Impact Factor ISSN: 1837-9664
J Cancer 2026; 17(5):952-967. doi:10.7150/jca.126434 This issue Cite
Review
The Role of Traditional Chinese Medicine in Metabolic Reprogramming, Tumor microenvironment, and Macrophage Regulation: Mechanisms and Advances in Preclinical Studies
1. Hangzhou TCM Hospital of Zhejiang Chinese Medical University (Hangzhou Hospital of Traditional Chinese Medicine), No. 453 Tiyuchang Road, Xihu District, Hangzhou 310007, China
2. Zhejiang Hospital, No. 12 Linyin Road, Xihu District, Hangzhou 310007, China.
Received 2025-10-9; Accepted 2026-2-10; Published 2026-4-22
Abstract
Background: Cancer metabolism reprogramming is a hallmark of tumorigenesis, playing a critical role in tumor progression and therapeutic resistance. Macrophages in the Tumor microenvironment (TME) play a crucial role in tumor progression and immune system evasion. Traditional Chinese Medicine (TCM) has shown potential in influencing cancer metabolism and immune functions, offering new therapeutic possibilities.
Objective: This review explores the intersection of TCM, metabolic reprogramming, and macrophage polarization in cancer, focusing on their molecular mechanisms and therapeutic implications. The study further seeks to summarize preclinical evidence supporting TCM's role in modulating tumor metabolism and immune microenvironments, offering a holistic approach to cancer therapy.
Methods: In this study, we systematically reviewed the latest research progress in the last five years on the role of traditional Chinese medicine in regulating tumor metabolic reprogramming, TME and macrophage polarization. A comprehensive literature reviews the metabolic shifts in cancer cells, including glycolysis, oxidative phosphorylation, and lipid metabolism, and how TCM compounds restore metabolic balance. Studies on macrophage polarization (M1/M2) and their regulatory roles in cancer immunity, as influenced by TCM, were analyzed. Preclinical research highlighting TCM's dual impact on tumor metabolism and the TME was also evaluated.
Results: TCM compounds demonstrate the ability to target key metabolic pathways, such as glycolysis and lipid metabolism, restoring metabolic homeostasis in cancer cells. Additionally, TCM has been shown to regulate macrophage polarization within the TME, enhancing M1 macrophage-mediated anti-tumor immunity and suppressing M2 macrophage-mediated tumor promotion. Preclinical studies further support TCM's capacity to modulate the TME and improve therapeutic outcomes when used in combination with conventional cancer therapies.
Conclusions: TCM integrates metabolic reprogramming and immunomodulation to provide a comprehensive treatment strategy for cancer. However, translating these findings into clinical practice remains challenging. Future research should focus on validating the exact mechanisms of TCM in tumor metabolism and immunity through clinical trials and investigating the synergistic effects of TCM with modern cancer therapies, such as immunotherapy and targeted therapies. This may facilitate the use of TCM as an adjuvant therapy to improve the efficacy of existing cancer treatments.
Keywords: Traditional Chinese Medicine (TCM), Metabolic Reprogramming, Macrophage Polarization
1. Introduction
Cancer remains a leading cause of death worldwide. According to statistics, an estimated 20 million people will be diagnosed with cancer by 2022, and the number of diagnoses is expected to increase by 77 percent by 2050 to 35 million [1]. And despite significant advances in treatment modalities, including surgery, chemotherapy, and immunotherapy, therapeutic resistance and relapse continue to challenge effective management. Increasing evidence indicates that tumor progression and therapy resistance are closely linked to coordinated alterations in cellular metabolism and immune regulation within the TME [2]. A hallmark feature of cancer is metabolic reprogramming, wherein tumor cells alter their metabolic pathways to support uncontrolled growth, survival, and metastasis. Importantly, tumor metabolic reprogramming not only fuels malignant cell growth but also reshapes the functional state of immune cells, particularly tumor-associated macrophages, thereby reinforcing immune suppression within the TME [3]. Recent studies have identified tumor-associated macrophages (TAMs) as key players in shaping the TME (TME), modulating both cancer progression and the immune response [4-6]. Macrophages can exist in two major phenotypic states: M1 macrophages, which are anti-tumoral, and M2 macrophages, which promote tumor progression and immune evasion. The polarization of macrophages within the TME can significantly influence tumor progression and the response to treatment [7-9].
Traditional Chinese Medicine (TCM) has a long history of use in cancer therapy and is gaining increasing attention due to its potential to modulate multiple biological pathways, including metabolic reprogramming and immune modulation [10-12]. TCM offers a holistic approach to cancer treatment by balancing the body's internal environment, improving systemic immunity, and influencing cancer cell metabolism [13-15]. This multi-target and system-level regulatory capacity positions TCM as a potential immunometabolic modulator rather than a single-target cytotoxic intervention [16]. Many compounds present in TCM, such as flavonoids, alkaloids, and polysaccharides, have been found to inhibit cancer cell growth by modulating key metabolic pathways such as glycolysis, oxidative phosphorylation, and lipid metabolism [17-19].
Emerging evidence suggests that TCM may also impact the macrophage polarization within the TME, shifting the balance from an M2 (tumor-promoting [4] phenotype to an M1 (tumor-suppressing) phenotype, thereby enhancing anti-tumor immunity and potentially overcoming immune evasion mechanisms [20-22]. Such macrophage reprogramming is increasingly recognized as a key determinant of therapeutic responsiveness to chemotherapy and immunotherapy. The combination of metabolic reprogramming and immune modulation through TCM could provide a novel therapeutic strategy for cancer treatment [23-25].
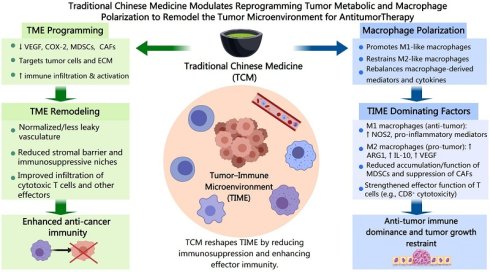
As shown in Figure 1, this review aims to explore the intersection of TCM, metabolic reprogramming, and macrophage modulation in cancer therapy, with an emphasis on the molecular mechanisms through which TCM compounds regulate both cancer metabolism and macrophage polarization. We will also discuss preclinical and clinical studies that demonstrate the potential of TCM to enhance the efficacy of conventional cancer therapies by modulating the TME, thereby offering a promising adjunctive approach to cancer treatment.
Mechanism of tumor suppression via immune-metabolic modulation by TCM.
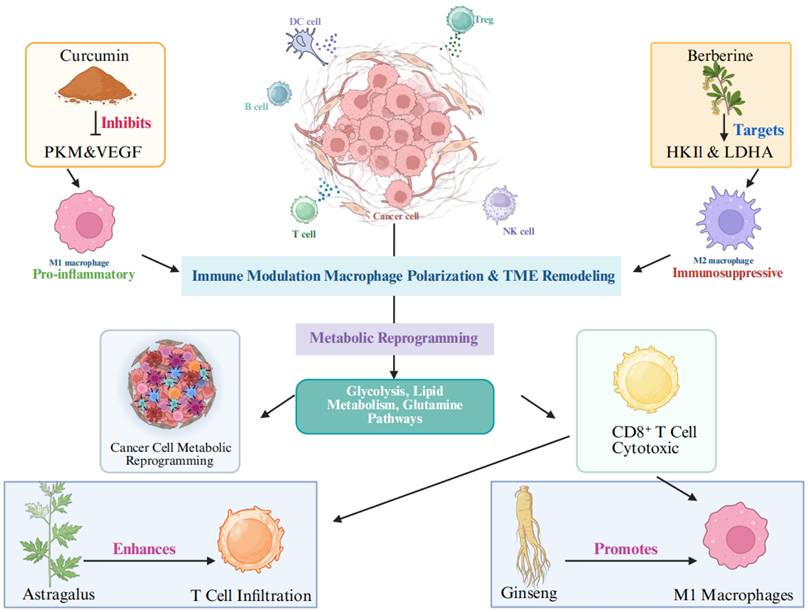
2. Cancer Metabolic Reprogramming
2.1. Overview of metabolic reprogramming in cancer
Metabolic reprogramming enables tumor cells to adapt to the hostile microenvironment, sustain rapid proliferation, and evade cell death [26-27]. This feature was formally recognized as one of the “Hallmarks of Cancer”. In healthy cells, glucose metabolism primarily relies on oxidative phosphorylation (OXPHOS) in mitochondria. However, even under aerobic conditions, cancer cells favor glycolysis for adenosine triphosphate (ATP) production, a phenomenon known as the Warburg effect. This metabolic shift not only provides rapid ATP but also supplies intermediates for biosynthesis, essential for cellular proliferation [28-29].
As shown in Figure 2, beyond the Warburg effect, cancer cells exhibit reprogramming in several other metabolic pathways, including enhanced lipid metabolism, glutamine dependency, and lactate-driven immune suppression. These pathways collectively form the foundation for tumor proliferation, survival, and metastasis [30-32].
TCM target key metabolic reprogramming pathways in cancer.
