Impact Factor ISSN: 1837-9664
J Cancer 2020; 11(21):6348-6355. doi:10.7150/jca.44431 This issue Cite
Research Paper
Pristimerin induces apoptosis and inhibits proliferation, migration in H1299 Lung Cancer Cells
1. Guangdong Provincial Key Laboratory of Molecular Target & Clinical Pharmacology, School of Pharmaceutical Sciences and the Fifth Affiliated Hospital, Guangzhou Medical University, Guangzhou 511436, P. R. China.
2. School of Chinese Medicine, Hong Kong Baptist University, Hong Kong, P. R. China.
3. Guangzhou Institute of Pediatrics/Guangzhou Women and Children's Medical Center, Guangzhou Medical University, Guangzhou 510623, P. R. China.
4. Institute of Immunology and School of Medicine, Shanxi Datong University, Datong 037009, P. R. China.
*These authors contributed equally to this work.
Received 2020-1-31; Accepted 2020-7-26; Published 2020-9-2
Abstract
Background: The natural occurring pristimerin, a quinonemethide triterpenoid, is extracted from a variety of species of the Celastraceae and Hippocrateaceae family. This research investigated the in vitro anti-cancer potential of pristimerin on NSCLC cells NCI-H1299 and elucidated the molecular mechanism.
Methods: Cell growth inhibition by pristimerin was assessed using the MTT assay. Apoptosis was detected using the Annexin V/propidium iodide (PI) test. The colony forming assay was used to investigate the anti-proliferative effects of pristimerin. Wound healing assay and the transwell cell migration assay were utilized to determine the inhibitory effects of migration and invasion, respectively. Western blot was used to detect the protein expression, and real-time-quantitative (RT-q) PCR was used to analyze the mRNA expression.
Results: The results showed that pristimerin inhibited the proliferation of H1299 cells with an IC50 value of 2.2 ± 0.34 µM and induced apoptosis in a dose-dependent manner. The colony formation ability was reduced in a dose-dependent manner. A marked inhibition of migration and invasion against H1299 cells was observed in a dose- or time-dependent manner. Moreover, the decreased protein levels of vimentin, F-actin, integrin β1, matrix metalloproteinase (MMP2) and Snail revealed the potential inhibition of epithelial-to-mesenchymal transition (EMT). The regulated mRNA levels of integrin β1, MMP2 and Snail indicated the great potential in the treatment of NSCLC.
Conclusion: In conclusion, our study demonstrated that pristimerin suppressed NSCLC cells NCI-H1299 in vitro, exhibited potent activities of proliferation inhibition and apoptosis induction. Furthermore, the treatment of pristimerin decreased migration and invasion of H1299, which was correlated with EMT-related proteins and mRNA.
Keywords: Anti-lung cancer, NSCLC, pristimerin, anti-apoptosis, migration, invasion
Introduction
Lung cancer is one of the most common malignant tumor throughout the world, accounting for the highest diagnostic rate (11.6%) and cancer death rate (18.4%), approximately 80% of which belongs to non-small cell lung cancer (NSCLC) [1, 2]. Although the targeted molecular therapy and immunotherapy has made great progresses [3], until recently, for early-stage lung cancer patients, chemotherapy remained the non-operative first-line approach (other than radiofrequency ablation and standard radiotherapy) [4]. Traditional chemotherapeutic drugs are still faced with drug-resistance and related-toxicities and active development of new anti-cancer agents is warranted and promising.
Natural product contains abundant resources of bioactive agents, the anti-cancer activities of natural product which attracts increasing attention [5-8]. The natural occurring pristimerin, a quinonemethide triterpenoid, is isolated from varied Celastraceae and Hippocrateaceae family [9-11]. Recently, pristimerin has been reported to widely induce apoptosis and autophagy, inhibit angiogensis and metastasis against cancer [12]. However, the potential efficacy of pristimerin against lung cancer is relatively deficient. In this article, we investigated the in vitro anti-cancer potential of pristimerin on NSCLC cells NCI-H1299 and elucidated the molecular mechanism.
Materials and Methods
Cell culture and reagents
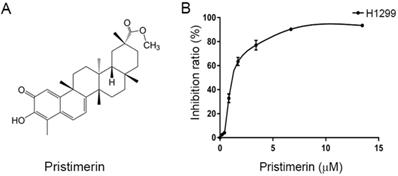
The human lung adenocarinoma epithelial cell line NCI-H1299 (lack expression of p53 protein) was obtained from iCell Bioscience Inc., Shanghai, China, and authenticated by short-tandem repeat (STR) profiling. The cells were maintained in RPMI-1640 medium supplemented with 100 μM streptomycin, 100 U/mL penicillin and 10% fetal bovine serum (FBS) (TIANHANG Biotech Co., Ltd., Zhejiang, China). Cells were cultured in a humidified atmosphere containing 5% CO2 at 37°C. Pristimerin (Fig. 1A) with a purity of >98% was purchased from the Spring & Autumn Biological Engineering Co., Ltd. (Nanjing, China).
