Impact Factor ISSN: 1837-9664
J Cancer 2018; 9(24):4618-4626. doi:10.7150/jca.27673 This issue Cite
Research Paper
α-1,2-Mannosidase MAN1C1 Inhibits Proliferation and Invasion of Clear Cell Renal Cell Carcinoma
1. Department of Urology, The First Affiliated Hospital of Harbin Medical University, Harbin, Heilongjiang Province, China
2. Department of Neurosurgery, The First Affiliated Hospital of Harbin Medical University, Harbin, Heilongjiang Province, China
Received 2018-6-5; Accepted 2018-10-1; Published 2018-11-24
Abstract
Background: This study investigated the biological function of the gene MAN1C1 α-mannosidase in renal cell carcinoma. It has been reported that MAN1C1 is probably a potential tumor suppressor gene in Wilms. However, the role of MAN1C1 in human clear cell renal cell carcinoma (ccRCC) has not been reported.
Methods: In this study, MAN1C1 gene over-expression was used to transfect human renal cancer cell lines 786-O and OS-RC-2 to study apoptosis and the underlying mechanisms which influence epithelial-mesenchymal transition.
Results: MAN1C1 was down-regulated in ccRCC and related to the clinicopathological factors and prognosis of ccRCC. We revealed that over-expression MAN1C1 showed anti-tumor effect by inducing apoptosis, as determined by Cell Counting Kit-8 (CCK-8) assay, cell cycle analysis, and western blot analysis. What's more, MAN1C1 over-expression remarkably increased the ratio of Bax/Bcl-2 and inhibited epithelial-mesenchymal transition by increasing the expression of E-CA. In addition, the ratio of Bax/Bcl-2 and E-CA were also increased in MAN1C1 gene over-expression renal cancer cells compared with the control cells.
Conclusion: We find that re-expression of silenced MAN1C1 in ccRCC cell lines inhibited cell viability, colony formation, induced apoptosis, suppressed cell invasion and migration. In conclusion, MAN1C1 is a novel functional tumor suppressor in renal carcinogenesis. This is the first time that the function of MAN1C1 gene has been verified in the renal tumor tissue so far.
Keywords: tumor growth, MAN1C1, Bax/Bcl-2, gene over-expression
Introduction
Renal cell carcinoma (RCC) is a common urologic malignancy and accounts for ~3% of adult malignancies. Every year, over 300,000 new patients are diagnosed around the world[1] with an estimated 102,000 deaths[2]. As the most common type among renal carcinomas, clear cell RCC (ccRCC) constitutes 80% of all cases[3]. In addition to surgery, RCC relative radiotherapy, chemotherapy and targeted therapy are not ideal[4, 5]. Approximately 30% of RCC patients present with metastatic disease at the time of diagnosis and nearly half of the remainder will subsequently develop, and ultimately die from metastasis[6]. Major clinical challenges for RCC are the lack of a screening method for early-stage diagnosis and the lack of biomarkers for prediction of progression. Thus, it has become a focus of basic and clinical RCC research to search for novel tumor biomarkers for patients with RCC and novel anti-RCC treatment. N-glycosylation is significant for the formation of mature glycoproteins in eukaryotes[7].
Previous gene array studies indicate that the myriad of glycosyl and mannosyltransferases are potential targets[8], which act in concert to control the pattern of protein N-glycosylation in the endoplasmic reticulum and Golgi complex. α-1,2-mannosidases are an essential enzyme for the maturation of N-glycans and is involved in the degradation of N-glycans as well[9]. Therefore, class I α-1,2-mannosidases are significant for Asn-linked oligosaccharide maturation in the endoplasmic reticulum (ER) and Golgi complex[10]. The process of N-glycosylation consists of a covalent linkage of a specific oligosaccharide (Glc3Man9GlcNAc2) to a nascent protein. Once the oligosaccharide is transferred, several subsequent steps of maturation occur along the secretory pathway[11].
The four class I α-1,2 mannosidases in humans are ER a-mannosidaseI (MAN1B1) and three Golgi a1,2-mannosidases (MAN1A1, MAN1A2, MAN1C1) [12]. They work in concert to trim the mannose residues in the oligosaccharides to form oligosaccharide complexes, which become specific enzymes in the inner and cross Golgi compartments. These modifications affect cell growth, cell-cell adhesion, cell motility and protein phosphorylation; therefore, the activity of the enzymes involved in Nglycosylation must be tightly regulated because N-glycan composition determines the fate of the protein, including whether or not the protein will be folded in the ER lumen or retrotranslocated into the cytosol and degraded[1, 13]. Thus, α-1,2-mannosidases are both involved in protein folding and play a role in misfolded protein degradation1[14]. Its metabolic abnormalities can cause various diseases including cancers[15]. Most of the current studies are caused by congenital erythropoiesis[16] and α-mannosidase accumulation diseases[17]. It has also been reported that MAN1B1 promotes hepatocellular carcinoma (HCC) formation[18]. In addition, two subtypes of α-1,2 mannosidases are reported to have different functions, with MAN1A1 possessing oncogenic qualities and MAN1C1 potentially acting as a tumor suppressor[19].
A. C. V. Krepischi carried out Array-CGH analysis in Wilms tumor (WT) and found that MAN1C1 gene was down-regulated[20]. Moreover, Brahim Aissani, Ph. D, advanced that MAN1C1 gene may have important associations with the size of uterine fibroids[21]. Hence, we wonder whether MAN1C1 gene can play a role in suppressing tumors.