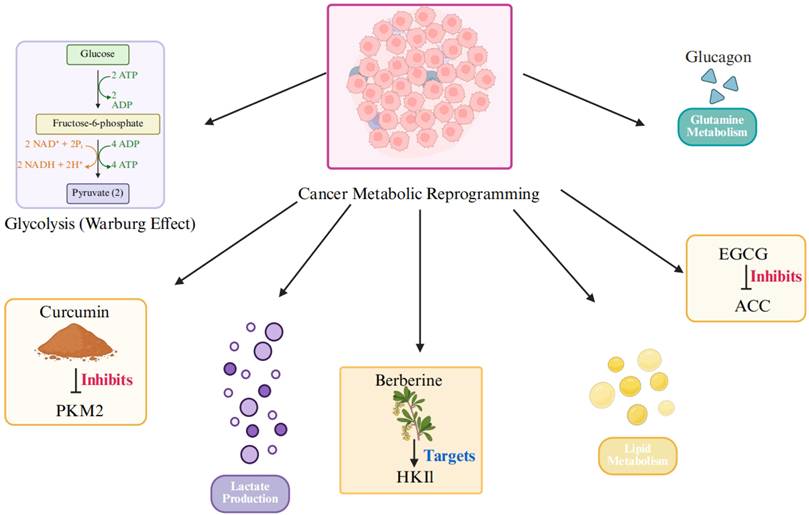
2.2. Major Reprogrammed Metabolic Pathways in Cancer
2.2.1. Permission to reuse and Copyright
The Warburg effect is characterized by the preference of cancer cells for glycolysis over OXPHOS, even in the presence of oxygen. Glycolysis allows cancer cells to quickly generate metabolic intermediates required for nucleotide, protein, and lipid synthesis [33-34]. Key enzymes in this pathway, such as Hexokinase II (HKII), Pyruvate Kinase M2 (PKM2), and Lactate Dehydrogenase A (LDHA), are often overexpressed in cancer cells and regulated by oncogenes like MYC and HIF-1α [35-36]. Targeting glycolysis has shown significant therapeutic potential [37-38]. For instance, Berberine, a bioactive compound from Coptis chinensis, activates AMPK and downregulates HKII expression, reducing glycolytic flux and lactate production, ultimately suppressing tumor growth [39]. Similarly, Curcumin inhibits PKM2 activity, preventing the conversion of pyruvate to lactate, which significantly impairs colorectal cancer cell proliferation [40].
2.2.2. Lactate Metabolism and Tumor
Microenvironment lactate, once considered a waste product of glycolysis, has emerged as a critical regulator of the TME. Accumulated lactate acidifies the TME, which suppresses the activity of effector T cells and NK cells, while promoting the expansion of regulatory T cells (Tregs), thereby facilitating immune evasion [41-44]. Baicalin, a flavonoid from Scutellaria baicalensis, has been shown to reduce lactate production, reprogramming the TME and enhancing T cell-mediated anti-tumor immunity [45-47].
2.2.3. Lipid Metabolism
Lipid Metabolism is essential for cancer cells to support membrane biosynthesis, energy storage, and signaling. Enhanced expression of Fatty Acid Synthase (FAS) and Acetyl-CoA Carboxylase (ACC) is a hallmark of reprogrammed lipid metabolism in various cancer cells, including breast and prostate cancer [48]. Epigallocatechin gallate (EGCG), a green tea-derived polyphenol, inhibits FAS activity, leading to reduced lipid synthesis and subsequent inhibition of tumor growth and invasion [49,50].
2.2.4. Glutamine Metabolism
Many cancer cells exhibit glutamine addiction, relying on glutamine as a source of carbon and nitrogen for nucleotide, amino acid, and lipid biosynthesis. Through the action of Glutaminase (GLS1), glutamine is converted into glutamate, which enters the TCA cycle to support anabolic metabolism. Astragalus polysaccharides, extracted from Astragalus membranaceus, downregulate GLS1 expression, reducing glutamine dependency and suppressing tumor growth [51,52].
2.3. Metabolic Reprogramming and Therapy Resistance
2.3.1. Chemotherapy Resistance
Enhanced glycolysis increases ATP production, enabling cancer cells to effectively pump out chemotherapeutic agents via ATP-dependent efflux pumps, reducing drug sensitivity. Overexpression of HKII and LDHA has been closely linked to resistance to drugs like cisplatin and doxorubicin [53].
2.3.2. Immunotherapy Resistance
Lactate accumulation in the TME suppresses the activity of effector immune cells, diminishing the efficacy of immune checkpoint inhibitors such as anti-PD-1 and anti-CTLA-4 antibodies. Baicalin, by reducing lactate production, has been shown to enhance the efficacy of these therapies in preclinical models [54].
2.4. The Roles of Traditional Chinese Medicine in Targeting Metabolic Reprogramming
2.4.1. Restoring Metabolic Balance
Berberine activates AMPK and inhibits mTOR signaling, effectively disrupting the metabolic demands of cancer cells and restoring energy homeostasis. Tanshinone IIA, derived from Salvia miltiorrhiza, modulates the PPARγ pathway, reducing lipid accumulation and significantly inhibiting breast cancer cell migration and invasion [55-56].
2.4.2. Improving the Metabolic Environment
Baicalin reduces lactate levels in the TME, alleviating the acidic microenvironment and enhancing T cell function [54]. Curcumin targets the PDK1/LDHA axis, decreasing lactate production and reversing immune suppression in the TME [57].
2.4.3. Improving the Metabolic Environment
EGCG inhibits FASN, sensitizing cancer cells to chemotherapeutic agents such as paclitaxel [58]. Astragalus polysaccharides have demonstrated synergistic effects with PD-1 inhibitors, significantly enhancing anti-tumor efficacy in mouse models [59-60].
3. The TME: An Expanded Background
3.1. Overview of the TME
The TME (TME) is a dynamic and complex ecosystem surrounding cancer cells, comprising various stromal cells, immune cells, vascular endothelial cells, fibroblasts, extracellular matrix (ECM) components, and signaling molecules. Unlike normal tissue environments, the TME plays a dual role in tumor development: it can either suppress tumor progression in its early stages or promote cancer growth, metastasis, and resistance to therapy as the tumor evolves [61-62].
The TME is highly heterogeneous and constantly evolving, influenced by interactions between cancer cells and their surrounding components. These interactions shape cancer progression, immune evasion, angiogenesis, and resistance to conventional and targeted therapies. By modifying the TME, cancer cells create an environment conducive to their survival and proliferation. Importantly, the TME is not only a passive bystander but an active participant in the cancer progression process, making it an important therapeutic target in modern oncology [63-65].
3.2. Key Components of the TME
3.2.1. Immune Cells and Immune Modulation
The TME hosts a variety of immune cells, including tumor-associated macrophages (TAMs), myeloid-derived suppressor cells (MDSCs), regulatory T cells (Tregs), dendritic cells (DCs), natural killer (NK) cells, and cytotoxic CD8+ T cells. These immune cells are critical in determining the fate of tumor growth and metastasis, as they mediate immune surveillance and immune evasion mechanisms [66-67].
3.2.2. Tumor-Associated Macrophages (TAMs)
TAMs are highly plastic and can polarize into either the M1 phenotype (pro-inflammatory and anti-tumorigenic) or the M2 phenotype (anti-inflammatory and pro-tumorigenic). TAMs in the TME are predominantly polarized towards the M2 phenotype, secreting cytokines like IL-10 and TGF-β, which suppress immune responses and promote tumor progression. High M2 macrophage infiltration is associated with poor prognosis in many cancers, including breast, lung, and ovarian cancer [68-70].
3.2.3. Regulatory T Cells (Tregs)
Tregs are recruited to the TME through chemokines like CCL22 and CCL28. Once in the TME, Tregs suppress cytotoxic T cell function and secrete immunosuppressive cytokines, such as IL-10 and TGF-β, creating an immunosuppressive microenvironment. This immune evasion mechanism allows tumors to grow unchecked and resist immune-based therapies [71-72].
3.2.4. Effector Cells (CD8+ T Cells and NK Cells)
Despite their tumor-killing potential, effector immune cells often become dysfunctional in the TME due to metabolic competition, hypoxia, and inhibitory signaling pathways. For instance, lactate accumulation from the Warburg effect directly inhibits CD8+ T cell function and prevents NK cell activation [73-74].
3.2.5. Vasculature and Angiogenesis
Tumor angiogenesis is one of the hallmarks of cancer and plays a critical role in providing nutrients and oxygen to rapidly proliferating cancer cells. The process is driven by vascular endothelial growth factor (VEGF), which stimulates the formation of new blood vessels. However, these tumor-associated blood vessels are often aberrant, leaky, and poorly organized, leading to hypoxia within the TME. Hypoxia further promotes tumor progression by stabilizing hypoxia-inducible factor 1-alpha (HIF-1α), which enhances glycolysis, angiogenesis, and metastasis. Abnormal vasculature also limits the delivery of chemotherapeutic agents and immune cells to the tumor core, posing significant challenges to effective treatment [75,76].
3.2.6. Cancer-Associated Fibroblasts (CAFs)
Cancer-associated fibroblasts are a major stromal component of the TME and contribute to tumor progression by remodeling the ECM, secreting pro-tumorigenic cytokines, and enhancing angiogenesis. CAFs secrete fibroblast growth factor (FGF), transforming growth factor-beta (TGF-β), and matrix metalloproteinases (MMPs), which degrade the ECM and facilitate tumor invasion and metastasis. CAFs also play a role in creating drug resistance by forming physical barriers that prevent drug penetration into the tumor [77,78].
