Impact Factor ISSN: 1837-9664
J Cancer 2026; 17(4):847-854. doi:10.7150/jca.127661 This issue Cite
Review
Orientin and Cancer Suppression: Molecular Mechanisms and Synergistic Effects
Department of Biological Sciences, College of Science, King Faisal University, Al-Ahsa, 31982, Saudi Arabia.
Received 2025-11-1; Accepted 2026-1-8; Published 2026-4-8
Abstract
Orientin, a C-glycosyl flavonoid (luteolin-8-C-glucoside) naturally present in plants such as Ocimum sanctum (holy basil), Trollius chinensis, rooibos tea, and Passiflora species, exhibits a wide range of pharmacological activities, including antioxidant, anti-inflammatory, and anticancer effects. In cancer research, orientin has been identified as a potent natural compound that can regulate several fundamental hallmarks of tumor progression, including excessive cell proliferation, resistance to apoptosis, angiogenic signaling, and metastatic dissemination. This review highlights orientin as a low-toxicity, naturally derived flavonoid that is generally well tolerated by healthy cells and may offer cytoprotective and antioxidant benefits, in contrast to conventional chemotherapeutics that often damage normal tissue, while selectively targeting cancer cells. The underlying molecular mechanisms responsible for orientin's inhibition of cancer progression, as well as its potential synergy with other therapies, are summarized. Collectively, these properties suggest a favorable pharmacological profile, supporting its consideration for clinical applications, especially in combination treatment approaches.
Keywords: orientin, cancer cell invasion and metastasis, anti-angiogenesis, combination treatments
Introduction
Orientin, a naturally occurring C-glycosyl flavone widely distributed in medicinal plants such as Ocimum sanctum (holy basil), Phyllostachys spp. (bamboo), and Passiflora incarnata, has emerged as a promising bioactive compound with diverse pharmacological properties. Among its many biological effects, increasing evidence highlights orientin's significant role in cancer prevention and therapy. As a multifunctional phytochemical, orientin exerts strong antioxidant, anti-inflammatory, and DNA-protective activities that contribute to maintaining cellular homeostasis 1-3. However, in vitro and in vivo studies demonstrated that orientin selectively targets malignant cells by modulating redox balance, inducing apoptosis, and suppressing tumor proliferation, invasion, and angiogenesis, while exhibiting minimal toxicity toward normal tissues4-6. Mechanistically, orientin acts through the regulation of key signaling pathways such as NF-κB, COX-2, Hedgehog, and HIF-1α/VEGF, thereby inhibiting tumor-promoting inflammation and angiogenic processes7, 8. Its dual role as both a cytoprotective antioxidant in normal cells and a pro-oxidant in cancer cells underscores its selective therapeutic potential.
Another structurally related compound to orientin is the isoorientin (ISO, luteolin-6-C-glucoside), a C-glycosyl flavone that has been recently reviewed for its effects on cancer cells proliferation, apoptosis, migration, and key signaling pathways across multiple in vitro cancer models, including lung, brain, oral, liver, pancreatic, and gastric cancers9. However, which has not been highlighted before, in this review we focus on orientin, a low-toxicity, naturally derived flavonoid that is generally well tolerated by healthy cells and may offer cytoprotective and antioxidant benefits but selectively targeting cancer cells. The underlying molecular mechanisms responsible for orientin's inhibition of cancer progression, as well as its potential synergy with other therapies, are summarized.
Natural resources of orientin
Among natural sources, Phyllostachys spp. (bamboo) have consistently been reported as the richest repositories of orientin, where it can constitute a large fraction of the leaf flavonoid pool (≈4.9-7.8% of the total extractable flavonoid fraction) and, in some measurements, reach very high absolute levels, around 38mg/g dry leaf1, 3. Passiflora spp., particularly Passiflora incarnata, are also high-yielding sources (≈3.3 mg/g in some dried extracts) and commonly co-contain vicenin-2 and isoorientin, which together contribute to the pharmacological profile10. Ocimum sanctum (holy basil, Tulsi) is another well-documented medicinal source, in which orientin is among the dominant C-glycosyl flavonoids in leaves as identified by LC-MS/MS and HPLC profiling2. Other plant genera, including Fagopyrum, Trollius, Adonis vernalis, Jatropha gossypifolia, Hordeum vulgare, and Anadenanthera, typically contain moderate to low or trace amounts depending on plant part, cultivar, and extraction or analysis method 1, 3. Importantly, reported yields vary widely with analytical approach (HPTLC, HPLC, LC-MS), extraction solvent, plant part (young vs. mature leaves or sprouts), and environmental or developmental factors (light intensity, temperature, growth stage), all of which must be considered when comparing data across studies1, 3.