Since the potential function of MAN1C1 in cancer has not yet been studied, we carried out the identification of MAN1C1 down-regulation from ccRCC samples and The Cancer Genome Atlas (TCGA) Data in the present study. Our data reveal that MAN1C1 may play vital roles in renal cell cancer and provide a potential target for the treatment of renal cell carcinoma. As far as we know, this is the first time that MAN1C1 gene has been found to play a role in renal tumors.
Materials and methods
Data sets collection and ccRCC patient samples
A total 42 malignant kidney clear cell carcinoma patients, without any chemotherapy or radiotherapy, were enrolled in our study. Tumor tissue samples and corresponding matched adjacent noncancerous tissue samples were taken from the surgical resection tissue of kidney clear cell carcinoma patients of the First Affiliated Hospital of Harbin Medical University. All patients have signed the informed consents. What's more, the study had obtained approval and supervision of Ethics Committee of the First Affiliated Hospital of Harbin Medical University.
TCGA ccRCC RNA-Seq and matching clinical data were collected from Synapse website (http://synapse.org;syn1461159). RNA-Seq analysis was carried out with the TCGA data of 532 ccRCC and 72 adjacent renal tissues.
RNA extraction and quantitative real-time PCR
According to the manufacturer's instructions, total RNA was extracted from fresh samples preserved and frozen in liquid nitrogen using TRIzol Reagent (Invitrogen, Carlsbad, CA , USA). Then total RNA (1 µg) was reverse transcribed into cDNA using High-Capacity cDNA Reverse Transcriptase Kits (TOYOBO, JAPAN). The relative level of MAN1C1 to the housekeeping gene GAPDH was determined by qRT-PCR using a lightcycler-480 (Roche). PCR amplification was carried out in triplicate, first at 95°C for 10 m, 40 cycles of 95°C for 15 s, and 60°C for 60 s. The relative expression levels of MAN1C1 were normalized to the housekeeping gene GAPDH (Invitrogen, Carlsbad, Calif, USA) and quantified with a NanoDrop ND-1000. The changes in the threshold cycle (CT) values were calculated by the equation ΔCT = CT (target) - CT (endogenous control) and fold difference was calculated as 2-Δ (ΔCT) and -ΔCT. Primers were designed by Oligo 6 software and primer pairs were as following: MAN1C1-F 5ˊ-GTGTCCGTCTGTTCCATCATTC-3ˊ, MAN1C1-R 5ˊ-TCCCTTACCATCGCTTTCTCC-3ˊ.
Cell culture and gene over-expression
OS-RC-2 and 786-O human RCC cell lines were obtained from the Chinese Academy of Science (Shanghai, China) and cultured in Roswell Park Memorial Institute-1640 (RPMI 1640) (Gibco, Carlsbad, CA, USA) supplemented with 10% (v/v) heat-inactivated fetal bovineserum (Gibco), 1% streptomycin/ penicillin, at 37 °C and 5% CO2. The OS-RC-2 and 786-O cell lines were cultured all night in a six-well plate (2.0 x 105 cells per well) and then transfected with the pcDNA-MAN1C1 plasmid or control vector(Vigenebio, China) when cells were grown to 75%. Cells were transfected with Lipofecamine 2000 (Invitrogen, Carlsbad, CA, USA).
Cell proliferation assay
According to the manufacturer's protocol, cell proliferation was measured by using the cell counting Kit-8 (Beyotime, Shanghai, China). In brief, 4 × 103 cells were seeded in a 96-well plate. Cell proliferation was assessed for 24, 48, and 72 h. After adding 10 μl WST-1 reagents per well, cultures were incubated for 2h and the absorbance was measured at 450 nm with a microplate reader (BioTek, VT, United States).
Colony formation assay
500 cells per well were seeded in 6-well plates for the colony formation assay. Visible colonies were observed with the naked eye 10 days later, fixed with 4% formaldehyde and stained with 0.1% crystal violet. Colonies whose diameters are greater than 1 mm were counted.
Flow cytometry
According to the instructions of manufacturer, the Annexin V-PE Apoptosis detection kit (BD Biosciences, San Jose, CA) was used to examine cell apoptosis. In brief, cells were washed and resuspended in 1 ×Binding Buffer. Then, 100 μl of the cell solution (1 × 105 cells) was mixed with 5 μl of Annexin V-PE and 5μl of 7-AAD, and incubated for 15 min at room temperature. Afterwards, 400 μl of 1 × binding buffer was added to each tube, and apoptosis analysis was performed in a FACScan instrument (Becton Dickinson, Mountain View, CA, USA).
Western blotting
Proteins were extracted from ccRCC cell lines by using urea buffer (2 M Thiourea, 40% CHAPS, 40mM Tris-Base, 40mM DTT, 2% Pharmalyte). Equal amounts of proteins were electrophoresed at sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and then transferred to PVDF membranes with cold transfer buffer. Membranes were blocked with 5% non-fat dry milk in TBS-T for 1 h at room temperature (RT), and incubated with corresponding primary antibodies at 4°C overnight. Antibodies used are listed as following: β-actin (1: 1000 dilution), Bcl-2 (1: 500 dilution), Bax (1: 500 dilution), E-cadherin (1: 500 dilution ), N-cadherin (1: 500 dilution), Vimentin (1: 500 dilution), cleaved-caspase3 (1: 500 dilution), cleaved-caspase 12 (1: 500 dilution), from Wanleibio, China and MAN1C1 (1:500 dilution; Abcam, USA). Secondary antibodies, goat anti-mouse-HRP and goat anti-rabbit-HRP, were diluted at 1:2000.
