Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(14):4372-4378. doi:10.7150/jca.57413 This issue Cite
Research Paper
Network Meta-analysis Comparing Efficacy, Safety and Tolerability of Anti-PD-1/PD-L1 Antibodies in Solid Cancers
Department of Medical Oncology and Hematology, Princess Margaret Cancer Center, University Health Network, Faculty of Medicine, University of Toronto, Toronto, Canada.
Abstract
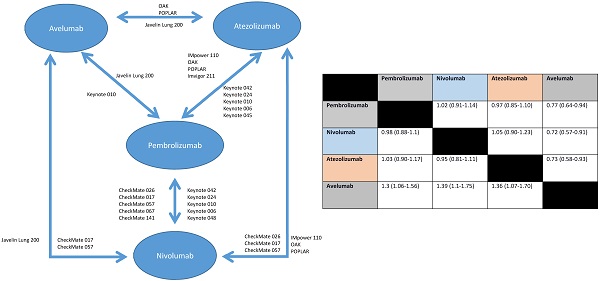
Background: Multiple anti-PD-1/PD-L1 antibodies have been approved, and in some diseases, there is a choice of more than one. Comparative efficacy, safety and tolerability are unknown.
Methods: Randomized trials (RCTs) supporting the registration of single agent anti-PD1 or anti-PDL1 inhibitors between 2015-2019 were identified. We extracted the hazard ratio (HR) for overall survival (OS) and calculated the odds ratio (OR) for commonly reported safety and tolerability outcomes. We then performed a network meta-analysis, reporting multiple pair-wise comparisons between different anti-PD-1/PD-L1 antibodies.
Results: Sixteen RCTs comprising 10673 patients were included; 10 in non-small-cell lung cancer, 2 in melanoma, 2 in head and neck squamous cell carcinoma and 2 in urothelial cancer. Compared to pembrolizumab, efficacy was similar for nivolumab (HR: 1.02 95% CI: 0.91-1.14) and for atezolizumab (HR: 0.97 95% CI: 0.85-1.10), however, avelumab appeared inferior (HR: 1.30, 95% CI: 1.06-1.56). Pembrolizumab showed similar odds of serious adverse events (SAEs) as nivolumab (OR: 1.12, 95% CI: 0.56-2.27) and atezolizumab (OR: 1.05, 95% CI: 0.55-2.04). Compared to nivolumab, atezolizumab was associated with more SAEs (OR: 2.14, 95% CI: 1.47-3.12). Avelumab had the lowest odds of grade 3-4 adverse events compared to pembrolizumab (OR: 0.42, 95% CI: 0.24-0.74), nivolumab (OR: 0.38, 95% CI: 0.24-0.62) and atezolizumab (OR: 0.21, 95% CI: 0.14-0.33). The odds of treatment discontinuation without progression were similar between nivolumab and atezolizumab (OR: 1.20, 95% CI: 0.73-2.00), and between pembrolizumab and nivolumab (OR: 1.35, 95% CI: 0.83-2.17), but was higher with atezolizumab compared to nivolumab (OR: 2.56, 95% CI: 1.29-5.00). Pembrolizumab was associated with higher OR of immune-related adverse events (IRAEs) compared to nivolumab (OR: 2.12, 95% CI: 1.49-3.03) and atezolizumab (OR: 1.63, 95% CI: 1.09-2.43).
Conclusions: Pembrolizumab, nivolumab, and atezolizumab have similar efficacy. Avelumab appears less efficacious. Safety and tolerability seem better with avelumab, but worse with atezolizumab and pembrolizumab.
Keywords: Immunotherapy, Programmed Cell Death 1 Receptor, Review, Clinical Trial, Phase III as Topic

 Global reach, higher impact
Global reach, higher impact