3.2.7. Extracellular Matrix (ECM)
The ECM is a structural scaffold that provides mechanical and biochemical support to cells within the TME. Tumor cells interact with the ECM via integrins and other cell surface receptors, which mediate cell adhesion, migration, and survival. Tumor-associated ECM undergoes continuous remodeling, driven by proteases such as MMPs. This remodeling creates tracks for cancer cell invasion and alters signaling pathways that promote tumor growth [79-80].
3.2.8. Metabolic Reprogramming in the TME
Cancer cells in the TME often undergo metabolic reprogramming to survive in nutrient-deprived and hypoxic conditions. The reliance on glycolysis (Warburg effect) leads to lactate accumulation, acidifying the TME and suppressing immune cell activity. In addition, tumor cells compete with immune cells for nutrients like glucose and glutamine, depriving immune effector cells of the energy needed for their functions [61,34,80]. Metabolic competition is a critical driver of immune evasion within the TME.
3.3. The Role of the TME in Cancer Progression
The TME (TME) actively participates in multiple stages of cancer progression by fostering tumor growth through the provision of essential growth factors such as EGF and VEGF, which sustain uncontrolled cellular proliferation. It facilitates immune evasion by recruiting immunosuppressive cells like Tregs and MDSCs, thereby creating a permissive environment for tumors to escape immune surveillance. Additionally, the TME promotes metastasis via CAF-mediated ECM remodeling, enabling tumor cells to invade surrounding tissues and enter the bloodstream for distant dissemination. Furthermore, hypoxia, aberrant vasculature, and ECM modifications within the TME contribute to therapy resistance by establishing physical and biochemical barriers that diminish the efficacy of chemotherapy, radiotherapy, and immunotherapy [81].
3.4. Targeting the TME in Cancer Therapy
The TME (TME) plays a central role in cancer development, which makes it a highly promising therapeutic target. Current therapeutic strategies for TME are focused on four key directions. In the area of immune regulation, researchers have effectively restored the body's immune surveillance function by reprogramming tumor-associated macrophages (TAMs) from a pro-tumorigenic M2 phenotype to an anti-tumorigenic M1 phenotype, while blocking the recruitment of regulatory T cells (Tregs) [69,82].
Anti-angiogenic therapy is another important strategy, and VEGF-targeted drugs represented by bevacizumab are able to normalize the disordered tumor vascular system, thus significantly improving the delivery efficiency of other therapeutic agents [83].
In terms of metabolic intervention, metabolic crosstalk between tumor cells and mesenchymal stromal cells within the TME can be effectively disrupted by strategies such as inhibiting lactate dehydrogenase A (LDHA) or targeting key enzymes of glycolysis [55,84].
Important advances have also been made in interventions targeting the extracellular matrix (ECM), which not only inhibit tumor invasion and metastasis, but also significantly enhance the tissue penetration of chemotherapeutic agents by inhibiting matrix metalloproteinase (MMP) activity or blocking ECM proteins secreted by cancer-associated fibroblasts (CAF). These multipronged therapeutic strategies provide new solution ideas to overcome TME-mediated therapeutic resistance [25,85].
3.5. The Role of Traditional Chinese Medicine in TME Modulation
Traditional Chinese Medicine (TCM) offers a unique advantage in targeting the TME due to its multi-target and multi-pathway mechanisms. Baicalin from Scutellaria baicalensis has been shown to inhibit IL-10 and TGF-β secretion, reprogramming TAMs from the M2 to M1 phenotype, thereby enhancing anti-tumor immunity [86]. Curcumin reduces VEGF expression and stabilizes tumor vasculature, improving oxygen delivery and reducing hypoxia-induced tumor growth [87-88]. Astragalus polysaccharides; an extract from Astragalus membranaceus plant enhance T cell infiltration into the TME and boost the efficacy of immune checkpoint inhibitors [89-90].
4. Macrophage Regulation: Detailed Insights into M1 and M2 Polarization Pathways and the Role of Traditional Chinese Medicine
4.1. Overview of Macrophage Polarization
Macrophages exhibit remarkable plasticity and can adopt diverse phenotypes in response to environmental signals. Broadly, macrophages are polarized into two distinct functional states. Known as “classically activated” macrophages, M1 macrophages are induced by pro-inflammatory signals such as interferon-gamma (IFN-γ) and lipopolysaccharide (LPS). These macrophages produce pro-inflammatory cytokines such as TNF-α, IL-12, and IL-1β, and are involved in pathogen clearance and anti-tumor immunity [18,91]. Referred to as “alternatively activated” macrophages, M2 macrophages are polarized by anti-inflammatory signals such as IL-4, IL-10, and TGF-β [18,92]. They produce cytokines like IL-10 and TGF-β, which suppress immune responses, promote tissue remodeling, and enhance tumor progression (Figure 3).
Dynamic M1/M2 macrophage polarization orchestrates TME remodeling through metabolic-immune crosstalk.
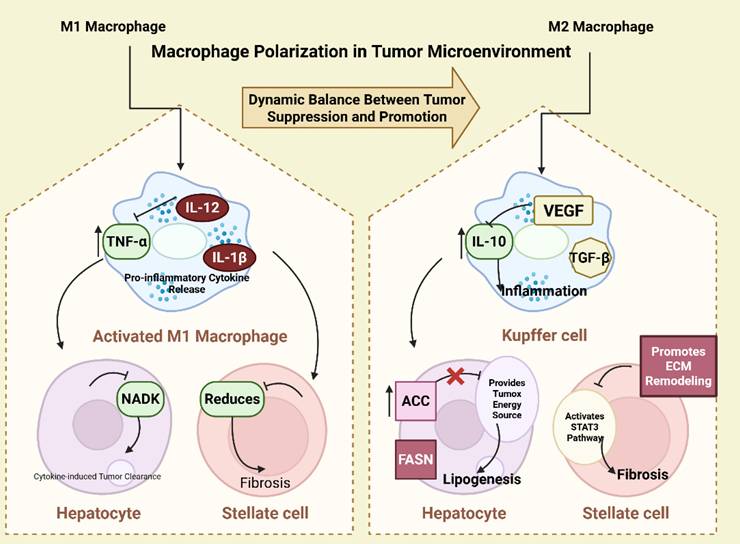
In the TME (TME), tumor-associated macrophages (TAMs) are predominantly polarized to the M2 phenotype, which promotes angiogenesis, immune suppression, and tumor invasion. Reprogramming TAMs from the M2 to M1 phenotype is a promising therapeutic strategy to restore anti-tumor immunity.
4.2. Key Signaling Pathways Driving M1 and M2 Polarization
4.2.1. STAT3 Pathway
TCM Mechanism: Tripterygium wilfordii (Thunder God Vine) contains triptolide, which inhibits STAT3 activation, reducing IL-10 and Arg-1 expression in TAMs (Figure 4). The Signal Transducer and Activator of Transcription 3 (STAT3) pathway plays a pivotal role in driving M2-like polarization of tumor-associated macrophages (TAMs) within the TME. Persistent STAT3 activation, commonly induced by cytokines such as IL-6 and IL-10, promotes the transcription of immunosuppressive genes, including IL-10, VEGF, and arginase-1 (Arg-1), thereby reinforcing immune suppression and tumor progression [93].
Synergistic mechanisms of multi-component traditional Chinese Medicine in remodeling the TME: triple-targeting angiogenesis, TAMs polarization, and immune ecosystem.
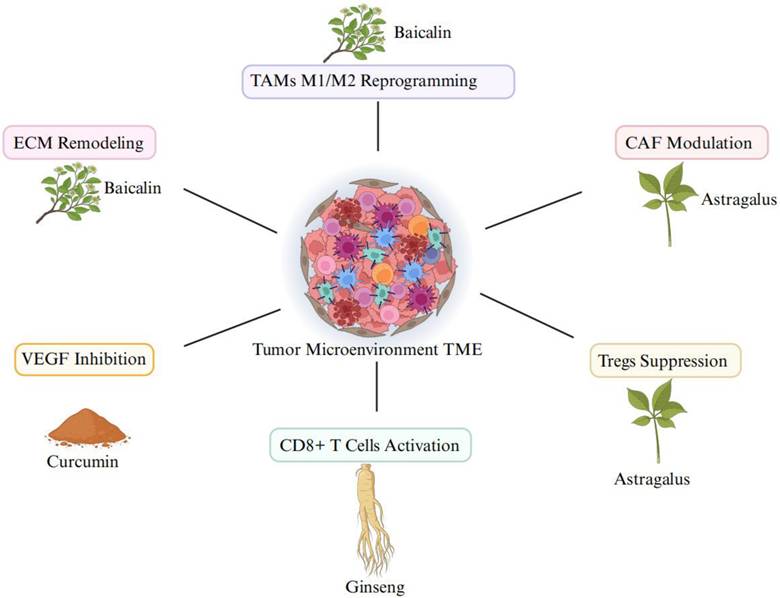
Importantly, STAT3 signaling is closely linked to macrophage metabolic adaptation, favoring anti-inflammatory and tumor-supportive programs that stabilize the M2 phenotype [93]. Inhibition of STAT3 has therefore been recognized as a critical strategy for reprogramming TAMs toward an M1-like, antitumor state.
Representative TCM components, such as triptolide derived from Tripterygium wilfordii, have been reported to suppress STAT3 activation, leading to reduced IL-10 and Arg-1 expression and attenuation of M2-associated immunosuppressive features [94]. Collectively, these findings indicate that TCM-mediated modulation of STAT3 functions as a pathway-centered mechanism to reshape macrophage immunometabolic states, rather than isolated compound-specific effects.