Cell migration and invasion assay
Wound-healing assay was carried out to evaluate the migration rate of 786-O and OS-RC-2 cells transfected with MAN1C1 or control. At first, 1.5x106 cells/well were seeded in 6-well plates and incubated overnight until the cells reached 90% confluence. A straight scratch was made by a sterile pipette tip. The destroyed cells were rinsed off by using PBS 3 times gently and incubated in medium for another 24 h. Cell migration was observed and imaged at 0 and 24 h with a digital camera (Leica DFC300FX). The invasion assays were performed in the following way. 786-O and OS-RC-2 cells pre-transfected with MAN1C1 or control (2x104cells/well) were placed in transwell cell culture chambers (8 mm pore size; Merck Millipore Corp,Billerica, MA, USA), coated with Matrigel (Becton-Dickinson, East Rutherford, NJ, USA). Cell suspension was placed in the upper chamber of the insert and the lower chamber was filled with complete medium (containing 20% FBS) as a chemoattractant. Cells were incubated for another 24 h. Non-invading cells on the upper membranes were removed and the invasive cells were fixed in 95% ethanol and stained with hematoxylin. What's more, Cells were examined, photographed and quantified under a light microscope at 100x in five random fields per membrane. Each sample was assayed in triplicate.
Statistical analysis
The Kaplan-Meier method and log-rank test were performed to show survival differences respectively according to MAN1C1 expression. The time for overall survival was calculated from the time of surgery until the occurrence of death. Survival data were assessed by using univariate. The differences between the groups in our vitro experiments were analyzed by using Student's t-test. Spearman correlation coefficients were calculated for correlation analysis. P<0.05 was considered statistically significant. All experiments were carried out in triplicate, and the SPSS 18.0 software system (SPSS, Chicago, IL) was used for statistical analysis. All statistical tests were two-sided and P< 0.05 was considered statistically significant.
Results
MAN1C1 mRNA and protein expression are frequently downregulated or silenced in primary ccRCC tumor tissues
To identify potentially important genes that contribute to renal cancer suppression, we first searched the TCGA database of published microarray data from matched malignant and non-malignant samples. We collected TCGA RNAseqV2 data from 532 ccRCC samples and 72 non-tumor samples. It was found that MAN1C1 gene was present with lower expression in the tumor than in normal tissue (Fig. 1A). Then, the expression levels of MAN1C1 in 42 pairs of human primary ccRCC and adjacent normal tissues of patients from the First Affiliated Hospital of Harbin Medical University were analyzed by qRT-PCR. Consistent with the data of TCGA, MAN1C1 expression in ccRCC tissues was significantly reduced compared to the pair-matched adjacent normal tissues (P< 0.001,Fig. 1B).
Relative MAN1C1 expression in ccRCC tissues and cell lines. (A) Relative expression of MAN1C1 between ccRCCs (n=532) and normal renal tissue samples (n=71) (TCGA). (B) The down-regulated MAN1C1 expression in ccRCC tissues was confirmed by qRT-PCR. (C) Expression of MAN1C1 in ccRCC cell lines was demonstrated by qRT-PCR. (D) Expression of MAN1C1 in ccRCC cell lines was demonstrated by Western blot analysis. (E) MAN1C1 expression was examined by western blot analysis in lysates prepared from human tumors (Ca) or uninvolved adjacent tissue (N) from the same patient. Actin as loading control
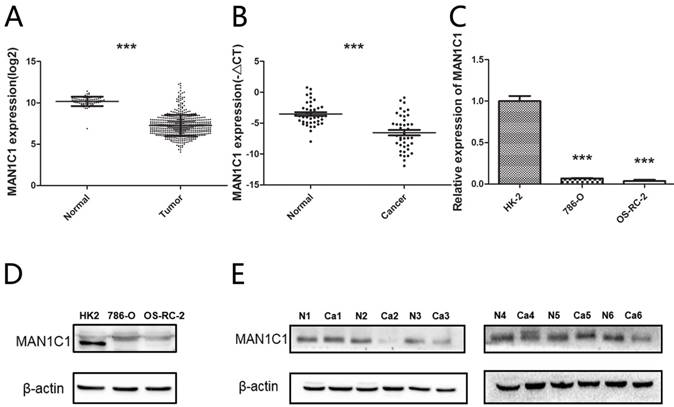
To identify the relevant cell line models for utilization in further experiment, a reduced expression of MAN1C1 mRNA was also observed in two different ccRCC cell lines (786-O and OS-RC-2) compared to a normal kidney cell line (HK-2) (Fig. 1C). In addition, we found that MAN1C1 protein (~71 kDa) was lowly expressed in cell lines (Fig. 1D) and tumor tissues (Fig. 1E). In 6 pairs of renal cell carcinoma and adjacent normal tissues, the expression of 4 pairs of MAN1C1 protein was lower than adjacent normal tissues. Briefly, all the results indicate that MAN1C1 is down-regulated in ccRCC.