Predicting the anticancer potential of orientin via in silico approaches
In silico studies have highlighted multiple molecular targets of orientin relevant to cancer therapy. Molecular docking analyses demonstrated that orientin interacts with matrix metalloproteinase-13 (MMP-13) - a promotor for tumor invasion, metastasis, and angiogenesis- by hydrogen bonding of orientin with Pro242 within the catalytic pocket, suggesting inhibition of substrate access and extracellular matrix degradation as well as potential inhibition tumor invasion11. These interactions suggest that the catalytic site of MMP-13 is a key targetable region, where orientin can potentially block extracellular matrix remodeling and metastasis. Additionally, orientin also demonstrated a high binding affinity for dual-specificity tyrosine phosphorylation-regulated kinase 2 (DYRK2). Molecular docking analyses suggested that it is stabilized within the kinase's ATP-binding site through hydrogen bonds and hydrophobic interactions, which could underlie its observed anti-proliferative activity in U87 glioblastoma and Caco-2 colorectal cell lines12. Binding to the ATP-binding pocket of DYRK2 implies potential modulation of downstream cell cycle regulators, including cyclin D1 and p53-mediated apoptotic pathways. Orientin also forms stable interactions near the ATP-binding pocket of JAK2, particularly at Lys882 and Asp939, residues essential for kinase catalysis, consistent with predicted inhibition of JAK2/STAT signaling, a central regulator of cell proliferation and survival 13-15. These docking results confirm the ATP-binding pocket and key catalytic residues of JAK2 as potential molecular targets for orientin. In the meantime, quinone oxidoreductase, a redox-regulating enzyme implicated in hepatocellular carcinoma, was predicted to accommodate orientin via hydrogen bonds at its catalytic site, potentially modulating oxidative stress and cytotoxic signaling16. Targeting quinone oxidoreductase may lead to regulation of ROS-dependent signaling pathways such as Nrf2 and MAPK, contributing to apoptosis and inhibition of tumor growth. However, nano-formulated orientin (NF-O) further improved predicted target engagement and bioavailability, enhancing interactions with angiogenesis-related proteins such as HIF-1α and VEGF, as indicated by stable binding to the PAS domain of HIF-1α, which may reduce tumor neovascularization17. The PAS domain of HIF-1α is a critical site for orientin binding in angiogenesis-related protein inhibition. These in silico findings suggest that orientin interacts with key hotspot residues across multiple cancer-related proteins, offering mechanistic insight into its multi-targeted anticancer activity and highlighting its potential for preclinical testing and future clinical application.
