Impact Factor ISSN: 1837-9664
J Cancer 2021; 12(11):3164-3179. doi:10.7150/jca.48661 This issue Cite
Research Paper
Signature of gene aberrant alternative splicing events in pancreatic adenocarcinoma prognosis
1. Department of General Surgery, the First Affiliated Hospital of Soochow University, Suzhou, Jiangsu, 215006, China.
2. Pancreatic Disease Research Centre, The First Affiliated Hospital of Soochow University, Suzhou, Jiangsu, 215006, China.
3. Department of Plastic Surgery, Suzhou Municipal Hospital, Suzhou, Jiangsu, 215006, China.
4. Department of Oncology, the First Affiliated Hospital of Soochow University, Suzhou, Jiangsu, 215006, China.
* Jun Yao and Yu-Chen Tang contributed equally to this work.
Abstract
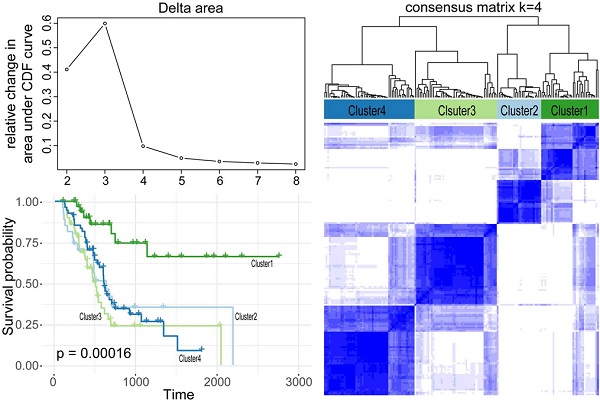
Alternative splicing (AS), as an effective and universal mechanism of transcriptional regulation, is involved in the development and progression of cancer. Therefore, systematic analysis of alternative splicing in pancreatic adenocarcinoma (PAAD) is warranted. The corresponding clinical information of the RNA-Seq data and PAAD cohort was downloaded from the TCGA data portal. Then, a java application, SpliceSeq, was used to evaluate the RNA splicing pattern and calculate the splicing percentage index (PSI). Differentially expressed AS events (DEAS) were identified based on PSI values between PAAD cancer samples and normal samples of adjacent tissues. Kaplan-Meier and Cox regression analyses were used to assess the association between DEAS and patient clinical characteristics. Unsupervised cluster analysis used to reveal four clusters with different survival patterns. At the same time, GEO and TCGA combined with GTEx to verify the differential expression of AS gene and splicing factor. After rigorous filtering, a total of 45,313 AS events were identified, 1,546 of which were differentially expressed AS events. Nineteen DEAS were found to be associated with OS with a five-year overall survival rate of 0.946. And the subtype clusters results indicate that there are differences in the nature of individual AS that affect clinical outcomes. Results also identified 15 splicing factors associated with the prognosis of PAAD. And the splicing factors ESRP1 and RBM5 played an important role in the PAAD-associated AS events. The PAAD-associated AS events, splicing networks, and clusters identified in this study are valuable for deciphering the underlying mechanisms of AS in PAAD and may facilitate the establishment of therapeutic goals for further validation.
Keywords: genomic analysis, alternative splicing, pancreatic adenocarcinoma, risk model, alternative splicing factors.

 Global reach, higher impact
Global reach, higher impact