Down-regulation of MAN1C1 is associated with ccRCC TNM stages and poor prognosis
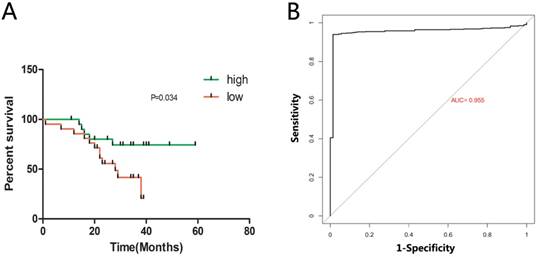
Next, we detected the potential clinical significance of MAN1C1 in ccRCC. According to the relative expression levels of MAN1C1 (with the median value as the cutoff) in cancer/adjacent tissues, patients were divided into two groups: the MAN1C1 high expression group (n = 21) and the MAN1C1 low expression group (n = 21). The correlation between MAN1C1 expression and clinicopathological characteristics is shown in Table 1. Low MAN1C1 expression was positively associated with high TNM stage (P = 0.038) and advanced pathological grade (P = 0.04). However, the expression level of MAN1C1 was not significantly associated with present lymph node metastasis as well as gender or age. Kaplan-Meier survival analysis indicated that patients with lower MAN1C1 expression had a shorter overall survival time compared to the ones with high MAN1C1 expression (Fig. 2A , P = 0.048). Univariate and multivariate Cox proportional hazards analyses showed that MAN1C1, TNM stage and pathological grade were identified to be independent prognostic factors for survival in ccRCC patients (Table 2). These results suggest that MAN1C1 may be a good diagnostic biomarker for ccRCC. We then used a receiver operating characteristic (ROC) curve to study the diagnostic value of MAN1C1 in distinguishing between ccRCC tissue and adjacent non-tumour tissue. The area under the curve (AUC) was calculated for the ROC curve and the AUC of the expression level of MAN1C1 was 0.955 (Fig. 2B), demonstrating that MAN1C1 has good potential as a biomarker.
Associations of MAN1C1 expression with clinicpathological factors
| Number of patients | MAN1C1 | χ2 | P value | ||
|---|---|---|---|---|---|
| High expression (%) | Low expression (%) | ||||
| Gender | 0.389 | 0.533 | |||
| Male | 24 | 11(52.4) | 13(61.9) | ||
| Female | 18 | 10(47.6) | 8(38.1) | ||
| Age | 0.618 | 0.432 | |||
| ≤ 65 | 34 | 16(76.2) | 18(85.7) | ||
| >65 | 8 | 5(23.8) | 3(14.3) | ||
| TNM Stage | 4.286 | 0.038 | |||
| I+II | 35 | 20(95.2) | 15(71.4) | ||
| III+IV | 7 | 1(4.8) | 6(28.6) | ||
| Grade | 4.200 | 0.04 | |||
| Grade 1+2 | 30 | 18(8.7) | 12(57.1) | ||
| Grade 3+4 | 12 | 3(14.3) | 9(42.9) | ||
| Lymph node metastasis | 1.543 | 0.214 | |||
| No | 35 | 19(90.5) | 16(76.2) | ||
| Yes | 7 | 2(9.5) | 5(23.8) | ||
| Distant metastasis | 0.359 | 0.549 | |||
| M0 | 39 | 20(95.2) | 19(90.5) | ||
| M1 | 3 | 1(4.8) | 2(9.5) | ||
P value when expression levels were compared using the Pearson Chi-square test
Relative MAN1C1 expression and its relationship with overall survival of ccRCC patients. (A) Kaplan-Meier survival analysis demonstrated that patients with low MAN1C1 expression had a shorter overall survival time compared to those with high MAN1C1 expression ( P = 0.047). (B) based on high and low MAN1C1 expression in patients with ccRCC (TCGA). “ MAN1C1 low” (N = 266), “MAN1C1 high” ( N = 266). The ROC analysis revealed that a larger area under the curve for MAN1C1 in ccRCC indicates greater potential as a biomarker (AUC = 0.955) .
Univariate and multivariate regression analyses of parameters associated with the prognosis of ccRCC patients
| Characteristics | Subset | Univariate analysis | Multivariate analysis | ||
|---|---|---|---|---|---|
| Hazard ratio(95% CI) | P value | Hazard ratio(95% CI) | P value | ||
| Gender | Male/Female | 0.900(0.342-2.3702) | 0.832 | 0.633(0.199-2.015) | 0.439 |
| Age | ≤60/> 60 | 1.798(0.630-5.132) | 0.273 | 2.324(0.589-9.171) | 0.228 |
| Tumor Stage | I+II/III+IV | 1.790(0.581-5.509) | 0.310 | 0.009 (0.000-0.298) | 0.008 |
| Tumor Grade | Grade(1+2)/Grade(3+4) | 5.904(2.223-15.683) | <0.001 | 10.025(2.295-43.782) | 0.002 |
| Lymph node metastasis | Yes/No | 3.001(1.046-8.611) | 0.041 | 3.933(0.212-72.931) | 0.358 |
| Distant metastasis | Yes/No | 7.603(1.955-29.565) | 0.003 | 41.871(2.881-608.639) | 0.006 |
| MAN1C1 | High/Low | 2.970(1.033-8.541) | 0.043 | 3.783(1.047-13.666) | 0.042 |
HR hazard ratio, 95% CI 95% confidence interval
MAN1C1 inhibits the proliferation and promotes apoptosis of ccRCC cells in vitro
To determine whether MAN1C1 is directly involved in promoting cell death and inhibits tumor growth, we performed over-expression experiments through transfection with MAN1C1. From the CCK8 assay, we observed that the over-expression of MAN1C1 reduced the proliferation of 786-O and OS-RC-2 cells compared with the negative control (NC) cells transfected (Fig. 3A). The ccRCC suppressing role of MAN1C1 was confirmed by the results of the clonogenic assay demonstrating that over-expression of MAN1C1 reduced colony numbers in both ccRCC cell lines (Fig. 3B). In addition, over-expression of MAN1C1 resulted in increased apoptosis in both 786-O and OS-RC-2 cells compared with mimic, B2 (PE Annexin V positive representing the cells in early apoptosis) and B4 (PE Annexin V and 7-AAD double positive representing the cells in late apoptosis) regions in the plot, which were accounted as the apoptotic cells (Fig. 3C). These results suggest that MAN1C1 inhibits the proliferation and promotes apoptosis of ccRCC cells in vitro.