Chemical structure of pristimerin and its ability to inhibit NCI-H1299 cells proliferation. A. Chemical structures of pristimerin. B. Pristimerin shows potent cytotoxicity to H1299 cells measured by MTT assay. The cells having grown for 24 h were exposed to a full range of concentrations of pristimerin for 72 h.
Cell viability assay
The MTT assay was utilized to evaluate the cytotoxic activity of pristimerin. 4 × 103 cells were transferred into each well of 96-well plates in a final volume of 190 μL. After cells attached for 24 h, 10 μL pristimerin were added to the medium at a series of different concentrations for 72 h incubation. 20 μL MTT (5 mg/mL) was added to each well for 4 h reaction. Next, the supernatant was discarded, and the formed formazan product was dissolved in 100 μL DMSO/well. The optical density (OD) was detected at an absorbance wavelength of 540 nm using a Model 550 Microplate reader (Bio-Rad Laboratories, Berkeley, CA, USA), with reference filter of 655 nm. The IC50 value was used to express the inhibitory action of pristimerin, which was obtained according to the concentration-response curve using Bliss software. Percent survival rate was obtained using the formula as described previously [5].
Annexin V/ propidium iodide (PI) test
Annexin V-FITC/PI double staining assay was applied to determine apoptosis, using an Annexin V-FITC/PI apoptosis detection kit (KeyGen Biotech Co., Ltd., Nanjing, China). H1299 cells were cultured in the presence of 0.9, 1.8 and 3.6 μM pristimerin for 48 h, then collected and resuspended in 0.5 mL binding buffer/sample for 5 min. Subsequently, 5 μL FITC-labeled Annexin-V and 5 μL PI were added for 15 min staining at 37°C in dark. The stained samples were measured using a flow cytometer (Beckman Coulter, Brea, CA, USA) and analyzed by CytExpert software version 2.2 (Beckman Coulter, Brea, CA, USA). The rate of apoptosis (%) was determined by dividing the number of apoptotic cells by the number of total cells observed, and multiplying with 100 [7]. All experiments were repeated three times.
Colony forming assay
Cells were split and transferred into 12-well plates with 400 cells/well. Until the colonies of 30-40 cells formed, cells were incubated with pristimerin of 1.8 μM for 24, 48 and 72 h, respectively. The wells were washed three times with PBS, then fixed with methanol and stained with 0.5% (w/v) crystal violet. Images were captured using ChemiDoc™ XRS+ (Bio-Rad Laboratories, Berkeley, CA, USA). The colony forming assay was repeated three times in duplicate.
Wound healing assay
Cells were digested using trypsin and resuspended in serum-free medium. The cells were transferred into 6-well plates and grown to reach confluence overnight. Subsequently, linear wounds were scraped with pipette tips in the cell monolayer. Next, the debris was removed and the edge of the wound was smoothed by washing the cells once. The cells were incubated with pristimerin (0.9, 1.8 and 3.6 μM), respectively. After 0, 24 and 48 h of incubation, wounds were observed and photographed using a light microscope (Olympus Life Science, Tokyo, Japan) at ×40 magnification. Cell motility was quantified by the percentage of the repaired area. The percent wound closure (%) was obtained through the following formula: percent mobility rate = migrated cell surface area/total surface area × 100 [13].
Invasion assay
The bottom of transwell apparatus (Corning Costar, Corning, NY, USA) was pre-coated with matrigel (Corning Costar, Corning, NY, USA), which was diluted by 5 times with basal medium. Cells were transferred into the upper chamber of the transwell apparatus at a density of 8 × 103 cells/well in 200 μL of serum-free RPMI-1640 medium, which contained the pristimerin (0.9, 1.8 and 3.6 μM). The lower chamber contained 600 μL RPMI-1640 medium adding 10% FBS, which was a chemoattractant. After incubation of 48 h, cells that migrated into the lower chamber were fixed with methanol and stained with 0.5% (w/v) crystal violet [14]. Cells on the top layer were removed using a cotton swab, and the migrated cells were photographed using an inverted microscope (Leica Microsystems, Wetzlar, Germany) at ×200 magnification. Nine random fields were acquired to quantify attached and migrated cells. The cell number was quantified by IMAGE-J software (version 1.51j8, Bethesda, MD, USA) (http://imagej.nih.gov/ij/). Cell counts were expressed as the mean number of cells per field of view.