4.2.2. NF-κB Pathway
The nuclear factor kappa B (NF-κB) pathway is a central regulator of inflammation and macrophage polarization within the TME. Activation of the canonical NF-κB signaling cascade by pro-inflammatory stimuli such as lipopolysaccharide (LPS) and interferon-γ promotes M1-like macrophage polarization and induces the production of antitumor cytokines, including TNF-α, IL-12, and IL-1β [95].
However, under chronic inflammatory or tumor-derived signaling conditions, NF-κB activation may also contribute to immune tolerance by driving IL-10 expression and cooperating with transcriptional co-regulators that favor M2-like programs [96]. Notably, NF-κB signaling is tightly linked to macrophage metabolic states, as sustained activation can support glycolysis-dependent inflammatory responses or, alternatively, immunosuppressive adaptation depending on microenvironmental cues.
Representative TCM-derived compounds, such as curcumin from Curcuma longa, have been shown to modulate NF-κB signaling by inhibiting IκB kinase activity and preventing NF-κB nuclear translocation, thereby attenuating M2-associated inflammatory and metabolic features while promoting antitumor immune responses [97]. Collectively, these findings highlight NF-κB as a signaling hub through which TCM exerts pathway-centered regulation of macrophage immunometabolic plasticity rather than compound-specific effects.
4.2.3. PI3K/AKT Pathway
The phosphatidylinositol-3-kinase/protein kinase B (PI3K/AKT) pathway is a key signaling axis governing macrophage survival, metabolism, and polarization. Activation of PI3K/AKT signaling by cytokines such as IL-4 and IL-10 promotes M2-like polarization through upregulation of immunosuppressive markers, including arginase-1 and CD206, and enhances the production of TGF-β [98,99].
From a metabolic perspective, PI3K/AKT signaling supports mitochondrial fitness and lipid metabolic reprogramming in macrophages, thereby stabilizing tumor-supportive M2 phenotypes within nutrient-rich or hypoxic tumor niches. In contrast, inhibition of PI3K/AKT signaling has been associated with a shift toward M1-like polarization and enhanced pro-inflammatory cytokine production [100].
Representative TCM interventions, such as polysaccharides derived from Astragalus membranaceus, have been reported to suppress AKT phosphorylation, resulting in reduced M2 polarization and increased expression of TNF-α and IL-12 in macrophages [101,102]. These observations suggest that TCM-mediated modulation of PI3K/AKT signaling functions at the pathway level to reprogram macrophage immunometabolic states, rather than acting through isolated downstream targets.
4.2.4. TGF-β/SMAD Pathway
The transforming growth factor-β (TGF-β)/SMAD signaling pathway is a critical driver of M2-like macrophage polarization and immunosuppressive remodeling within the TME. Elevated TGF-β levels in the TME activate SMAD2 and SMAD3, which translocate to the nucleus and induce the transcription of genes associated with immune suppression, extracellular matrix remodeling, and tumor progression [68,103]. Through these effects, TGF-β/SMAD signaling not only stabilizes M2 macrophage phenotypes but also contributes to the formation of a physical and biochemical barrier that limits effective antitumor immune responses.
Representative TCM-derived compounds, such as baicalin from Scutellaria baicalensis, have been shown to inhibit TGF-β signaling by reducing SMAD3 phosphorylation. This modulation attenuates M2-associated immunosuppressive features and enhances T cell-mediated antitumor activity [104]. Collectively, these findings suggest that TCM-mediated interference with the TGF-β/SMAD axis functions as a pathway-centered mechanism to reprogram macrophage phenotypes and reshape the immunosuppressive TME.
4.2.5. Hypoxia-Inducible Factor-1 Alpha (HIF-1α)
Hypoxia is a defining feature of the TME, and hypoxia-inducible factor-1 alpha (HIF-1α) serves as a master regulator of cellular adaptation to low oxygen conditions. In macrophages, HIF-1α activation promotes M2-like polarization by upregulating pro-tumorigenic factors such as vascular endothelial growth factor (VEGF) and arginase-1 (Arg-1). In parallel, HIF-1α drives metabolic reprogramming toward glycolysis, reinforcing the functional and metabolic stability of immunosuppressive macrophage phenotypes under hypoxic stress [105].
From a therapeutic perspective, representative TCM interventions such as Salvia miltiorrhiza (Danshen) and its active compound tanshinone IIA have been reported to inhibit HIF-1α stabilization under hypoxic conditions. This suppression leads to reduced VEGF expression and promotes the reprogramming of tumor-associated macrophages toward a more M1-like, antitumor phenotype [106]. These observations highlight HIF-1α as a key immunometabolic checkpoint through which TCM exerts pathway-level regulation of macrophage plasticity within hypoxic tumor niches. To facilitate comparison of representative traditional Chinese medicine compounds and their immunometabolic regulatory roles in macrophage polarization, a summary table is provided (Table 1).
Immunometabolic regulatory roles of representative TCM compounds in tumor-associated macrophages.
| TCM compound / herb | Primary signaling pathway | Metabolic regulation | Immune modulation (TAMs) | Key functional outcome |
|---|---|---|---|---|
| Triptolide (Tripterygium wilfordii) | STAT3 | Suppression of M2-associated metabolic programs | ↓ IL-10, ↓ Arg-1, reduced M2 polarization | Restoration of antitumor immune activity |
| Curcumin (Curcuma longa) | NF-κB | Modulation of inflammatory metabolism | Shift from M2 to M1 phenotype | Enhanced pro-inflammatory immune response |
| Astragalus polysaccharides (Astragalus membranaceus) | PI3K/AKT | Regulation of lipid and mitochondrial metabolism | ↓ M2 markers, ↑ TNF-α / IL-12 | Promotion of M1-like macrophage polarization |
| Baicalin (Scutellaria baicalensis) | TGF-β/SMAD | Attenuation of immunosuppressive signaling | Inhibition of M2 polarization | Enhanced T cell-mediated antitumor immunity |
| Tanshinone IIA (Salvia miltiorrhiza) | HIF-1α | Reduction of glycolysis under hypoxia | Reprogramming of TAMs toward M1 | Alleviation of hypoxia-driven immune suppression |
5. Traditional Chinese Medicine in Metabolic Reprogramming
Cancer cells exhibit characteristic metabolic reprogramming to support their uncontrolled growth and survival. A key feature of this reprogramming is the Warburg effect, where cancer cells shift from oxidative phosphorylation (OXPHOS) to glycolysis, even under normal oxygen conditions. This shift allows for faster ATP production and provides metabolic intermediates necessary for the biosynthesis of nucleotides, lipids, and proteins, which are essential for cell division and survival. In addition to glycolysis, tumor cells also exhibit altered glutamine metabolism, lipid metabolism, and mitochondrial dysfunction, all of which contribute to the aggressive nature of tumors [27,64,107].
Traditional Chinese Medicine (TCM) compounds have shown promise in restoring metabolic balance by targeting these altered metabolic pathways. For example, Berberine, a bioactive alkaloid from Coptis chinensis, has been extensively studied for its anti-cancer effects. Berberine inhibits glycolysis by downregulating Hexokinase II (HKII), a key enzyme in the glycolytic pathway, and activating the AMP-activated protein kinase (AMPK) signaling pathway. AMPK activation leads to the suppression of mTOR, a major regulator of cell growth and metabolism, thereby reprogramming cancer cells away from glycolysis and promoting mitochondrial oxidative phosphorylation [26,42,85]. Berberine also inhibits fatty acid synthase (FAS), which is crucial for lipid biosynthesis in cancer cells, and thereby reduces lipid accumulation, which is essential for tumor cell proliferation [108].
Many preclinical studies have confirmed the efficacy of curcumin, the active compound in Curcuma longa, in targeting cancer metabolism. Curcumin has been shown to suppress glycolysis by inhibiting Hexokinase II (HKII) and Pyruvate Kinase M2 (PKM2), both of which are overexpressed in cancer cells [109]. Additionally, curcumin has been found to modulate mitochondrial dynamics, promoting mitochondrial biogenesis while inhibiting mitochondrial fission. This helps restore the normal energy balance in tumor cells, reducing their proliferative capacity. Some studies have shown that curcumin is able to affect mitochondrial function, thereby reducing the proliferation and invasion of cancer cells [28,29,110,111].
Moreover, epigallocatechin gallate (EGCG), a polyphenol derived from Camellia sinensis (green tea), has been shown to modulate lipid metabolism in cancer cells by inhibiting acetyl-coA carboxylase (ACC), a key enzyme in fatty acid synthesis. EGCG suppresses the mTORC1 pathway, which not only inhibits protein synthesis but also promotes autophagy, further impairing the metabolic capacity of tumor cells [28]. These findings underscore the potential of TCM compounds in reprogramming cancer metabolism and restoring normal cellular energy processes.
6. Traditional Chinese Medicine in the TME
The TME (TME) plays a pivotal role in cancer progression by influencing the behavior of tumor cells and immune cells. One of the key features of the TME is the presence of tumor-associated macrophages (TAMs), which are crucial for promoting tumor growth, metastasis, and immune evasion. TAMs can be polarized into two distinct phenotypes: M1 macrophages, which exhibit anti-tumor activity by secreting pro-inflammatory cytokines such as TNF-α, IL-12, and IFN-γ, and M2 macrophages, which promote tumor progression and immune suppression through the secretion of immunosuppressive cytokines such as IL-10 and TGF-β [31,76]. The balance between M1 and M2 macrophages in the TME is crucial in determining the overall immune landscape of the tumor.