Overexpression of MAN1C1 affects tumor cell growth and western blot analysis of apoptotic-related proteins. (A) Overexpression of MAN1C1 in 786-O and OS-RC-2 cells by transfecting resulted in decrease cellular growth (B) and colony formation (C) and promotion of apoptosis (D) in 786-O and OS-RC-2 cell lines. The results are presented as the mean ± SD (n=3). *, P < 0.05; **, P < 0.01; ***, P < 0.001. D: 786-O and OS-RC-2 cells were transfected with MAN1C1 or control for 72 hours before being subjected to protein extraction and western blot with the indicated antibodies. (E) experiments were quantified from D measuring the intensity of apoptotic-related proteins relative to the GAPDH (loading control) (*, P < 0.05 and **, P < 0.01). The bars indicate mean ± SD (n=3).Abbreviations: SD, standard deviation; MAN1C1 overexpression plasmid; control,empty plasmid.
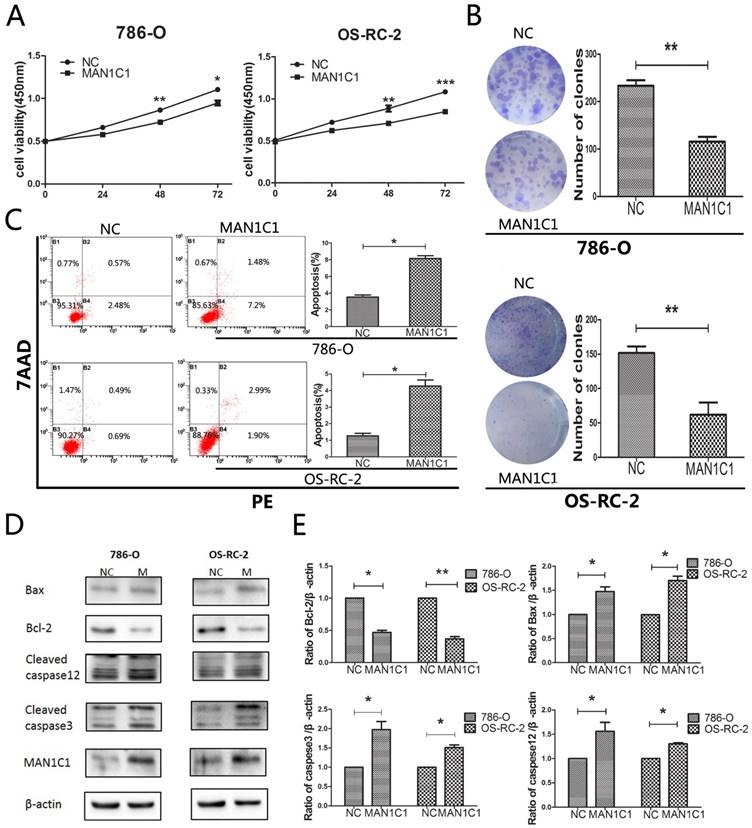
Next we confirmed an increased expression of MAN1C1 in cells transfected with pcDNA-MAN1C1 plasmid by using western blotting. By contrast, the cells transfected with empty vector alone had very low levels of MAN1C1 expression (Fig. 3D, E). We also examined whether MAN1C1 over-expression is correlated with alteration in the expression of apoptosis regulating proteins Bax and Bcl-2. As shown in figure, western blot results demonstrated that over-expression of MAN1C1 increased the levels of Bax by approximately 1.48-fold (P<0.05) in 786-O cells, and 1.71-fold (P<0.05) in OS-RC-2 cells. On the contrary, the levels of Bcl-2 in 786-O and OS-RC-2 cells were decreased appropriate 53% (P<0.01, Bcl-2 in 786-O) and 62% (P<0.01, Bcl-2 in OS-RC-2).
To further assess the role of MAN1C1 in apoptosis, we subsequently evaluated the expression of apoptosis-related proteins, including proapoptotic cleaved caspase-3 and caspase-12. Western blot results indicated that over-expression of MAN1C1 increased the levels of cleaved caspase-3 by approximately 1.97-fold (P<0.05) in 786-O cells, 1.51-fold (P<0.05) in OS-RC-2 cells, and increased the levels of cleaved caspase-12 by approximately 1.6-fold (P<0.05) in 786-O cells, 1.3-fold (P<0.05) in OS-RC-2 cells.