Western blot
Cultured cells from different samples were collected and washed twice with ice-cold PBS. The pellet was vortexed and lysed in lysis buffer (Cell Signaling Technology, Danvers, MA, USA), supplementary with Proteinase Inhibitor Cocktail (Cell Signaling Technology, Danvers, MA, USA) and phenylmethylsulfonyl fluoride (PMSF) (Cell Signaling Technology, Danvers, MA, USA). 25 μg/lane proteins were separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and subsequently transferred onto PVDF membranes (Millipore, Boston, MA, USA). The membranes were then blocked with TBST buffer containing 5% dried non-fat milk, subsequently incubated with primary antibodies overnight at 4°C. After 3 times washed with TBST buffer, the membranes were incubated to bind horseradish peroxidase (HRP) conjugated secondary antibodies for 2 h at room temperature. Blots were visualized by Western Lightning® Plus-ELC kit (PerkinElmer, Waltham, Ma, USA) and exposed by ChemiDoc™ XRS+ (BIO-RAD, Berkeley, CA, USA). Antibodies used in this study included: rabbit anit-Integrin beta 1 (1:1000 dilution) (#9699, Cell Signaling Technology), rabbit anit-MMP2 (1:1000 dilution) (ab92536, Abcam), rabbit anit-vimentin (1:1000 dilution) (#5741, Cell Signaling Technology), mouse anti-F-actin (0.2 µg/mL) (ab4792, Abcam), rabbit anti-Snail+Slug (1:1000 dilution) (ab180714, Abcam), mouse anti-GAPDH (1:10000 dilution) (MB001, Bioworld Technology), GAPDH (1A6) monoclonal antibody (1:10000 dilution) (MB001, Bioworld Technology), Anti-rabbit IgG, HRP-linked Antibody (1:2000 dilution) (#7074, Cell Signaling Technology) and HRP-conjugated goat anti-mouse (1:5000 dilution) (BS12478, Bioworld Technology).
mRNA extraction, reverse transcription and real-time-quantitative (RT-q) PCR
Total RNA of H1299 cells stimulated with pristimerin (0.9, 1.8, 2.7 and 3.6 μM) after 24 h was extracted using TRIzol (Thermo, Waltham, MA, USA). The PrimeScript™ RT Master Mix (Takara Biomedical Technology, Kusatsu, Japan) was used to transcribe mRNAs into cDNA. The forward and reverse primers (Takara Biomedical Technology, Kusatsu, Japan) were designed to detect the expression of integrin β1, matrix metallopeptidase 2 (MMP2) and Snail. SYBR® Premix Ex Taq™ (TliRNaseH Plus) (Takara Biomedical Technology, Kusatsu, Japan) was utilized for RT-qPCR and GAPDH was used to normalize mRNA expression. RT-qPCR was carried out using a QuantStudio 5 (Thermo Fisher scientific, Waltham, MA, USA). The program setting for qPCR is as below: firstly, initial denaturation at 95°C for 30 sec; then 40 reaction cycles were followed by denaturation at 95°C for 5 sec, annealing at 60°C for 34 sec, and then elongation at 72°C for 30 sec [15]. The data were calculated using the 2‑ΔΔCq method and GAPDH was used as control gene for normalization [16]. The primer sequences designed are listed in Table 1.
The primer sequences employed in RT-qPCR
| Primer name | Primer sequence (5'--3') | Company |
|---|---|---|
| GAPDH Forward | GGAAGGTGAAGGTCGGAGTCA | Invitrogen, Inc. |
| GAPDH Reverse | GTCATTGATGGCAACAATATCCACT | Invitrogen, Inc. |
| Integrin β1 Forward | GACGCCGCGCGGAAAAGATG | TaKaRa, Inc. |
| Integrin β1 Reverse | GCACCACCCACAATTTGGCCC | TaKaRa, Inc. |
| Snail Forward | CCAGACCCACTCAGATGTCAAG | TaKaRa, Inc. |
| Snail Reverse | GGGCAGGTATGGAGAGGAAGA | TaKaRa, Inc. |
| MMP2 Forward | CTCATCGCAGATGCCTGGAA | TaKaRa, Inc. |
| MMP2 Reverse | TTCAGGTAATAGGCACCCTTGAAGA | TaKaRa, Inc. |
Statistical analysis
All experiments were performed in triplicate unless otherwise stated. The differences between groups were analyzed using ANOVA followed by Tukey's post-hoc test. Statistical differences in each assay were analyzed by SPSS software version 16.0 (SPSS, Inc., Chicago, IL, USA). Differences between groups were considered statistically significant if P < 0.05.