Cytotoxicity of orientin in normal and cancer cells
Orientin was found to have selective cytotoxicity toward cancer cells while exerting minimal toxic effects on or even enhancing antioxidant capacity in normal cells, highlighting its potential as a safe and effective anticancer flavonoid. For instance, orientin was shown to have a minimal toxicity to NIH-3T3 fibroblasts (IC₅₀ >100 µM), consistent with a favorable safety margin in non-malignant cells and above 50 µM in H9c2 rat cardiomyocytes, indicating that orientin exerts its effects without noticeably compromising the viability or structural integrity of normal cells18. Similarly, orientin-treated normal epithelial and endothelial cells maintained normal proliferation and mitochondrial activity, suggesting a low risk of oxidative or apoptotic damage19. Moreover, recent cellular studies (e.g., keratinocyte and skin models; equine stromal cells) demonstrate orientin's protective effects against UV or oxidative stress and no evidence of harming normal cell viability at concentrations used for protection, reinforcing its low cytotoxic profile in normal cells 20, 21. In parallel with that, in human neuroblastoma SH-SY5Y cells, orientin was non-cytotoxic up to 20 µM and even enhanced antioxidant defense under hydrogen peroxide-induced stress, further confirming its biocompatibility22. Together, these observations support that orientin is well tolerated by normal cells and may exert cytoprotective or antioxidant effects, distinguishing it from conventional cytotoxic agents that often compromise normal tissue integrity 1, 23. On the other hand, in vitro studies have demonstrated that orientin significantly inhibits the proliferation of human cancer cell lines. These included hepatocellular carcinoma, colorectal carcinoma4, 24, 25, breast cancer8, 26, Colorectal (HCT-116) lung carcinoma (A549) cells, in a dose-dependent manner5. Moreover, in contrast, in multiple tumor models including colorectal, breast, and liver cancers, orientin triggered caspase-dependent apoptosis and mitochondrial dysfunction, hallmarks of selective cytotoxic action27. Overall, these results demonstrate that orientin exhibits selective cytotoxicity—potently targeting cancer cells while sparing normal ones—making it an attractive natural compound for anticancer therapy with a potentially wide therapeutic margin.
Orientin promotes apoptosis in cancer cells
In EC-109 esophageal cancer cells, orientin induced dose-dependent apoptosis over 24-72 h. Colony formation assays showed increasing inhibition with concentrations from 5 to 80 µM. Flow cytometry analysis demonstrated that orientin at 20 µM, 40 µM, and 80 µM increased apoptotic rates progressively. In the meantime, morphological changes consistent with apoptosis, such as cell shrinkage, membrane blebbing, and nuclear condensation, were also observed at these doses23. In a study investigated the effects of 100 µM orientin on T24 human bladder carcinoma cells, orientin inhibited cell proliferation, induced cell cycle arrest, and promoted apoptosis via decreasing the expression of anti-apoptotic protein Bcl-2 and an increase in pro-apoptotic protein BAX and cleaved caspase-3. That was associated with the suppression of the NF-kappaB and Hedgehog signaling pathways7. However, treatment of HT29 human colorectal carcinoma cells with orientin resulted in a marked, dose-dependent induction of apoptosis accompanied by suppression of cell proliferation. Orientin exposure increased the proportion of apoptotic cells, with significant elevation of early and late apoptosis observed at 6.25 µM and 12.5 µM. At the molecular level, Orientin altered the BAX/BCL-2 ratio in favor of apoptosis via enhancing the mitochondrial release of cytochrome c and Smac/DIABLO into the cytosol, followed by activation of caspase-9 and caspase-3 and cleavage of PARP25. The study also reported DNA damage induction, evidenced by increased γH2AX expression, linking orientin's pro-apoptotic action to oxidative or genotoxic stress mechanisms. On the other hand, Flax straw, a byproduct of Linum usitatissimum L., contains high levels of C-glucosyl flavonoids, including orientin, vitexin, and isoorientin. Extracts from flax straw suppressed proliferation of MCF-7 cells and triggered apoptosis by increasing Bax and caspase-7, -8, and -9 expression, while decreasing Bcl-2 levels26. Conclusively and as summarized in table 2, orientin seems to induce the intrinsic apoptotic pathway by causing DNA damage, elevating oxidative stress, and impairing mitochondrial function, ultimately resulting in cytochrome c release and activation of caspases.
