Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(16):4819-4829. doi:10.7150/jca.58670 This issue Cite
Research Paper
Upregulation of V-ATPase by STAT3 Activation Promotes Anoikis Resistance and Tumor Metastasis
1. Guangdong Immune Cell therapy Engineering and Technology Research Center, Center for Protein and Cell-based Drugs, Institute of Biomedicine and Biotechnology, Shenzhen Institute of Advanced Technology, Chinese Academy of Sciences, Shenzhen, 518055, China.
2. University of Chinese Academy of Sciences, Beijing, 100864, China.
3. School of Basic Medical Science, Jinzhou Medical University, Jinzhou, 121000, China.
Abstract
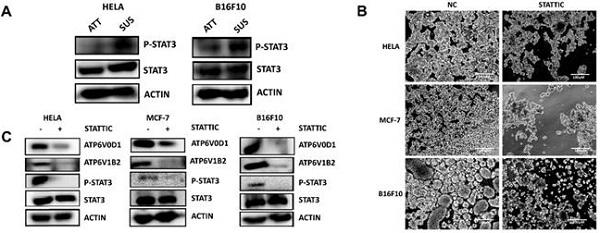
Most cancer mortality results from metastatic tumor cells and not the localized tumor. Overcoming anoikis is one of the most important steps for detached tumor cells to migrate and metastasize. However, the molecular mechanisms remain to be fully deciphered. Herein, our study revealed upregulation of vacuolar ATPase (V-ATPase) in cancer cells during ECM detachment plays a key role in anoikis evasion. V-ATPase is an enzyme complex that utilizes energy from ATP hydrolysis to maintain cellular homeostasis and had been reported to enhance cancer progression. In this study, V-ATPase inhibition sensitized human cervical cancer, breast cancer, and murine melanoma cells to anoikis via increased ROS production, accumulation of misfolded protein, and impaired pulmonary metastasis in vivo. Scavenging ROS restored anoikis resistance and clearance of misfolded protein accumulation in the tumor cells. Mechanistically, STAT3 upregulates V-ATPase expression while blockade of STAT3 activity repressed V-ATPase expression in these tumor cells as well as sensitized cells to anoikis, increased ROS production, and misfolded protein accumulation. Altogether, our data demonstrate an unreported role of STAT3 in mediating the upregulation of V-ATPase to promote anoikis resistance, thus provides an alternative option to target cancer metastasis.
Keywords: Anoikis, Metastasis, V-ATPase, STAT3, Misfolded protein accumulation

 Global reach, higher impact
Global reach, higher impact