Results
Pristimerin exhibits a notable cytotoxicity against H1299 cells
Cell growth inhibition by pristimerin was assessed using MTT assay. Pristimerin (Fig 1A) inhibited the proliferation of H1299 cells with an IC50 value of 2.2 ± 0.34 μM. The growth inhibition occurred in a dose-dependent manner (Fig. 1B). The results indicated the cytotoxicity of pristimerin against H1299 cells.
Pristimerin shows a potent apoptotic-inducing effect against H1299 cells
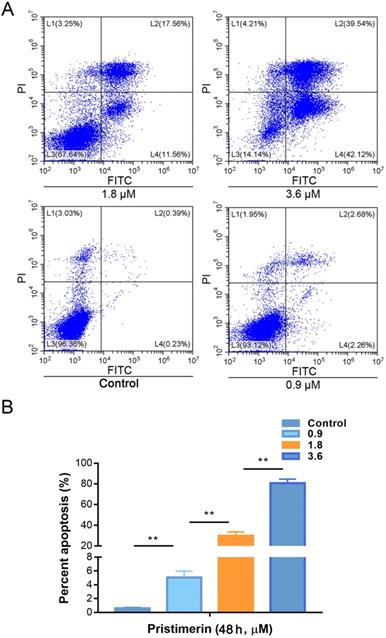
To investigate whether pristimerin is able to induce apoptosis of H1299 cells. The cells were incubated with 0.9, 1.8 and 3.6 μM of pristimerin for 48 h, then apoptosis was detected by flow cytometry based on annexin V and PI staining to discriminate healthy cells from early/late apoptotic cells and necrotic cells. The results showed that the rates of apoptosis were 0.61 ± 0.11, 5.08 ± 0.90, 29.79 ± 0.88 and 80.64 ± 3.92% for control, 0.9, 1.8 and 3.6 μM of pristimerin, respectively (Fig. 2).
Pristimerin induces lung cancer H1299 cells apoptosis in vitro. A. The lung cancer H1299 cells were treated with pristimerin for 48 h and apoptosis were detected by FACS. B. Quantitative results of (A). Data were quantified as mean ± standard deviation. **P < 0.01, n ≥ 3.
Pristimerin decreases H1299 cells colony formation
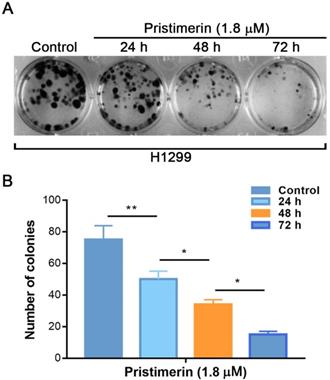
The colony forming assay was performed to further investigate the anti-proliferative effects of pristimerin. The results showed that incubation of H1299 cells with pristimerin for 24, 48 and 72 h reduced the colony formation in a time-dependent manner which was characterized by specific colony numbers and sizes of H1299 cells (Fig. 3).
Pristimerin of 1.8 µM leads to a significant inhibition of colony formation of H1299 cells in a time-dependent manner. A. H1299 cells were treated with different concentrations of pristimerin and colony efficiency was observed via a colony forming cell assay. B. Quantitative results of (A). Data were quantified as mean ± standard deviation. *P < 0.05, **P < 0.01, n ≥ 3.
Pristimerin reduces H1299 cells migration and invasion
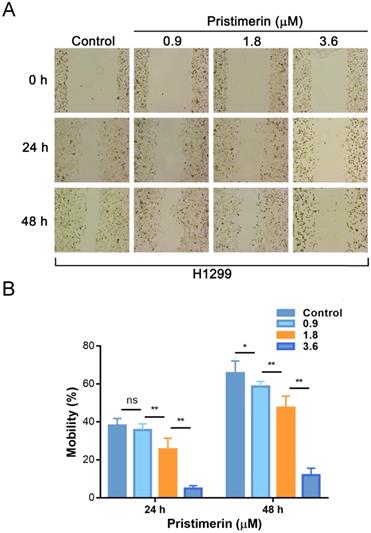
Wound healing assay was applied to evaluate the inhibitory effect of pristimerin on NSCLC cells migration. After H1299 cells were exposed to pristimerin for 24 h at a concentration of 0.9, 1.8 and 3.6 μM, the mobility ratios with respect to control were 38.03 ± 3.71, 35.67 ± 3.23, 25.54 ± 5.76 and 4.91 ± 1.47%, respectively. Moreover, after exposure to pristimerin for 48 h at the same series of concentration, the mobility ratios were 65.66 ± 6.42, 58.61 ± 3.72, 47.36 ± 6.18, 11.92 ± 3.63%, respectively (Fig. 4). The result suggested that pristimerin markedly inhibited H1299 cells migration in a dose-dependent manner.