Orientin promotes apoptosis in cancer cells
| Cell Line / Cancer Model | Orientin Conc. / Treatment | Apoptotic Markers / Pathways Affected | References |
|---|---|---|---|
| Esophageal cancer (EC-109) | 80 µM | ↑ p53; ↓ Bcl-2 | An et al., 2015 |
| Human bladder carcinoma (T24) | 20-100 µM | ↓ Bcl-2; ↑ Bax and cleaved caspase-3 | Tian et al., 2019 |
| Human colorectal carcinoma (HT-29) | 6.25-12.5 µM for 24 h (IC50 ≈ 6 µM) | ↑ Bax; ↓ Bcl-2; cytochrome c & Smac/DIABLO release; ↑ cleaved caspase-9, caspase-3, PARP; ↓ XIAP, survivin; ↑ p53, γH2AX; indicates intrinsic mitochondrial apoptosis | Thangaraj et al., 2019 |
| Human bladder carcinoma (T24) | 10-40 µM for 24-48 h | Morphological apoptosis: nuclear condensation, DNA fragmentation; increased apoptotic index (Annexin V/PI staining) | Shen et al., 2020 |
Comprehensive summary of orientin's anticancer mechanisms
| Cancer Type / System | Mechanism of Action | Model System | Reference |
|---|---|---|---|
| Colorectal Cancer | Inhibits angiogenesis via HIF-1α/VEGFA suppression; attenuates NF-κB-mediated inflammation | In vitro (cancer stem cells, CSCs); in vivo (DMH-induced rat model) | Ghosh et al. (2025); Thangaraj & Vaiyapuri (2017) |
| Esophageal Cancer | Induces apoptosis via mitochondrial pathway activation | EC-109 cell line | An et al. (2015) |
| Hepatocellular Carcinoma | Inhibits proliferation and migration; modulates EMT markers | HepG2 cells; xenograft models | Tao et al. (2023) |
| Breast Cancer | Suppresses invasion by downregulating MMP-9 and IL-8 via PKCα/ERK/AP-1/STAT3 signaling | MCF-7 cells (TPA-induced) | Kim et al. (2018) |
| Liver Cancer | Exhibits cytotoxic activity; C-glycoside moiety identified as pharmacophore | HepG2 cell line | Sharma et al. (2016) |
| Bladder Cancer | Triggers apoptosis through inhibition of NF-κB and Hedgehog signaling pathways | T24 cell line | Tian et al. (2019) |
| Vascular Endothelium | Reduces oxidative stress and inflammation by enhancing SESN1-mediated autophagy | HUVECs (ox-LDL-induced model) | Gao et al. (2024) |
| Radiation Protection | Protects lymphocytes and bone marrow cells by minimizing chromosomal damage | In vitro (human lymphocytes); mouse models | Vrinda & Uma Devi (2001); Nayak & Devi (2005) |
| Chemosensitization | Enhances the efficacy of 5-FU in colorectal CSCs | In vitro and in vivo models | Ghosh et al. (2025) |
Orientin inhibits proliferation, migration, and cell cycle progression in cancer cells
Orientin was found to exhibit potent antiproliferative and antimigratory activities in several cancer models (Figure 1). In hepatocellular carcinoma cells, Tao et al. reported that orientin significantly inhibited proliferation and migration of HepG2 and Huh7 cells in a dose-dependent manner5. This effect was accompanied by decreased levels of PCNA and MMP-2/9, suggesting disruption of DNA synthesis and extracellular matrix remodeling. In addition, orientin induced G0/G1 cell cycle arrest by downregulating cyclin D1 and