Impact Factor ISSN: 1837-9664
J Cancer 2021; 12(5):1270-1283. doi:10.7150/jca.51621 This issue Cite
Research Paper
A pharmacogenetics study of platinum-based chemotherapy in lung cancer: ABCG2 polymorphism and its genetic interaction with SLC31A1 are associated with response and survival
1. Center for Medical Research and Innovation, Shanghai Pudong Hospital and Pudong Medical Center, Shanghai Medical College, Fudan University, Shanghai, China.
2. Ministry of Education Key Laboratory of Contemporary Anthropology and Department of Anthropology and Human Genetics, School of Life Sciences, Fudan University, Shanghai, China.
3. Department of Respiratory and Critical Care Medicine, Changhai Hospital, the Second Military Medical University, Shanghai, China.
4. Department of Pulmonary Medicine, Zhongshan Hospital of Fudan University, Shanghai, China.
5. Department of Pneumology, Chest Hospital, Shanghai Jiao Tong University, Shanghai, China.
6. Department of Cardiothoracic Surgery, Changzheng Hospital of the Second Military Medical University, Shanghai, China.
7. Department of Clinical Pharmacology, Xiangya Hospital; Hunan Key Laboratory of Pharmacogenomics, Institute of Clinical Pharmacology, Central South University, Changsha, China.
Abstract
Objective: The expression and function of platinum transporters affect drug tissue concentration and therapeutic effects. We had previously characterized functional variant of platinum intake transporter SLC31A1 gene. We aimed to investigate the association of platinum efflux transporter gene ABCG2 polymorphism and combined ABCG2 and SLC31A1 polymorphisms with clinical outcomes of NSCLC patients receiving platinum-based chemotherapy.
Methods: We genotyped thirteen tagging and functional SNPs of ABCG2 in 1004 patients, and assessed their association with response, toxicity and survival using unconditional logistic regression and Cox proportional hazards regression analyses respectively.
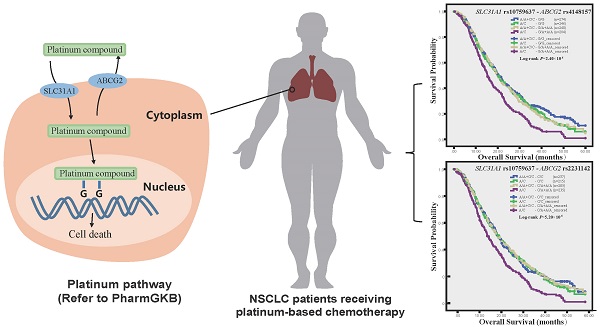
Results: Nonsynonymous rs2231142 (odds ratio [OR] 2.07; 95 % confidence interval [CI] 1.26-3.63), rs1871744 (OR 0.60; 95 % CI 0.42-0.87) and their haplotype and diplotype were associated with objective response. Rs4148157 was associated with shorter overall survival (Log-rank P = 0.002; hazard ratio [HR] 1.22; 95 % CI 1.05-1.42). Furthermore, the combined SLC31A1 rs2233914 and ABCG2 rs1871744 genotype was significantly associated with poor response (OR 0.31; 95 % CI 0.17-0.56; Pinteraction = 0.003). And the combined genotypes of the functional rs10759637 of SLC31A1 and the nonsynonymous rs2231142 (Log-rank P = 5.20×10-5; HR 1.47; 95 % CI 1.19-1.81; Pinteraction = 0.007) or linked rs4148157 of ABCG2 were significantly associated with poor survival.
Conclusion: This study reveals divergent association of ABCG2 polymorphism with response and survival of NSCLC patients receiving platinum-based chemotherapy, demonstrates the combined effects of functional variants of ABCG2 and SLC31A1 on clinical outcomes, and highlights pharmacogenetic relevance of platinum transporter genes interaction.
Keywords: NSCLC, pharmacogenetics, platinum transporter, ABCG2, SLC31A1, SNP

 Global reach, higher impact
Global reach, higher impact