Impact Factor ISSN: 1837-9664
J Cancer 2021; 12(6):1815-1825. doi:10.7150/jca.51099 This issue Cite
Research Paper
Androgen Plays a Carcinogenic Role in EOC via the PI3K/AKT Signaling Pathway in an AR-Dependent Manner
1. Department of Anatomy, Hebei Medical University, Shijiazhuang, China
2. Department of Obstetrics and Gynecology, The Second Hospital of Hebei Medical University, Shijiazhuang, China
3. Neuroscience Research Center, Hebei Medical University, Shijiazhuang, China.
4. Hebei Key Laboratory of Neurodegenerative Disease Mechanism, Shijiazhuang, China.
Abstract
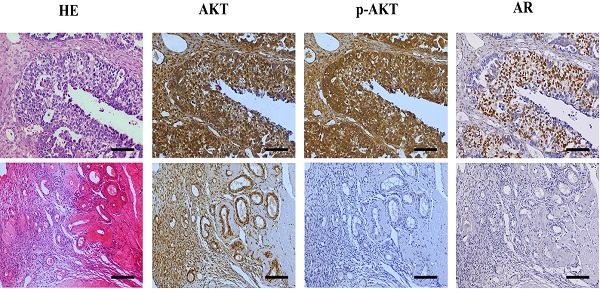
Background: Epithelial ovarian cancer (EOC) is one of the most common gynecological cancers with the highest mortality rate. Studies indicate that androgens contribute to initiation or progression of EOC through poorly understood mechanisms, however, in the phase II clinical studies of antiandrogen therapy for EOC, neither flutamide nor bicalutamide showed good antitumor effects. Based on the contradictions, the purpose of this study was to explore the role of androgen receptor (AR) in the androgen pathogenesis of EOC and the possible mechanism, and further to find an indicator to screen the anti-androgen therapy sensitive cases.
Methods: In this study, 70 EOC biopsies and 17 para-cancerous tissues with complete medical information were collected and analyzed. The expression of the androgen receptor (AR) was detected by immunohistochemistry. In addition, ovarian cancer cell lines were used for in vitro studies to further explore the role of androgen in cell proliferation and the possible mechanisms.
Results: The results showed that the expression of AR in ovarian cancer tissues was significantly elevated compared to the para-cancerous tissues, particularly in low-grade EOC, and the presence of high AR expression often suggested a worse prognosis. The in vitro study indicated that testosterone promoted the proliferation of the AR-positive SKOV3 cell line, which could be blocked by flutamide, but not in the AR-negative A2780 cell line. Next, we showed that testosterone-promoted proliferation in SKOV3 cells was abolished after we knocked out the AR. The mechanism studies revealed that the p-AKT expression in the ovarian cancer tissue was increased compared to the para-cancerous tissues, following a pattern similar to the increase of AR expression. Furthermore, the deletion and overexpression of SKOV3 cells' ARs lead to corresponding changes in the p-AKT levels. In addition, the BEZ235, an inhibitor of the PI3K/AKT signaling pathway blocked the proliferative effect of testosterone in SKOV3 cells.
Conclusion: We showed that testosterone was able to promote the proliferation of ovarian cancer cells through activating the PI3K/AKT signaling pathway in an AR dependent manner and AR may be a screening indicator for anti-androgen therapy sensitive cases of EOC.
Keywords: ovarian carcinoma, androgen receptor (AR), tissue microarray (TMA), proliferation promotion, PI3K/AKT signalling pathway

 Global reach, higher impact
Global reach, higher impact