MAN1C1 inhibits ccRCC cell migration and invasion
According to the characteristics of renal cell carcinoma metastasis, studies of a variety of tumor types in the past decade have indicated that EMT is often activated during cancer progression and metastasis. Cell migration has an important role in cancer metastasis. To investigate the potential role of MAN1C1 in ccRCC progression, we over-expressed MAN1C1 exogenously in two ccRCC cell lines: 786-O and OS-RC-2. The effects of MAN1C1 expression on the migration and invasion ability of ccRCC cells were also evaluated by scratch and transwell assays.
The results showed that the inhibition of cell migration in 786-O and OS-RC-2 cells was remarkable upon MAN1C1 over-expression in wound-healing experiments (Fig. 4A, B). The analysis of 786-O and OS-RC-2 cell invasion also demonstrated MAN1C1 over-expression significantly decreased the invasive properties of these tumour cells (Fig. 4C, D). To further confirm the results, we investigated the effect of MAN1C1 over-expression on protein expression levels of the epithelial marker E-cadherin and the mesenchymal markers N-cadherin and vimentin. As shown in (Fig. 4E, F), western blot results indicated that MAN1C1 over-expression increased the levels of E-cadherin by approximately 1.34-fold (P<0.05) in 786-O cells and 1.26-fold (P<0.05) in OS-RC-2 cells.
The inhibition of MAN1C1 reduces cell motility of renal cancer. ccRCC cell lines 786-O and OS-RC-2 were transfected with MAN1C1 or control, respectively. (A) The migration rate of 786-O and OS-RC-2 cells pre-treated as described for 24 h was observed through wound-healing assays. (B) Histogram represents the statistical analysis of wound-healing assays (***, P < 0.001 vs control group). (C) Trans-well invasion assays were conducted to observe the invasive cells in 786-O and OS-RC-2. (D) Histogram represents the statistical analysis of trans-well invasion assays(***, P<0.001 vs control group). (E) The expression of tumor metastasis-related proteins (E-Ca/N-Ca/Vime) in 786-O and OS-RC-2 cells was detected through western blotting. β-ACTING was used as an endogenous reference. (F) Histogram represents the statistical analysis of the changes in the expression of metastasis-related proteins. The bars showed means ± SD of three independent experiments.
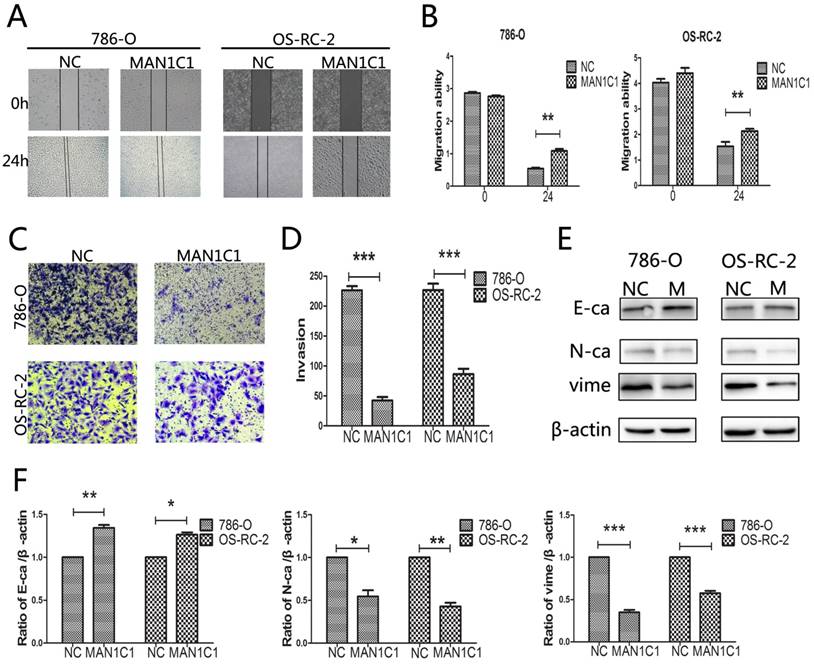
On the contrary, the levels of N-cadherin and vimentin in 786-O and OS-RC-2 cells were reduced approximately 46% (P<0.05, N-cadherin in 786-O), 57% (P<0.01, N-cadherin in OS-RC-2), and 75% (P<0.05, vimentin in 786-O), 43% (P<0.01, vimentin in OS-RC-2). This indicated that EMT is suppressed following MAN1C1 over-expression. In conclusion, our results suggest that MAN1C1 plays a tumor-suppressive role via regulation of cell proliferation and migration in ccRCC.
Discussion
Results from the previous studies suggest that increased expression of α-1,2 mannosidases has been associated with many cancers[22], and our observation that MAN2C1 associates with prostate tumourigenesis[23] is consistent with the report that MAN2C1 is associated with tumourigenic activity in B cells and nasopharynegeal carcinoma[24, 25]. Interestingly, recently-developed MAN2C1 transgenic mice are prone to enhance tumour growth, invasion and metastasis[26]. Also, MAN1A1 is associated with breast cancer[27], MAN1A2 is a prognostic indicator in B cell lymphoma[28] and MAN1B1 is up-regulated and promotes transformation phenotypes in HCC[18]. Whereas MAN1C1 was dramatically suppressed at an early stage of liver cancer development (stageI)[19], which is consistent with the report that MAN1C1 is probably a potential tumor suppressor in Wilms tumors[20]. In our study, MAN1C1 was down-regulated in ccRCC tissues and was closely associated with advanced TNM stage and poorer overall survival.