The TME has shown considerable promise in modulating the polarization of macrophages within the TME. Baicalin, a flavonoid derived from Scutellaria baicalensis, has been demonstrated to promote M1 macrohage polarization while inhibiting the M2 phenotype. This shift in macrophage polarization leads to enhanced anti-tumor immunity and better tumor clearance. Baicalin exerts these effects by reducing the production of IL-10 and TGF-β, which are key cytokines involved in M2 polarization [30,88].
Similarly, Ginsenosides an active compound found in Panax ginseng, known for its immunomodulatory effects, enhances M1 macrophage polarization by upregulating TNF-α and IL-12 expression. Ginseng also modulates the NF-κB pathway, a critical signaling pathway in the immune response, to activate anti-tumor immune responses [33,88]. Studies in mouse models have shown that Ginseng supplementation results in an increased infiltration of M1 macrophages in the TME and a concomitant reduction in tumor growth [112].
Curcumin has also been shown to modulate the TME by altering macrophage polarization. By regulating NF-κB, STAT3, and PI3K/Akt signaling pathways, curcumin promotes M1 polarization and suppresses M2 macrophage activity. This leads to increased expression of pro-inflammatory cytokines and a stronger anti-tumor immune response [113]. Additionally, Astragalus membranaceus has been demonstrated to modulate the TME by improving the function of immune cells, including macrophages, by upregulating IL-12 and promoting T cell activation [34,36,95].
7. Traditional Chinese Medicine in Macrophage Regulation
Macrophages are central players in the immune system, with their ability to either promote or suppress tumor progression depending on their polarization. The balance between M1 (anti-tumor) and M2 (pro-tumor) macrophages is crucial for shaping the TME and modulating immune responses. M1 macrophages are pro-inflammatory and have anti-tumor activity, whereas M2 macrophages promote tumor progression, immune suppression, and tissue remodeling.
Studies have shown that TCM compounds have shown efficacy in modulating macrophage polarization, particularly in shifting macrophages from the M2 to the M1 phenotype. For instance, Astragalus membranaceus, a well-known TCM herb, has been demonstrated to promote M1 polarization by activating the PI3K/Akt pathway, which enhances pro-inflammatory cytokine production and suppresses M2 macrophage function [18,35,58]. Similarly, Tripterygium wilfordii has been shown to inhibit the STAT3 pathway, a key regulator of M2 macrophage polarization, thereby reducing the immunosuppressive environment within the TME and enhancing the anti-tumor immune response [68].
Polygonum cuspidatum has been reported to inhibit M2 polarization and promote M1 polarization by modulating NF-κB signaling [114]. This regulation of macrophage polarization leads to enhanced cytotoxic T cell activity and improved anti-tumor immunity.
Additionally, Curcumin has been shown to reduce M2 macrophage infiltration in the TME and promote the conversion of M2 macrophages to the M1 phenotype by modulating various signaling pathways, including NF-κB and STAT3. By promoting M1 macrophage polarization, TCM compounds can not only enhance anti-tumor immunity but also prevent immune evasion mechanisms commonly employed by tumors [109].
8. Preclinical Studies of Traditional Chinese Medicine
Preclinical studies have consistently demonstrated the potential of TCM in modulating critical pathways involved in cancer progression, particularly through metabolic reprogramming, immune regulation, and microenvironmental remodeling. A growing body of evidence indicates that TCM compounds can serve as primary agents or adjuncts to enhance the efficacy of existing cancer therapies [115].
For instance, Huangqi (Astragalus membranaceus), widely used for its immunomodulatory properties, has been shown to promote macrophage polarization towards the M1 phenotype in breast cancer (4T1) mouse models. This shift not only enhanced cytotoxic T cell activity but also reduced tumor growth and metastasis [35]. Similarly, Triptolide, a diterpenoid derived from Tripterygium wilfordii, has demonstrated potent anti-cancer activity in prostate cancer models by targeting the STAT3 signaling pathway, thereby inhibiting M2 macrophage polarization and reducing the immunosuppressive environment within the TME [14,116].
In addition to immune modulation, compounds such as Baicalin have been reported to exhibit anti-metastatic properties. In preclinical models of liver cancer (HepG2), Baicalin reduced metastatic nodules in the lungs and liver by modulating the PI3K/Akt signaling pathway, which is critical for regulating macrophage polarization and tumor cell migration [30]. Moreover, the potential of TCM extends beyond individual herbs. Formulae such as Shenqi Fuzheng, a combination of Astragalus and Codonopsis, have been shown to synergistically inhibit tumor growth by enhancing immune cell infiltration into the TME [117].
9. Mechanistic Insights of Traditional Chinese Medicine
Targeting Metabolic Pathways: TCM compounds such as berberine, curcumin, and epigallocatechin gallate (EGCG) disrupt cancer cell metabolism by modulating pathways like AMPK/mTOR and glycolysis. Berberine, for instance, activates AMPK while downregulating glycolysis-related enzymes, leading to reduced lactate production and ATP generation [26]. Curcumin similarly inhibits PKM2 and mitochondrial fission, restoring oxidative metabolism in cancer cells [29].
Immune Regulation: Herbs like Astragalus and Ginseng enhance immune surveillance by promoting T cell infiltration and modulating the balance of macrophage polarization towards the M1 phenotype. This reprogramming reduces the immunosuppressive signals in the TME, such as IL-10 and TGF-β, while enhancing pro-inflammatory cytokines like TNF-α and IL-12 [33,36].
Anti-Angiogenic Effects: TCM compounds like Salvianolic acid B, derived from Salvia miltiorrhiza, inhibit angiogenesis by suppressing VEGF expression, thereby depriving tumors of essential nutrients and oxygen. This dual effect of immune activation and vascular suppression significantly reduces tumor growth [118].
10. Synergistic Effects of TCM with Conventional Therapies
10.1. Enhancing Chemotherapy Efficacy
The combination of TCM and chemotherapy has been extensively studied for its ability to reduce drug resistance and improve therapeutic outcomes (Figure 5). For example, berberine combined with cisplatin in ovarian cancer models demonstrated enhanced cytotoxicity against tumor cells by sensitizing them to DNA damage, while also mitigating cisplatin-induced nephrotoxicity. Mechanistically, berberine enhances cisplatin efficacy by inhibiting glycolytic pathways and activating oxidative stress responses, which synergistically promote apoptosis in cancer cells [100].
The tripartite synergy bridge of TCM-chemotherapy-immunotherapy: augmenting drug sensitivity, alleviating immune suppression, and enhancing T-cell infiltration.
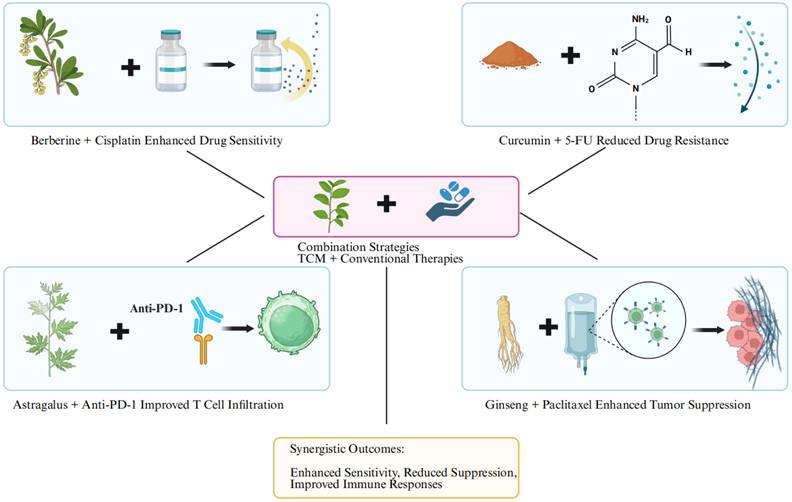
Similarly, curcumin potentiates the efficacy of 5-fluorouracil (5-FU) in colorectal cancer models by downregulating NF-κB and multidrug resistance proteins such as P-glycoprotein. This not only reduces drug resistance but also promotes cancer cell apoptosis and limits proliferation [34]. Preclinical studies have also highlighted the potential of ginsenoside Rg3, a major active compound in Ginseng, which enhances the efficacy of paclitaxel in breast cancer by modulating the PI3K/Akt/mTOR pathway, thereby reducing tumor growth and preventing metastasis [42].
10.2. Improving Immunotherapy Outcomes
Immune checkpoint inhibitors (ICIs), such as anti-PD-1 or anti-CTLA-4 therapies, have revolutionized cancer treatment but remain ineffective in a significant subset of patients due to an immunosuppressive TME. TCM has shown promise in overcoming this challenge by modulating immune responses.
For instance, Astragalus membranaceus enhances the efficacy of anti-PD-1 therapy by increasing T-cell infiltration and reducing immunosuppressive cytokines like TGF-β. In a lung cancer model, combining Astragalus with anti-PD-1 therapy significantly improved tumor clearance and survival rates compared to either treatment alone [36]. Similarly, curcumin has been reported to remodel the immune landscape by inhibiting M2 macrophages and enhancing cytotoxic T-cell activity, thereby improving ICI responsiveness in melanoma models [100].