Treatment of pristimerin decreases the migrative ability of H1299 cells in vitro in a dose-dependent manner. A. H1299 cells were treated with different concentrations of pristimerin for 24 h and 48 h, the mobility ratio was observed via wound healing assays. B. Quantitative results of (A). Data are presented as the mean ± standard deviation. *P < 0.05, **P < 0.01, n ≥ 3.
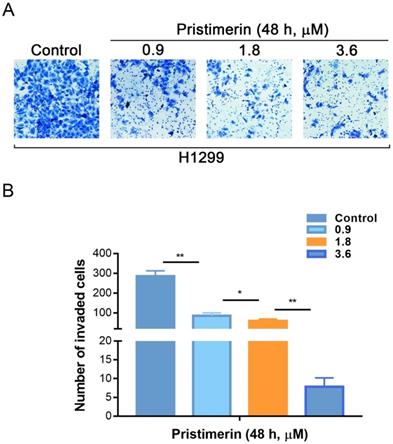
Invasion is another process of cancer proliferation and metastasis. Transwell assay was utilized to analyze the role of pristimerin in regulating the invasion of NSCLC cells. Consistent with the results of migration assay, pristimerin markedly inhibited NSCLC cells invasion. After treatment of H1299 cells with 0.9, 1.8 and 3.6 μM pristimerin for 48 h, the number of invaded cells decreased (Fig. 5). The above results indicated that pristimerin reduced cell invasion in a dose-dependent manner.
H1299 cells invasion is inhibited in vitro by pristimerin in a dose-dependent manner. A. The invasive ability of H1299 cells was detected via transwell assay. Following treatment of H1299 cells with different concentrations of pristimerin for 48 h, the number of invaded cells decreased in a dose-dependent manner. B. Quantitative results of (A). Data are presented as the mean ± standard deviation. *P < 0.05, **P < 0.01, n ≥ 3.
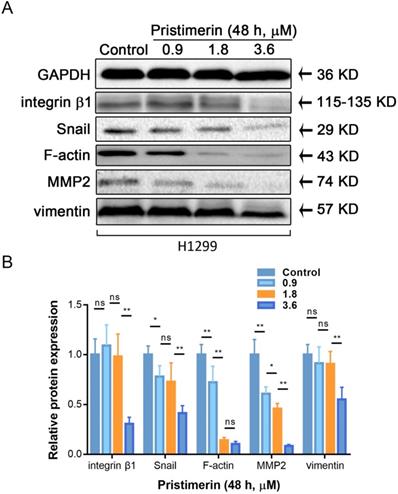
Pristimerin regulates protein expression
Cancer metastasis is a complicated process, which involved motility, adhesion and disruption of polarity of cell. To investigate whether pristimerin inhibited EMT of H1299 cells, western blot was used to detect the protein express of vimentin, F-actin integrin β1, MMP2 and Snail. After H1299 cells were incubated with 0.9, 1.8 and 3.6 μM pristimerin for 48 h, the protein expression of vimentin, F-actin, integrin β1, MMP2 and Snail were decreased in a dose-dependent manner (Fig. 6).
Pristimerin regulates migration and invasion-related proteins expression in H1299 cells. After H1299 cells were treated with pristimerin of 0.9, 1.8 and 3.6 μM for 48 h respectively, the whole-cell lysates were assayed by Western blot and corresponding antibodies. A. Down-regulations of vimentin, F-actin, integrin β1, MMP2 and Snail expression by pristimerin in a dose-dependent manner were observed. B. Data analysis of (A). Data are presented as the mean ± standard deviation. *P < 0.05, **P < 0.01, n ≥ 3.
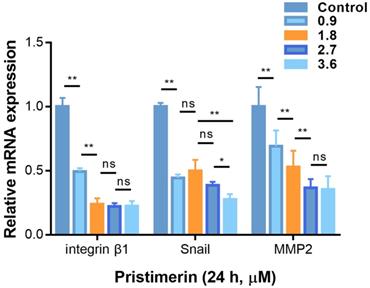
Pristimerin affects mRNA expression
To determine whether the anti-proliferative, anti-migration and anti-invasive activities of pristimerin were related to the specific mRNA levels, RT-qPCR was used to determine the mRNA expression. After H1299 cells were incubated with pristimerin (0.9, 1.8, 2.7 and 3.6 μM) for 24 h, the mRNA levels of integrin β1, MMP2 and Snail were down-regulated in a dose-dependent manner (Fig. 7).
