3.2
Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(10):2975-2981. doi:10.7150/jca.56207 This issue Cite
Research Paper
Lower-dose decitabine improves clinical response compared with best supportive care in lower-risk MDS patients: a prospective, multicenter phase 2 study
1. MDS Center, Department of Hematology, the First Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou 310003, Zhejiang Province, China.
2. Key Laboratory of Hematologic Malignancies of Zhejiang Province, Hangzhou 310009, Zhejiang Province, China.
3. Department of Hematology, Tongde Hospital of Zhejiang Province, Hangzhou 310012, Zhejiang Province, China.
4. Department of Hematology, Zhuji People's Hospital of Zhejiang Province, Zhuji 311800, Zhejiang Province, China.
5. Institute of Hematology, the First Hospital of Jiaxing City in Zhejiang Province, Jiaxing 314001, Zhejiang Province, China.
Abstract
Purpose: To explore the efficacy and safety of lower-dose decitabine in patients with lower-risk MDS, a prospective multicenter phase II study was conducted to compare decitabine with the best supportive care (BSC).
Methods: Patients diagnosed with lower-risk MDS from September 2013 to August 2018 were assigned to the decitabine group or the BSC group. Decitabine (12 mg/m2/day) was administered over 1 hour/day for 5 consecutive days in a 4-week cycle. BSC, including growth factors, transfusion, thalidomide, lenalidomide, and immunosuppressive agents were given consecutively. The endpoints included the proportion of patients who achieved overall response (OR) in the first 2 or 3 courses, event-free survival (EFS), and overall survival (OS).
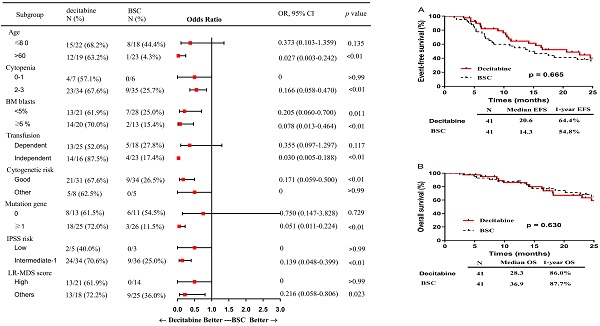
Results: A total of recruited 82 patients were analyzed. In the decitabine group, 65.9% (27/41) achieved OR after 2 or 3 cycles of treatment, compared with 22.0% (9/41) in the BSC group (p <0.01). Besides, 44.0% (11/25) in the decitabine group became independent of RBC/Platelets transfusion, compared with 27.8% (5/18) in the BSC group. Patients with gene mutation and treated with decitabine achieved a higher OR rate, compared with those without gene mutation [72.0% (18/25) vs 11.5% (3/26), p <0.01]. There was no significant difference in the median EFS between the decitabine and BSC groups (20.6 vs 14.3 months respectively, p = 0.665). In the decitabine group, the most significant adverse events were infections of any grades or neutropenic fever (46.3%, 19/41) and one patient (4.2%) died of acute cerebral infarction within 6 weeks of treatment.
Conclusion: Lower-dose decitabine demonstrated promising clinical response with acceptable toxicity profiles in patients with low- and intermediate 1-risk MDS. A higher response rate to decitabine was observed in patients with mutated genes. Therefore, lower-dose decitabine can be advocated for patients with low-risk MDS and mutated genes.
Keywords: myelodysplastic syndromes, lower risk, hypomethylating

 Global reach, higher impact
Global reach, higher impact