11. Discussion
11.1. Challenges in TCM Research
Despite the promising preclinical and clinical data, several challenges hinder the widespread adoption of TCM in mainstream cancer therapy:
Standardization and Quality Control: The variability in TCM formulations due to differences in plant cultivation, harvesting, and preparation methods results in inconsistent therapeutic effects. Establishing standardized extraction and quality control protocols is crucial for ensuring reproducibility.
Mechanistic Complexity: TCM often exerts effects through multiple targets and pathways, making it challenging to delineate precise mechanisms of action. This complexity necessitates the use of omics technologies (e.g., transcriptomics, proteomics, and metabolomics) to comprehensively evaluate its biological effects.
Regulatory Barriers: Many TCM compounds lack rigorous clinical validation, limiting their acceptance by regulatory bodies. Conducting large-scale, randomized controlled trials (RCTs) with standardized protocols is essential to overcome these barriers. Although limitations such as formulation standardization and variability in clinical evidence have been acknowledged, future efforts should further address these challenges through well-designed multicenter, large-sample clinical trials. Such studies are essential to enhance reproducibility, reduce center-specific bias, and provide robust evidence for the clinical efficacy and safety of traditional Chinese medicine (TCM) in oncology. In addition, future research should focus on the rational integration of TCM into existing cancer treatment paradigms. Rather than serving as a stand-alone intervention, TCM may function as an adjuvant modality that complements immunotherapy and targeted therapy by modulating the TME, alleviating treatment-related immunosuppression, and improving therapeutic responsiveness. Exploring combined treatment strategies in different tumor contexts may therefore facilitate the clinical translation of TCM and optimize its role in personalized cancer management.
11.2. Future Directions in TCM Research
Integration of Advanced Technologies: The use of molecular docking, high-throughput screening, and AI-based prediction models can accelerate the discovery of bioactive compounds in TCM and their molecular targets. For example, AI can be used to predict synergistic combinations of TCM and conventional therapies.
Development of Combination Therapies: Synergizing TCM with modern cancer therapies such as immunotherapy and targeted therapies, and exploring the potential of combining TCM with emerging therapies such as CAR-T cells, oncolytic viruses, or nano-drug delivery systems may open new avenues for cancer treatment.
Personalized Medicine: Leveraging precision medicine approaches to tailor TCM treatments based on individual genetic and metabolic profiles could enhance efficacy and minimize side effects.
Global Collaboration: Encouraging international collaboration in TCM research, particularly with institutions specializing in cancer biology and pharmacology, can facilitate cross-disciplinary studies and clinical applications.
Clinical translation: Validation of the effects of TCM on metabolic pathways in human trials, e.g., designing phased clinical trials to objectively assess the effects of TCM interventions on metabolic reprogramming.
Bioavailability issues: The low bioavailability of many TCM compounds is addressed using nanotechnology or improved delivery systems.
Despite encouraging preclinical evidence, the clinical translation of TCM in oncology remains constrained by several challenges. These include heterogeneity in patient populations, variability in TMEal states, and the lack of standardized clinical endpoints that directly reflect immunometabolic modulation. To overcome these limitations, future studies should prioritize biomarker-guided clinical trial designs, integrate immune and metabolic profiling for patient stratification, and adopt multicenter, adequately powered trial frameworks. Importantly, TCM is more likely to exert clinical benefit as an adjuvant rather than a stand-alone therapy. Given the immunometabolic heterogeneity across tumor types, the integration of TCM with conventional treatments should therefore be context-dependent. Tumors characterized by hypoxia, lactate accumulation, and M2-dominant macrophage infiltration may benefit from TCM interventions targeting metabolic normalization and macrophage reprogramming to enhance responsiveness to immunotherapy. In contrast, tumors with pre-existing immune infiltration but pronounced therapy-induced immunosuppression may derive greater benefit from TCM-based adjuncts during chemotherapy. Together, these considerations highlight the potential of TCM as a flexible adjuvant modality that complements established anticancer therapies by reshaping the TME rather than replacing standard treatments.
Acknowledgements
Funding
This research was supported by grants from the Traditional Chinese Medicine Science and Technology Program (Special Project for Modernization of Traditional Chinese Medicine) of Zhejiang Province (2022ZX014): Clinical research on syndrome differentiation and treatment of chronic renal failure by traditional Chinese medicine based on real-world study and RCT (2022.01-2025.12); and the Key Research and Development Program (Pioneer and Leading Goose-X R&D Program) of Zhejiang Provincial Science and Technology Plan (2024C03212): Evidence-based research and mechanism exploration on the optimization of prevention and treatment protocols for chronic kidney disease with traditional Chinese medicine based on an intensive diagnosis and treatment model (2024.01-2026.12).
Author Contributions
YAA: Writing - original draft, review and editing, Conceptualization, Methodology, Project administration; CHY: Writing - review and editing, Conceptualization, Methodology; YDL: Writing - original draft, Conceptualization, Methodology.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Bray F, Laversanne M, Sung H. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-63
2. Yuan S, Sun R, Shi H. et al. VDAC2 loss elicits tumour destruction and inflammation for cancer therapy. Nature. 2025;640:1062-71
3. Zhu Y, Zhou Z, Du X. et al. Cancer cell-derived arginine fuels polyamine biosynthesis in tumor-associated macrophages to promote immune evasion. Cancer Cell. 2025;43:1045-60.e7
4. Hanahan D, Weinberg RA. Hallmarks of cancer: The next generation. Cell. 2011;144:646-74
5. Sanyal R, Polyak MJ, Zuccolo J. et al. MS4A4A: A novel cell surface marker for M2 macrophages and plasma cells. Immunol Cell Biol. 2017;95:611-9
6. Mao X, Xu J, Wang W. et al. Crosstalk between cancer-associated fibroblasts and immune cells in the tumor microenvironment: new findings and future perspectives. Mol Cancer. 2021;20:131
7. Liu H, Wang ZY, Zhou YC. et al. Immunomodulation of Chinese herbal medicines on NK cell populations for cancer therapy: A systematic review. J Ethnopharmacol. 2021;268:113561
8. Chapman NM, Chi H. Metabolic rewiring and communication in cancer immunity. Cell Chem Biol. 2024;31:862-83
9. Dai Z, Tan C, Wang J. et al. Traditional Chinese medicine for gastric cancer: An evidence mapping. Phytother Res. 2024;38:2707-23
10. Sun K, Wu L, Wang S. et al. Antitumor effects of Chinese herbal medicine compounds and their nano-formulations on regulating the immune system microenvironment. Front Oncol. 2022;12:949332
11. Wang S, Fu JL, Hao HF. et al. Metabolic reprogramming by traditional Chinese medicine and its role in effective cancer therapy. Pharmacol Res. 2021;170:105728
12. Zuo Q, Wu Y, Hu Y. et al. Targeting lipid reprogramming in the tumor microenvironment by traditional Chinese medicines as a potential cancer treatment. Heliyon. 2024;10:e30807
13. Atri C, Guerfali FZ, Laouini D. Role of human macrophage polarization in inflammation during infectious diseases. Int J Mol Sci. 2018;19:1801
14. Guerra AR, Duarte MF, Duarte IF. Targeting tumor metabolism with plant-derived natural products: emerging trends in cancer therapy. J Agric Food Chem. 2018;66:10663-85
15. Gao J, Liang Y, Wang L. Shaping polarization of tumor-associated macrophages in cancer immunotherapy. Front Immunol. 2022;13:888713