MAN1C1 Re-expression in 786-O and OS-RC2 kidney cancer cells indicated inhibition of cell proliferation and promotion of apoptosis. In this study, we found that transfection of 786-O and OS-RC-2 cancer cells with a MAN1C1 expression vector decreased cell number in culture (Fig. 3A). In addition, we observed an increase in apoptosis in these cells. Thus we believe that the decrease of cell proliferation is a consequence of the apoptotic role of MAN1C1. This result is similar to the one reported by Krepischi[20]. Our study has uncovered a novel function of MAN1C1 and suggested its possible contribution to cell apoptosis in cancer. We think in the early stages of ccRCC, the down-regulation of MAN1C1 causes the dysregulation of N-glycosylation and results in modest ER stress due to the accumulation of uncleaved glycoprotein precursors. However, at the later stages, the accumulation of a large number of uncleaved glycoprotein precursors may lead to the accumulation of greater ER stress and eventually cancer. Active experiments are currently underway to test this hypothesis.
Invasion and metastasis are two of the most important hallmarks of malignant tumors and are factors directly associated with mortality in human cancers[29]. Therefore, it is essential to identify factors involved in invasion and metastasis of tumor cells. Furthermore, EMT, which facilitates cell motility and invasion, is a key event in tumor invasion and metastasis[30,31]. For example, aberrant N-glycosylation of E-cadherin is associated with carcinogenesis secondary to altered cell-cell adhesion and communication[32].
We found that MAN1C1 over-expression in OS-RC-2 and 786-O cell lines can inhibit the transformation of cells into a more mesenchymal phenotype, which is evident from the upregulation of epithelial genes, such as E-cadherin, and the downregulation of mesenchymal genes, such as N-cadherin and vimentin (Fig. 4E).
In summary, here we have identified a novel role of MAN1C1 in apoptosis, which will help to understand the over-expression of MAN1C1 in suppressing the growth potential of cancer and leading to a significant reduction in cell proliferation, colony formation, migration and invasion. These data suggest that MAN1C1 may be a useful prognostic biomarker and potential therapeutic target in ccRCC, with the potential to lead to better outcomes for patients with these poor prognosis malignancies.
Acknowledgements
This work was supported by National Natural Science Foundation of China [grant No.81171996; 81272289; 81872084]. The First Affiliated Hospital of Harbin Medical University Science Foundation [grant No. 2015B011].
Author contributions
Li Haoming mainly carried out the experiments, organized the data and wrote the paper. Wang Gang and Jian Wengang helped with the experiments.Yu Yipeng helped analyze the data. Zhang Daming and Wang Yongquan helped improve the paper. Wang Tengda performed CCK8 assay. Meng Yuyang and Yuan Chao accomplished clinical sample collection. Zhang Cheng carried out the experiment design and manuscript drafting. All authors had edited and approved the final manuscript.
Ethics approval and consent to participate
This study was conducted at Department of Urology, The First Affiliated Hospital of Harbin Medical University, Harbin, Heilongjiang, China. This research study was approved by the Institutional Review Board of Harbin Medical University, and the subjects provided informed consent.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. International journal of cancer. 2010;127:2893-917
2. Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M. et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. International journal of cancer. 2015;136:E359-86
3. Rini BI, Campbell SC, Escudier B. Renal cell carcinoma. Lancet. 2009;373:1119-32
4. Ma W, Tao L, Wang X, Liu Q, Zhang W, Li Q. et al. Sorafenib Inhibits Renal Fibrosis Induced by Unilateral Ureteral Obstruction via Inhibition of Macrophage Infiltration. Cellular physiology and biochemistry: international journal of experimental cellular physiology, biochemistry, and pharmacology. 2016;39:1837-49
5. Tian X, Dai S, Sun J, Jiang S, Sui C, Meng F. et al. Inhibition of MDM2 Re-Sensitizes Rapamycin Resistant Renal Cancer Cells via the Activation of p53. Cellular physiology and biochemistry: international journal of experimental cellular physiology, biochemistry, and pharmacology. 2016;39:2088-98
6. McDermott DF, Regan MM, Clark JI, Flaherty LE, Weiss GR, Logan TF. et al. Randomized phase III trial of high-dose interleukin-2 versus subcutaneous interleukin-2 and interferon in patients with metastatic renal cell carcinoma. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2005;23:133-41
7. Roth J, Zuber C, Park S, Jang I, Lee Y, Kysela KG. et al. Protein N-glycosylation, protein folding, and protein quality control. Molecules and cells. 2010;30:497-506
8. Garcia-Vallejo JJ, Van Dijk W, Van Het Hof B, Van Die I, Engelse MA, Van Hinsbergh VW. et al. Activation of human endothelial cells by tumor necrosis factor-alpha results in profound changes in the expression of glycosylation-related genes. Journal of cellular physiology. 2006;206:203-10
