Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(14):4288-4294. doi:10.7150/jca.54169 This issue Cite
Research Paper
Hereditary cancer syndrome-associated pathogenic variants are common in patients with hematologic malignancies subsequent to primary solid cancer
1. Department of Laboratory Medicine, Sheikh Khalifa Specialty Hospital, Ras Al Khaimah, United Arab Emirates.
2. Division of hematology, Department of internal medicine, Yonsei University College of Medicine, Seoul, Korea.
3. Department of Laboratory Medicine, Yonsei University College of Medicine, Seoul, Korea.
4. Department of Laboratory Medicine, School of Medicine, Kyung Hee University, Seoul, Korea.
5. SD Genomics Co., Ltd., Seoul, Korea.
#Both authors have equally contributed to this article.
*Current affiliation: Genoplan Korea, Inc., Seoul. Korea
Abstract
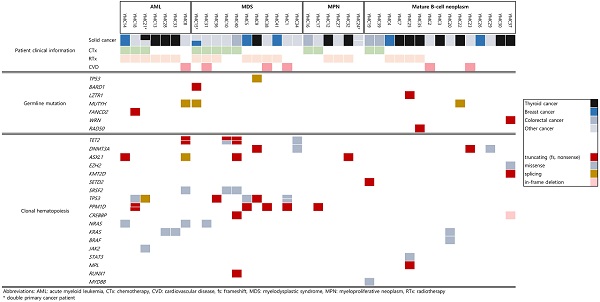
Background: As the number of long-term survivors of solid cancers keeps increasing, risk assessment of secondary hematologic malignancies is important for the prognosis of the patient. Germline genetic predisposition to secondary hematologic malignancy has been studied widely in myeloid neoplasms and rarely in lymphoid neoplasms. This study aimed to profile the mutational spectrums of patients with subsequent lymphoid tissue neoplasm to shed some light on the understudied area.
Methods: In total, 39 patients who had primary solid cancer and subsequent hematologic malignancies were enrolled. We performed two next-generation sequencing (NGS) panel tests encompassing hereditary cancer predisposition genes and genes related to clonal hematopoiesis of indeterminate potential (CHIP). All statistical analyses were performed using R 3.5.1.
Results: We found 8 of 39 patients with germline mutations in cancer predisposition genes; 4 of 18 patients had therapy-related myeloid neoplasms (22.2%); and 4 of 15 patients had secondary lymphoid malignancies (26.7%). Notably, of 14 patients who initially suffered from thyroid cancer, 5 patients (35.7%) had germline mutations. Malignancy of lymphoid tissue showed no association with radioactive iodine therapy but was observed to a greater extent in germline mutation-positive thyroid cancer patients regardless of their history of treatment. We observed that 24 of 39 patients (61.5%) were CHIP carriers. Patients who had secondary lymphoid malignancy were less likely to have CHIP than those who had myeloid malignancy.
Conclusions: In patients with primary solid cancer who are planning to undergo cytotoxic chemotherapy, radiotherapy, or radioactive iodine therapy, an initial assessment with germline mutation testing using an expanded NGS panel, including low, moderate, and high-risk cancer-associated genes, and somatic CHIP mutation testing can screen the patients who are at risk of developing therapy-related myeloid and lymphoid malignancies. Through careful screening and monitoring throughout the treatment process, patients can benefit from the early detection of secondary malignancies and receive proper treatment.
Keywords: hematologic malignancy, germline predisposition to cancer, clonal hematopoiesis of indeterminate potential, next-generation sequencing, therapy-related myeloid neoplasms

 Global reach, higher impact
Global reach, higher impact