Impact Factor ISSN: 1837-9664
J Cancer 2021; 12(6):1770-1778. doi:10.7150/jca.50371 This issue Cite
Research Paper
Preoperative Portal Vein Embolization for Liver Resection: An updated meta-analysis
1. Department of Hepatobiliary and Pancreatic Surgery, The Second Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou, Zhejiang 310009.
2. Key Laboratory of Precision Diagnosis and Treatment for Hepatobiliary and Pancreatic Tumor of Zhejiang Province, Hangzhou, Zhejiang 310009.
3. Research Center of Diagnosis and Treatment Technology for Hepatocellular Carcinoma of Zhejiang Province, Hangzhou, Zhejiang 310009.
4. Clinical Medicine Innovation Center of Precision Diagnosis and Treatment for Hepatobiliary and Pancreatic Disease of Zhejiang University, Hangzhou, Zhejiang 310009.
5. Clinical Research Center of Hepatobiliary and Pancreatic Diseases of Zhejiang Province, Hangzhou, Zhejiang 310009.
#These authors contributed equally to this work.
Abstract
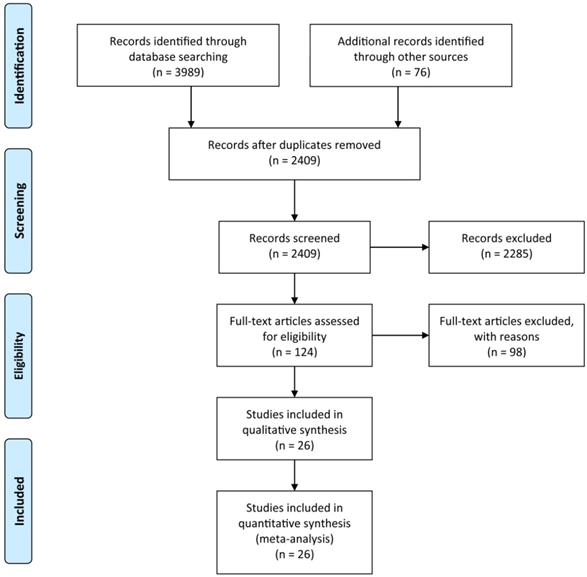
Background: Portal vein embolization (PVE) is performed before major liver resection to increase liver volume remnant, controversy remains on the adverse effect of PVE on liver tumor patients. The current study highlighted the effect of PVE on the degree of hypertrophy of future liver remnant (FLR) and summarized PVE-related complications, aiming to provide a guideline for surgeons.
Methods: A search of current published studies on PVE was performed. Meta-analysis was conducted to assess the effect of PVE on hypertrophy of FLR and summarized PVE-related complications.
Results: 26 studies including 2335 patients were enrolled in the meta-analysis. All enrolled studies reported data regarding FLR hypertrophy rate, pooled effect size (ES) for FLR hypertrophy rate using a fixed-effect model was 0.105 (95%CI: 0.094-0.117, p=0.000), indicating PVE is favored in inducing FLR hypertrophy. Metatrim method indicated no obvious evidence of publication bias in the present meta-analysis. 247 (10.6%) patients exhibited PVE-related complications, receiving expectant treatment without affecting planned liver resection. Total 1782 patients (76%) underwent a subsequent liver resection after PVE, which is an encouraging result comparing with traditional resection rate in liver tumor patients.
Conclusions: PVE is a safe and effective procedure with a low occurrence of related complications for inducing sufficient hypertrophy of FLR in liver tumor patients, which could elevate the resection rate of liver tumor patients. Careful patient cohort selection is crucial to avoid overuse of PVE in technically resectable patients. Further multiple central clinical trials are conducive to select optimal patient cohorts and provide a guideline for surgeons.
Keywords: portal vein embolization, liver tumor, future liver remnant, liver regeneration, liver resection

 Global reach, higher impact
Global reach, higher impact