9. Zeng Y, Pan YT, Asano N, Nash RJ, Elbein AD. Homonojirimycin and N-methyl-homonojirimycin inhibit N-linked oligosaccharide processing. Glycobiology. 1997;7:297-304
10. Tempel W, Karaveg K, Liu ZJ, Rose J, Wang BC, Moremen KW. Structure of mouse Golgi alpha-mannosidase IA reveals the molecular basis for substrate specificity among class 1 (family 47 glycosylhydrolase) alpha1,2-mannosidases. The Journal of biological chemistry. 2004;279:29774-86
11. Barlowe CK, Miller EA. Secretory protein biogenesis and traffic in the early secretory pathway. Genetics. 2013;193:383-410
12. Herscovics A. Structure and function of Class I alpha 1,2-mannosidases involved in glycoprotein synthesis and endoplasmic reticulum quality control. Biochimie. 2001;83:757-62
13. Molinari M. N-glycan structure dictates extension of protein folding or onset of disposal. Nature chemical biology. 2007;3:313-20
14. Pan S, Cheng X, Sifers RN. Golgi-situated endoplasmic reticulum alpha-1, 2-mannosidase contributes to the retrieval of ERAD substrates through a direct interaction with gamma-COP. Molecular biology of the cell. 2013;24:1111-21
15. Hazari YM, Bashir A, Haq EU, Fazili KM. Emerging tale of UPR and cancer: an essentiality for malignancy. Tumour biology: the journal of the International Society for Oncodevelopmental Biology and Medicine. 2016;37:14381-90
16. Bianchi P, Fermo E, Vercellati C, Boschetti C, Barcellini W, Iurlo A. et al. Congenital dyserythropoietic anemia type II (CDAII) is caused by mutations in the SEC23B gene. Human mutation. 2009;30:1292-8
17. Damme M, Morelle W, Schmidt B, Andersson C, Fogh J, Michalski JC. et al. Impaired lysosomal trimming of N-linked oligosaccharides leads to hyperglycosylation of native lysosomal proteins in mice with alpha-mannosidosis. Molecular and cellular biology. 2010;30:273-83
18. Pan S, Cheng X, Chen H, Castro PD, Ittmann MM, Hutson AW. et al. ERManI is a target of miR-125b and promotes transformation phenotypes in hepatocellular carcinoma (HCC). PloS one. 2013;8:e72829
19. Tu HC, Hsiao YC, Yang WY, Tsai SL, Lin HK, Liao CY. et al. Up-regulation of golgi alpha-mannosidase IA and down-regulation of golgi alpha-mannosidase IC activates unfolded protein response during hepatocarcinogenesis. Hepatology communications. 2017;1:230-47
20. Krepischi ACV, Maschietto M, Ferreira EN, Silva AG, Costa SS, da Cunha IW. et al. Genomic imbalances pinpoint potential oncogenes and tumor suppressors in Wilms tumors. Molecular cytogenetics. 2016;9:20
21. Aissani B, Zhang K, Wiener H. Follow-up to genome-wide linkage and admixture mapping studies implicates components of the extracellular matrix in susceptibility to and size of uterine fibroids. Fertility and sterility. 2015;103:528-34 e13
22. Olszewska E, Borzym-Kluczyk M, Rzewnicki I, Wojtowicz J, Rogowski M, Pietruski JK. et al. Possible role of alpha-mannosidase and beta-galactosidase in larynx cancer. Contemporary oncology. 2012;16:154-8
23. He L, Fan C, Kapoor A, Ingram AJ, Rybak AP, Austin RC. et al. alpha-Mannosidase 2C1 attenuates PTEN function in prostate cancer cells. Nature communications. 2011;2:307
24. Shi GX, Wang Y, Liu Y, Cui W, Zhao FT, Zhu LP. Long-term expression of a transferred gene in Epstein-Barr virus transformed human B cells. Scandinavian journal of immunology. 2001;54:265-72
25. Yue W, Jin YL, Shi GX, Liu Y, Gao Y, Zhao FT. et al. Suppression of 6A8 alpha-mannosidase gene expression reduced the potentiality of growth and metastasis of human nasopharyngeal carcinoma. International journal of cancer. 2004;108:189-95
26. Xiang ZG, Jiang DD, Liu Y, Zhang LF, Zhu LP. hMan2c1 transgene promotes tumor progress in mice. Transgenic research. 2010;19:67-75
27. Milde-Langosch K, Karn T, Schmidt M, zu Eulenburg C, Oliveira-Ferrer L, Wirtz RM. et al. Prognostic relevance of glycosylation-associated genes in breast cancer. Breast cancer research and treatment. 2014;145:295-305
28. Kim SJ, Sohn I, Do IG, Jung SH, Ko YH, Yoo HY. et al. Gene expression profiles for the prediction of progression-free survival in diffuse large B cell lymphoma: results of a DASL assay. Annals of hematology. 2014;93:437-47
29. Chen Z, Zhang J, Zhang Z, Feng Z, Wei J, Lu J. et al. The putative tumor suppressor microRNA-30a-5p modulates clear cell renal cell carcinoma aggressiveness through repression of ZEB2. Cell death & disease. 2017;8:e2859
30. Kang Y, Massague J. Epithelial-mesenchymal transitions: twist in development and metastasis. Cell. 2004;118:277-9
31. Thiery JP. Epithelial-mesenchymal transitions in tumour progression. Nature reviews Cancer. 2002;2:442-54
32. Pinho SS, Seruca R, Gartner F, Yamaguchi Y, Gu J, Taniguchi N. et al. Modulation of E-cadherin function and dysfunction by N-glycosylation. Cellular and molecular life sciences: CMLS. 2011;68:1011-20
Author contact
Corresponding author: Cheng Zhang, Department of Urology, The First Affiliated Hospital of Harbin Medical University, Harbin 150001, Heilongjiang, China. Email: doctorcheng77com

 Global reach, higher impact
Global reach, higher impact