Pristimerin regulates migration and invasion-related mRNA in H1299 cells. A. After H1299 cells were treated with pristimerin of 0.9, 1.8, 2.7 and 3.6 µM for 24 h respectively, the mRNA expression was detected by RT-qPCR. Down-regulation of Snail, and integrin β1and MMP2 mRNA expression were observed. B. Data analysis of (A). Data are presented as the mean ± standard deviation. *P < 0.05, **P < 0.01, n ≥ 3.
Discussion
Pristimerin (Fig. 1A), a quinonemethide triterpenoid, kills cancer cell lines in a wide spectrum [12], as well as potent in vivo anti-cancer activities [17, 18]. However, its activities against lung cancer are insufficience of evidence to support. It was reported that pristimerin has shown activities of proliferation-inhibiting and apoptosis-inducing in NSCLC cells [11, 19]. At the present research, pristimerin exhibited a powerful cytotoxicity to H1299 cells, with IC50 value of 2.2 ± 0.34 μM (Fig. 1B). Apoptosis and autophagy induced by pristimerin were widely revealed [20-22]. In our study, the annexin V-FITC/PI test was used to detect apoptosis. The results showed that apoptosis was strongly induced in a dose-dependent manner in H1299 cells (Fig. 2). Subsequently, colony forming cell assay was utilized to detect the anti-proliferative ability of pristimerin. The decreased cell numbers and sizes of H1299 cells showed that pristimerin time-dependently inhibited cell proliferation (Fig. 3).
Cancer metastasis is the most common reason that caused human cancer deaths, while only a minority of lung cancers are localised when diagnosis [23-26]. In order to investigate whether pristimerin inhibited motility of NSCLC cells, wound healing assay and transwell assay were applied. The experimental data indicated that pristimerin significantly decreased NSCLC cells migration in a dose-dependent manner (Fig. 4), as well as mobility (Fig. 5).
Western blot assay was subsequently utilized to find out the regulated protein targets related to this process. Vimentin controls the alignment of the cell traction forces to direct single-cell mesenchymal migration, and marks epithelial-mesenchymal transition (EMT) [27]. As the data showed, vimentin was dose-dependently down-regulated. Additionally, F-actin, another cytoskeleton polymers needed for migration [28], was also decreased by pristimerin. To complete cell adhesion, cellular cytoskeleton needs the assistant of integrin family to connect extracellular matrix (ECM) network [29], the decreased integrin β1 in our results indicated the inhibition of adhesive process [30]. Besides the cancer cell-cell adhension, it also needs the ability to destroy the histological barrier of cancer cell invasion. Matrix metallopeptidase (MMP) is considered as an important member of the invasive process, which cancer cell secreted to dissolve the components of ECM or basement membrane [31]. It was previously reported that pristimerin down-regulated MMP2 and matrix metallopeptidase 9 (MMP9) to inhibit migration and invasion of esophageal squamous cell carcinoma (ESCC) [32]. Similarly, we found an obviously decreased expression of MMP2 in our study. Zuo et al. demonstrated the inhibitory action on EMT activation in prostate cancer (PCa) PC-3 cells [33], which was confirmed by EMT markers including N-cadherin, fibronectin, vimentin and ZEB1. While in our study, the activities of pristimerin to reduce the cellular cytoskeleton of vimentin and F-actin, cellular adhesion of integrin β1, as well as cellular invasion of MMP2, exerted the multiple ways of anti-cancer potential. Furthermore, the key EMT upstream protein Snail was inhibited revealed the relationship of this process (Fig. 6) [34].
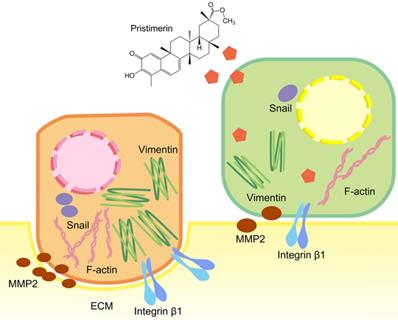
To further explore whether the inhibition of these proteins was corelated with mRNA levels, RT-qPCR was applied. Consistent with the protein levels, integrin β1, MMP2 and Snail were down-regulated in a dose-dependent manner after 24 h treatment. This indicated pristimerin may regulate integrin β1, MMP2 and Snail proteins via suppressing their mRNA translation (Fig. 7). The above molecular mechanisms of pristimerin are summarized in Fig. 8.
Brief summary of molecular mechanisms involved in pristimeirn-induced anti-proliferative, anti-migration and anti-invasive effects against human NSCLC cell line NCI-H1299.