16. Zhang J, Li N, Hu X. Metabolic reprograming of macrophages: A new direction in traditional chinese medicine for treating liver failure. J Immunol Res. 2024;2024:5891381
17. Wang L, Wu W, Zhu X. et al. The ancient Chinese decoction Yu-Ping-Feng suppresses orthotopic lewis lung cancer tumor growth through increasing M1 macrophage polarization and CD4+ T cell cytotoxicity. Front Pharmacol. 2019;10:1333
18. Boutilier AJ, Elsawa SF. Macrophage polarization states in the tumor microenvironment. Int J Mol Sci. 2021;22:6995
19. Zhang J, Gao J, Cui J. et al. Tumor-associated macrophages in tumor progression and the role of traditional Chinese medicine in regulating TAMs to enhance antitumor effects. Front Immunol. 2022;13:1026898
20. Zhang Y, Zhang Z. The history and advances in cancer immunotherapy: Understanding the characteristics of tumor-infiltrating immune cells and their therapeutic implications. Cell Mol Immunol. 2020;17:807-21
21. Shi JH, Liu LN, Song DD. et al. TRAF3/STAT6 axis regulates macrophage polarization and tumor progression. Cell Death Differ. 2023;30:2005-16
22. Zhao Q, Zhou J, Li F. et al. The role and therapeutic perspectives of Sirtuin 3 in cancer metabolism reprogramming, metastasis, and chemoresistance. Front Oncol. 2022;12:910963
23. Kong MY, Li LY, Lou YM. et al. Chinese herbal medicines for prevention and treatment of colorectal cancer: From molecular mechanisms to potential clinical applications. J Integr Med. 2020;18:369-84
24. Bao X, Wang D, Dai X. et al. An immunometabolism subtyping system identifies S100A9+ macrophage as an immune therapeutic target in colorectal cancer based on multiomics analysis. Cell Rep Med. 2023;4:100987
25. D'Urso A, Oltolina F, Borsotti C. et al. Macrophage reprogramming via the modulation of unfolded protein response with siRNA-loaded magnetic nanoparticles in a TAM-like experimental model. Pharmaceutics. 2023;15:1711
26. Vander Heiden MG, DeBerardinis RJ. Understanding the Intersections between Metabolism and Cancer Biology. Cell. 2017;168:657-69
27. Shi Z, Hu C, Zheng X. et al. Feedback loop between hypoxia and energy metabolic reprogramming aggravates the radioresistance of cancer cells. Exp Hematol Oncol. 2024;13:55
28. DeBerardinis RJ, Chandel NS. Fundamentals of cancer metabolism. Sci Adv. 2016;2:e1600200
29. Pavlova NN, Thompson CB. The emerging hallmarks of cancer metabolism. Cell Metab. 2016;23:27-47
30. Maj T, Wang W, Crespo J. et al. Oxidative stress controls regulatory T cell apoptosis and suppressor activity and PD-L1-blockade resistance in tumor. Nat Immunol. 2017;18:1332-41
31. Trejo-Solis C, Silva-Adaya D, Serrano-García N. et al. Role of glycolytic and glutamine metabolism reprogramming on the proliferation, invasion, and apoptosis resistance through modulation of signaling pathways in glioblastoma. Int J Mol Sci. 2023;24:17633
32. Li Y, Pan Y, Zhao X. et al. Peroxisome proliferator-activated receptors: A key link between lipid metabolism and cancer progression. Clin Nutr. 2024;43:332-45
33. Liberti MV, Locasale JW. The warburg effect: How does it benefit cancer cells? Trends Biochem Sci. 2016;41:211-8
34. Praharaj M, Shen F, Lee AJ. et al. Metabolic reprogramming of tumor-associated macrophages using glutamine antagonist JHU083 drives tumor immunity in myeloid-rich prostate and bladder cancers. Cancer Immunol Res. 2024;12:854-75
35. Liu VM, Vander Heiden MG. The role of pyruvate kinase M2 in cancer metabolism. Brain Pathol. 2015;25:781-3
36. Mambetsariev N, Lin WW, Wallis AM. et al. TRAF3 deficiency promotes metabolic reprogramming in B cells. Sci Rep. 2016;6:35349
37. Cui Y, Li C, Sang F. et al. Natural products targeting glycolytic signaling pathways-an updated review on anti-cancer therapy. Front Pharmacol. 2022;13:1035882
38. Zhao M, Wei F, Sun G. et al. Natural compounds targeting glycolysis as promising therapeutics for gastric cancer: A review. Front Pharmacol. 2022;13:1004383
39. Fu M, Liu Y, Cheng H. et al. Coptis chinensis and dried ginger herb combination inhibits gastric tumor growth by interfering with glucose metabolism via LDHA and SLC2A1. J Ethnopharmacol. 2022;284:114771
40. Mirzaei H, Masoudifar A, Sahebkar A. et al. MicroRNA: A novel target of curcumin in cancer therapy. J Cell Physiol. 2018;233:3004-15
41. Zhao Y, Chard Dunmall LS, Cheng Z. et al. Natural products targeting glycolysis in cancer. Front Pharmacol. 2022;13:1036502
42. Peng B, Zhang SY, Chan KI. et al. Novel anti-cancer products targeting AMPK: Natural herbal medicine against breast cancer. Molecules. 2023;28:740
43. Zhan L, Su F, Li Q. et al. Phytochemicals targeting glycolysis in colorectal cancer therapy: effects and mechanisms of action. Front Pharmacol. 2023;14:1257450
44. Xi Z, Dai R, Ze Y. et al. Traditional Chinese medicine in lung cancer treatment. Mol Cancer. 2025;24:57
45. Khan A, Siddiqui S, Husain SA. et al. Phytocompounds targeting metabolic reprogramming in cancer: An assessment of role, mechanisms, pathways, and therapeutic relevance. J Agric Food Chem. 2021
46. Pralea IE, Petrache AM, Tigu AB. et al. Phytochemicals as regulators of tumor glycolysis and hypoxia signaling pathways: Evidence from in vitro studies. Pharmaceuticals (Basel). 2022;15:808
47. Xing P, Zhong Y, Cui X. et al. Natural products in digestive tract tumors metabolism: Functional and application prospects. Pharmacol Res. 2023;191:106766
48. Wang F, Ma S, Chen P. et al. Imaging the metabolic reprograming of fatty acid synthesis pathway enables new diagnostic and therapeutic opportunity for breast cancer. Cancer Cell Int. 2023;23:83
49. Gu Y, Xue M, Wang Q. et al. Novel strategy of proxalutamide for the treatment of prostate cancer through coordinated blockade of lipogenesis and androgen receptor axis. Int J Mol Sci. 2021;22:13222
50. Liu HY, Zhang HS, Liu MY. et al. GLS1 depletion inhibited colorectal cancer proliferation and migration via redox/Nrf2/autophagy-dependent pathway. Arch Biochem Biophys. 2021;708:108964
51. Lin Z, Li J, Zhang J. et al. Metabolic reprogramming driven by IGF2BP3 promotes acquired resistance to EGFR inhibitors in non-small cell lung cancer. Cancer Res. 2023;83:2187-207
52. Wang R, Wang C, Lu L. et al. Baicalin and baicalein in modulating tumor microenvironment for cancer treatment: A comprehensive review with future perspectives. Pharmacol Res. 2024;199:107032
53. Ortiz LM, Lombardi P, Tillhon M. et al. Berberine, an epiphany against cancer. Molecules. 2014;19:12349-67
54. Cui Y, Liu J, Wang X. et al. Baicalin attenuates the immune escape of oral squamous cell carcinoma by reducing lactate accumulation in tumor microenvironment. J Adv Res. 2025;77:721-32
55. Guo W, Ding Y, Pu C. et al. Curcumin inhibits pancreatic cancer cell proliferation by regulating Beclin1 expression and inhibiting the hypoxia-inducible factor-1α-mediated glycolytic pathway. J Gastrointest Oncol. 2022;13:3254-62
56. Liu J, Zhang C, Liu S. et al. Tanshinone IIA promotes apoptosis by downregulating BCL2 and upregulating TP53 in triple-negative breast cancer. Naunyn Schmiedebergs Arch Pharmacol. 2023;396:365-74
57. Fu X, He Y, Li M. et al. Targeting of the tumor microenvironment by curcumin. Biofactors. 2021;47:914-32
58. Humbert M, Seiler K, Mosimann S. et al. Reducing FASN expression sensitizes acute myeloid leukemia cells to differentiation therapy. Cell Death Differ. 2021;28:2465-81
59. Tsukamoto S, Huang Y, Kumazoe M. et al. Sphingosine kinase-1 protects multiple myeloma from apoptosis driven by cancer-specific inhibition of RTKs. Mol Cancer Ther. 2015;14:2303-12
60. Zhou L, Liu Z, Wang Z. et al. Astragalus polysaccharides exerts immunomodulatory effects via TLR4-mediated MyD88-dependent signaling pathway in vitro and in vivo. Sci Rep. 2017;7:44822
61. Neophytou CM, Panagi M, Stylianopoulos T. et al. The role of tumor microenvironment in cancer metastasis: Molecular mechanisms and therapeutic opportunities. Cancers (Basel). 2021;13:2053
62. Zhao Y, Shen M, Wu L. et al. Stromal cells in the tumor microenvironment: accomplices of tumor progression? Cell Death Dis. 2023;14:587
63. Ni Y, Zhou X, Yang J. et al. The role of tumor-stroma interactions in drug resistance within tumor microenvironment. Front Cell Dev Biol. 2021;9:637675
64. Hu H, Chen Y, Tan S. et al. The research progress of antiangiogenic therapy, immune therapy and tumor microenvironment. 2022; 13: 802846.
