumina B, Her-2 enrich and TNBC were 54.6 months, 53.3 months, 59.5 months and 24.5 months separately. Side effects were minimal and manageable. Metronomic VNR can be an effective treatment for ABC either works as a switch maintenance or salvage therapy. In combination with target therapy or hormonal therapy, mVNR can further improve TTF and DCR with minimal toxicities. Further study should focus on the optimal dosage, schedule and combination regimen.
Keywords: metastatic breast cancer, advanced breast cancer, metronomic chemotherapy, vinorelbine, effect.

 Global reach, higher impact
Global reach, higher impact