Conclusion
In conclusion, our study demonstrated that pristimerin suppressed NSCLC cells NCI-H1299 in vitro, exhibited potent activities of proliferation inhibition and apoptosis induction. Furthermore, the treatment of pristimerin decreased migration and invasion of H1299 cells, which was correlated with EMT-related proteins and mRNA.
Acknowledgements
This work was supported by National Natural Science Foundation of China (U1903126, 81773888 and 81902152), Fund of Guangzhou Science and Technology Program (201707010048), Fund from Guangzhou Institute of Pediatrics/Guangzhou Women and Children's Medical Center (No: YIP-2018-031 and No: IP-2018-012).
Authors' Contributions
JZ and YY conceived and designed the experiments; JL, QG and XL performed the experiments; LZ and CS analyzed the data; YL, WZ and HC contributed analysis tools; HW and FW revised the paper; JL and JZ wrote the paper. All authors have read and approved the manuscript.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Travis WD, Brambilla E, Nicholson AG, Yatabe Y, Austin JHM, Beasley MB. et al. The 2015 World Health Organization classification of lung tumors: impact of genetic, clinical and radiologic advances since the 2004 classification. J Thorac Oncol. 2015;10:1243-60
2. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424
3. Osmani L, Askin F, Gabrielson E, Li QK. Current WHO guidelines and the critical role of immunohistochemical markers in the subclassification of non-small cell lung carcinoma (NSCLC): Moving from targeted therapy to immunotherapy. Semin Cancer Biol. 2018;52:103-9
4. Hirsch FR, Scagliotti GV, Mulshine JL, Kwon R, Curran WJ Jr, Wu YL. et al. Lung cancer: current therapies and new targeted treatments. Lancet. 2017;389:299-311
5. Lin M, Tang S, Zhang C, Chen H, Huang W, Liu Y. et al. Euphorbia factor L2 induces apoptosis in A549 cells through the mitochondrial pathway. Acta Pharm Sin B. 2017;7:59-64
6. Zhang JY, Lin MT, Tung HY, Tang SL, Yi T, Zhang YZ. et al. Bruceine D induces apoptosis in human chronic myeloid leukemia K562 cells via mitochondrial pathway. Am J Cancer Res. 2016;6:819-26
7. Tao YW, Lin YC, She ZG, Lin MT, Chen PX, Zhang JY. Anticancer activity and mechanism investigation of beauvericin isolated from secondary metabolites of the mangrove endophytic fungi. Anticancer Agents Med Chem. 2015;15:258-66
8. Liu Y, Sun H, Makabel B, Cui Q, Li J, Su C. et al. The targeting of noncoding RNAs by curcumin: Facts and hopes for cancer therapy (Review). Oncol Rep. 2019;42:20-34
9. Tang WH, Bai ST, Tong L, Duan WJ, Su JW, Chen JX. et al. Chemical constituents from Celastrus aculeatus Merr. Biochem Syst Ecol. 2014;54:78-82
10. Murayama T, Eizuru Y, Yamada R, Sadanari H, Matsubara K, Rukung G. et al. Anticytomegalovirus activity of pristimerin, a triterpenoid quinone methide isolated from Maytenus heterophylla (Eckl. & Zeyh.). Antivir Chem Chemother. 2007;18:133-9
11. Chang FR, Hayashi K, Chen IH, Liaw CC, Bastow KF, Nakanishi Y. et al. Antitumor agents. 228. five new agarofurans, Reissantins A-E, and cytotoxic principles from Reissantia buchananii. J Nat Prod. 2003;66:1416-20
12. Li JJ, Yan YY, Sun HM, Liu Y, Su CY, Chen HB. et al. Anti-cancer effects of pristimerin and the mechanisms: a critical review. Front Pharmacol. 2019;10:746
13. Das T, Safferling K, Rausch S, Grabe N, Boehm H, Spatz JP. A molecular mechanotransduction pathway regulates collective migration of epithelial cells. Nat Cell Biol. 2015;17:276-87
14. Qin J, Tang J, Jiao L, Ji J, Chen WD, Feng GK. et al. A diterpenoid compound, excisanin A, inhibits the invasive behavior of breast cancer cells by modulating the integrin β1/FAK/PI3K/AKT/β-catenin signaling. Life Sci. 2013;93:655-63
15. Wei M, Li J, Qiu J, Yan Y, Wang H, Wu Z. et al. Costunolide induces apoptosis and inhibits migration and invasion in H1299 lung cancer cells. Oncol Rep. 2020;43:1986-94
16. Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc. 2008;3:1101-8