65. Kim SK, Cho SW. The evasion mechanisms of cancer immunity and drug intervention in the tumor microenvironment. Front Pharmacol. 2022;13:868695
66. Tucci M, Passarelli A, Mannavola F. et al. Immune system evasion as hallmark of melanoma progression: The role of dendritic cells. Front Oncol. 2019;9:1148
67. Lei MML, Lee TKW. Cancer stem cells: Emerging key players in immune evasion of cancers. Front Cell Dev Biol. 2021;9:692940
68. Chung JY, Chan MK, Li JS. et al. TGF-β Signaling: From Tissue Fibrosis to Tumor Microenvironment. Int J Mol Sci. 2021;22:7575
69. Yang Q, Guo N, Zhou Y. et al. The role of tumor-associated macrophages (TAMs) in tumor progression and relevant advance in targeted therapy. Acta Pharm Sin B. 2020;10:2156-70
70. Han J, Dong L, Wu M. et al. Dynamic polarization of tumor-associated macrophages and their interaction with intratumoral T cells in an inflamed tumor microenvironment: from mechanistic insights to therapeutic opportunities. Front Immunol. 2023;14:1160340
71. Munir MT, Kay MK, Kang MH. et al. Tumor-associated macrophages as multifaceted regulators of breast tumor growth. Int J Mol Sci. 2021;22:6526
72. Sezginer O, Unver N. Dissection of pro-tumoral macrophage subtypes and immunosuppressive cells participating in M2 polarization. Inflamm Res. 2024;73:1411-23
73. Zheng Y, Wang X, Huang M. Metabolic Regulation of CD8+ T Cells: From Mechanism to Therapy. Antioxid Redox Signal. 2022;37:1234-53
74. Viel S, Vivier E, Walzer T. et al. Targeting metabolic dysfunction of CD8 T cells and natural killer cells in cancer. Nat Rev Drug Discov. 2025;24:190-208
75. Lv X, Li J, Zhang C. et al. The role of hypoxia-inducible factors in tumor angiogenesis and cell metabolism. Genes Dis. 2017;4:19-24
76. Zalpoor H, Aziziyan F, Liaghat M. et al. The roles of metabolic profiles and intracellular signaling pathways of tumor microenvironment cells in angiogenesis of solid tumors. Cell Commun Signal. 2022;20:186
77. Feng B, Wu J, Shen B. et al. Cancer-associated fibroblasts and resistance to anticancer therapies: Status, mechanisms, and countermeasures. Cancer Cell Int. 2022;22:166
78. Ahuja S, Sureka N, Zaheer S. Unraveling the intricacies of cancer-associated fibroblasts: A comprehensive review on metabolic reprogramming and tumor microenvironment crosstalk. Apmis. 2024;132:906-27
79. Jena BC, Das CK, Bharadwaj D. et al. Cancer associated fibroblast mediated chemoresistance: A paradigm shift in understanding the mechanism of tumor progression. Biochim Biophys Acta Rev Cancer. 2020;1874:188416
80. Tajaldini M, Poorkhani A, Amiriani T. et al. Strategy of targeting the tumor microenvironment via inhibition of fibroblast/fibrosis remodeling new era to cancer chemo-immunotherapy resistance. Eur J Pharmacol. 2023;957:175991
81. Arora L, Pal D. Remodeling of stromal cells and immune landscape in microenvironment during tumor progression. Front Oncol. 2021;11:596798
82. Jiang Z, Zhang W, Sha G. et al. Galectins are central mediators of immune escape in pancreatic ductal adenocarcinoma. Cancers (Basel). 2022;14:5475
83. Zhao ZT, Wang J, Fang L. et al. Dual-responsive nanoparticles loading bevacizumab and gefitinib for molecular targeted therapy against non-small cell lung cancer. Acta Pharmacol Sin. 2023;44:244-54
84. Fan P, Zhang N, Candi E. et al. Alleviating hypoxia to improve cancer immunotherapy. Oncogene. 2023;42:3591-604
85. Wu P, Gao W, Su M. et al. Adaptive mechanisms of tumor therapy resistance driven by tumor microenvironment. Front Cell Dev Biol. 2021;9:641469
86. Dandawate S, Williams L, Joshee N. et al. Scutellaria extract and wogonin inhibit tumor-mediated induction of T(reg) cells via inhibition of TGF-β1 activity. Cancer Immunol Immunother. 2012;61:701-11
87. Tung YT, Chen HL, Lai CW. et al. Curcumin reduces pulmonary tumorigenesis in vascular endothelial growth factor (VEGF)-overexpressing transgenic mice. Mol Nutr Food Res. 2011;55:1036-43
88. Wen R, Huang X, Long J. et al. Advances in traditional Chinese herbal medicine and their pharmacodynamic mechanisms in cancer immunoregulation: a narrative review. Transl Cancer Res. 2024;13:1166-87
89. Zhou X, Wang X, Sun Q. et al. Natural compounds: A new perspective on targeting polarization and infiltration of tumor-associated macrophages in lung cancer. Biomed Pharmacother. 2022;151:113096
90. Miao K, Liu W, Xu J. et al. Harnessing the power of traditional Chinese medicine monomers and compound prescriptions to boost cancer immunotherapy. Front Immunol. 2023;14:1277243
91. Fiani ML, Barreca V, Sargiacomo M. et al. Exploiting manipulated small extracellular vesicles to subvert immunosuppression at the tumor microenvironment through mannose receptor/CD206 targeting. Int J Mol Sci. 2020;21:6318
92. Goswami KK, Sarkar M, Ghosh S. et al. Neem leaf glycoprotein regulates function of tumor associated M2 macrophages in hypoxic tumor core: Critical role of IL-10/STAT3 signaling. Mol Immunol. 2016;80:1-10
93. Nikolskii AA, Shilovskiy IP, Barvinskaia ED. et al. Role of STAT3 transcription factor in pathogenesis of bronchial asthma. Biochemistry (Mosc). 2021;86:1489-501
94. Zhu H, Tong S, Yan C. et al. Triptolide attenuates LPS-induced activation of RAW 264.7 macrophages by inducing M1-to-M2 repolarization via the mTOR/STAT3 signaling. Immunopharmacol Immunotoxicol. 2022;44:894-901
95. Lawrence T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb Perspect Biol. 2009;1:a001651
96. Zhou L, Wang D, Qiu X. et al. DHZCP modulates microglial M1/M2 polarization via the p38 and TLR4/NF-κB signaling pathways in LPS-stimulated microglial cells. Front Pharmacol. 2020;11:1126
97. Xiang F, Zhang Z, Li Y. et al. Research progress in the treatment of schistosomiasis with traditional Chinese medicine. J Ethnopharmacol. 2024;333:118501
98. Merecz-Sadowska A, Sitarek P, Śliwiński T. et al. Anti-inflammatory activity of extracts and pure compounds derived from plants via modulation of signaling pathways, especially PI3K/AKT in macrophages. Int J Mol Sci. 2020;21:9605
99. Wang L, Lu Q, Gao W. et al. Recent advancement on development of drug-induced macrophage polarization in control of human diseases. Life Sci. 2021;284:119914
100. Zhan Y, Lang L, Wang F. et al. Hypothyroidism promotes microglia M1 polarization by inhibiting BDNF-promoted PI3K-Akt signaling pathway. Neuroendocrinology. 2025;115:34-47
101. Dong-Jian Y, Lin-Han L, Wan-Ting C. et al. [Knowledge of schistosomiasis in traditional Chinese medicine]. Zhongguo Xue Xi Chong Bing Fang Zhi Za Zhi. 2018;31:218-21
102. Chen S, Zeng J, Li R. et al. Traditional Chinese medicine in regulating macrophage polarization in immune response of inflammatory diseases. J Ethnopharmacol. 2024;325:117838
103. Krstic J, Santibanez JF. Transforming growth factor-beta and matrix metalloproteinases: functional interactions in tumor stroma-infiltrating myeloid cells. ScientificWorldJournal. 2014;2014:521754
104. Li J, Liu H, Lin Q. et al. Baicalin suppresses the migration and invasion of breast cancer cells via the TGF-β/lncRNA-MALAT1/miR-200c signaling pathway. Medicine (Baltimore). 2022;101:e29328
105. Semba H, Takeda N, Isagawa T. et al. HIF-1α-PDK1 axis-induced active glycolysis plays an essential role in macrophage migratory capacity. Nat Commun. 2016;7:11635
106. Wang K, Chen Q, Shao Y. et al. Anticancer activities of TCM and their active components against tumor metastasis. Biomed Pharmacother. 2021;133:111044
107. Xie B, Li J, Lou Y. et al. Reprogramming macrophage metabolism following myocardial infarction: A neglected piece of a therapeutic opportunity. Int Immunopharmacol. 2024;142:113019
108. Gu S, Song X, Xie R. et al. Berberine inhibits cancer cells growth by suppressing fatty acid synthesis and biogenesis of extracellular vesicles. Life Sci. 2020;257:118122
109. Shuvalov O, Kirdeeva Y, Daks A. et al. Phytochemicals target multiple metabolic pathways in cancer. Antioxidants (Basel). 2023;12:2012
110. Jung KH, Lee JH, Park JW. et al. Effects of curcumin on cancer cell mitochondrial function and potential monitoring with ¹⁸F-FDG uptake. Oncol Rep. 2016;35:861-8
111. Arora S, Joshi G, Chaturvedi A. et al. A perspective on medicinal chemistry approaches for targeting pyruvate kinase M2. J Med Chem. 2022;65:1171-205
112. Zhu Y, Wang A, Zhang S. et al. Paclitaxel-loaded ginsenoside Rg3 liposomes for drug-resistant cancer therapy by dual targeting of the tumor microenvironment and cancer cells. J Adv Res. 2023;49:159-73
113. Deswal B, Bagchi U, Kapoor S. Curcumin suppresses M2 macrophage-derived paclitaxel chemoresistance through inhibition of PI3K-AKT/STAT3 signaling. Anticancer Agents Med Chem. 2024;24:146-56
114. Sun Q, Nan XY, Wang H. et al. Polydatin retards the progression of osteoarthritis by maintaining bone metabolicbalance and inhibiting macrophage polarization. Front Bioeng Biotechnol. 2024;12:1514483
115. Chen F, Li J, Wang H. et al. Anti-tumor effects of Chinese medicine compounds by regulating immune cells in microenvironment. Front Oncol. 2021;11:746917
116. Lee B, Park Y, Lee Y. et al. Triptolide, a cancer cell proliferation inhibitor, causes zebrafish muscle defects by regulating notch and stat3 signaling pathways. Int J Mol Sci. 2024;25:4675
117. Ma Y, Qi Y, Zhou Z. et al. Shenqi Fuzheng injection modulates tumor fatty acid metabolism to downregulate MDSCs infiltration, enhancing PD-L1 antibody inhibition of intracranial growth in melanoma. Phytomedicine. 2024;122:155171
118. Katary MA, Abdelsayed R, Alhashim A. et al. Salvianolic acid B slows the progression of breast cancer cell growth via enhancement of apoptosis and reduction of oxidative stress, inflammation, and angiogenesis. Int J Mol Sci. 2019;20:5653
Author contact
Corresponding author: Professor Hongyu Chen (Email: 201912211504016edu.cn).

 Global reach, higher impact
Global reach, higher impact