17. Park JH, Kim JK. Pristimerin, a naturally occurring triterpenoid, attenuates tumorigenesis in experimental colitis-associated colon cancer. Phytomedicine. 2018;42:164-71
18. Yousef BA, Hassan HM, Guerram M, Hamdi AM, Wang B, Zhang LY. et al. Pristimerin inhibits proliferation, migration and invasion, and induces apoptosis in HCT-116 colorectal cancer cells. Biomed Pharmacother. 2016;79:112-9
19. Zhang Y, Wang J, Hui B, Sun W, Li B, Shi F. et al. Pristimerin enhances the effect of cisplatin by inhibiting the miR23a/Akt/GSK3beta signaling pathway and suppressing autophagy in lung cancer cells. Int J Mol Med. 2019;43:1382-94
20. Deeb D, Gao X, Liu YB, Pindolia K, Gautam SC. Pristimerin, a quinonemethide triterpenoid, induces apoptosis in pancreatic cancer cells through the inhibition of pro-survival Akt/NF-κB/mTOR signaling proteins and anti-apoptotic Bcl-2. Int J Oncol. 2014;44:1707-15
21. Zhao H, Wang C, Lu B, Zhou Z, Jin Y, Wang Z. et al. Pristimerin triggers AIF-dependent programmed necrosis in glioma cells via activation of JNK. Cancer Lett. 2016;374:136-48
22. Cevatemre B, Erkisa M, Aztopal N, Karakas D, Alper P, Tsimplouli C. et al. A promising natural product, pristimerin, results in cytotoxicity against breast cancer stem cells in vitro and xenografts in vivo through apoptosis and an incomplete autopaghy in breast cancer. Pharmacol Res. 2018;129:500-14
23. Mehlen P, Puisieux A. Metastasis: a question of life or death. Nat Rev Cancer. 2006;6:449-58
24. Langer CJ, Besse B, Gualberto A, Brambilla E, Soria JC. The evolving role of histology in the management of advanced non-small-cell lung cancer. J Clin Oncol. 2010;28:5311-20
25. McWilliams A, Lam B, Sutedja T. Early proximal lung cancer diagnosis and treatment. Eur Respir J. 2009;33:656
26. Fares J, Fares MY, Khachfe HH, Salhab HA, Fares Y. Molecular principles of metastasis: a hallmark of cancer revisited. Signal Transduct Target Ther. 2020;5:28
27. Costigliola N, Ding L, Burckhardt CJ, Han SJ, Gutierrez E, Mota A. et al. Vimentin fibers orient traction stress. Proc Natl Acad Sci U S A. 2017;114:5195-200
28. Dominguez R, Holmes KC. Actin structure and function. Annu Rev Biophys. 2011;40:169-86
29. Chong Y, Tang D, Xiong Q, Jiang X, Xu C, Huang Y. et al. Galectin-1 from cancer-associated fibroblasts induces epithelial-mesenchymal transition through β1 integrin-mediated upregulation of Gli1 in gastric cancer. J Exp Clin Cancer Res. 2016;35:175
30. Huang W, Yan Y, Liu Y, Lin M, Ma J, Zhang W. et al. Exosomes with low miR-34c-3p expression promote invasion and migration of non-small cell lung cancer by upregulating integrin α2β1. Signal Transduct Target Ther. 2020;5:39
31. Lyu Y, Xiao Q, Yin L, Yang L, He W. Potent delivery of an MMP inhibitor to the tumor microenvironment with thermosensitive liposomes for the suppression of metastasis and angiogenesis. Signal Transduct Target Ther. 2019;4:26
32. Tu Y, Tan F, Zhou J, Pan J. Pristimerin targeting NF-κB pathway inhibits proliferation, migration, and invasion in esophageal squamous cell carcinoma cells. Cell Biochem Funct. 2018;36:228-40
33. Zuo J, Guo Y, Peng X, Tang Y, Zhang X, He P. et al. Inhibitory action of pristimerin on hypoxia-mediated metastasis involves stem cell characteristics and EMT in PC-3 prostate cancer cells. Oncol Rep. 2015;33:1388-94
34. Zheng X, Carstens JL, Kim J, Scheible M, Kaye J, Sugimoto H. et al. Epithelial-to-mesenchymal transition is dispensable for metastasis but induces chemoresistance in pancreatic cancer. Nature. 2015;527:525-30
Author contact
Corresponding authors: Jianye Zhang, Guangdong Provincial Key Laboratory of Molecular Target & Clinical Pharmacology, School of Pharmaceutical Sciences and the Fifth Affiliated Hospital, Guangzhou Medical University, Guangzhou, 511436, P. R. China. E-mail: jianyezcom & jianyezedu.cn and Yanyan Yan, Institute of Immunology and School of Medicine, Shanxi Datong University, Datong 037009, P. R. China. E-mail: zwsanyancom.

 Global reach, higher impact
Global reach, higher impact