CDK4 while upregulating the cell cycle inhibitor p21, indicating its potential to limit hepatoma growth by modulating the cyclin-CDK pathway and preventing entry into the S phase. Furthermore, orientin regulates members of the Bcl-2 family and inhibitors of apoptosis proteins, while also modulating the tumor suppressor p53, contributing to the activation of programmed cell death pathways28. In human bladder carcinoma T24 cells, Tian et al. demonstrated that orientin suppressed cell proliferation and promoted apoptosis by inhibiting the NF-κB and Hedgehog signaling pathways7. This effect was associated with downregulation of the anti-apoptotic protein Bcl-2 and upregulation of Bax and cleaved caspase-3, indicating interference with key survival and proliferative mechanisms. In the context of colorectal carcinogenesis, Thangaraj and Vaiyapuri demonstrated that orientin significantly suppressed colonic cell proliferation and mitotic activity in rats with 1,2-dimethylhydrazine (DMH)-induced colorectal cancer4. This antiproliferative effect was associated with downregulation of PCNA, Ki-67, and cyclin D1, alongside inhibition of NF-κB-mediated inflammatory mediators such as COX-2 and TNF-α, indicating that orientin modulates both inflammatory and cell cycle-regulatory pathways. Moreover, administration of orientin (10 mg/kg b.w., i.p.) during the initiation, post-initiation, or throughout the entire experimental period markedly reduced tumor marker levels and preserved near-normal colonic histoarchitecture compared to untreated controls. Altogether, these findings highlight orientin as a promising natural agent for colorectal cancer prevention and therapy through combined anti-proliferative and anti-inflammatory mechanisms4. Similarly, Thangaraj et al. confirmed that orientin mitigated DMH-induced colonic aberrant crypt foci and cell proliferation, reinforcing its preventive role in chemically induced colon tumorigenesis24. In esophageal cancer EC-109 cells, An et al. reported that orientin, along with its structural analogue vitexin, markedly inhibited cell proliferation and promoted apoptosis, effects that were associated with upregulated p53 expression and downregulation of the anti-apoptotic protein Bcl-229. This modulation of p53-dependent signaling pathways contributes to orientin's cytostatic and pro-apoptotic activity. As a whole, these findings indicate that orientin exerts its antiproliferative and antimigratory effects by targeting multiple regulatory mechanisms, including NF-κB, Hedgehog, COX-2/PGE-2, and cyclin/CDK signaling pathways. By inducing G0/G1 arrest, downregulating MMPs and cyclins, and modulating apoptosis-related proteins, orientin demonstrates a multi-targeted approach to controlling tumor cell proliferation and migration across diverse cancer types.
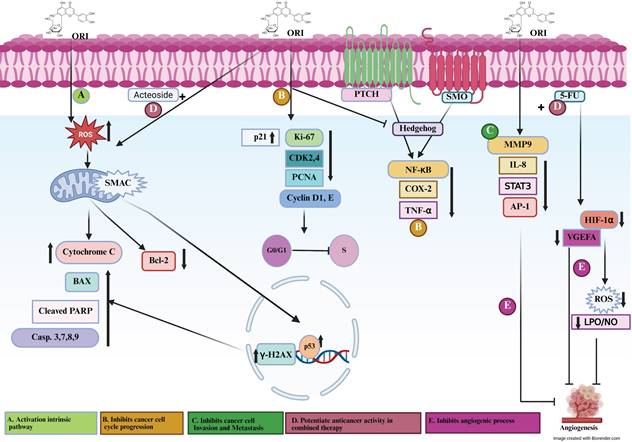
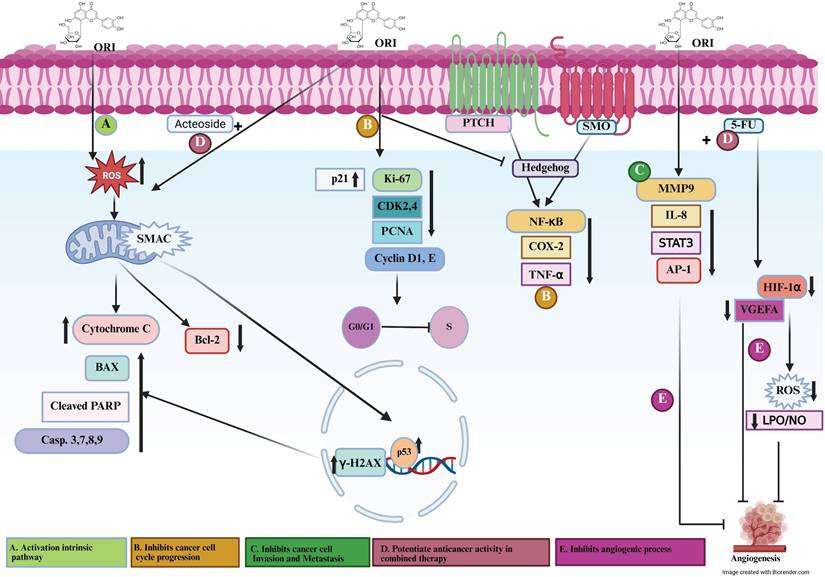
Signaling mechanisms involved in orientin preventing cancer progression. Figure 1. (A) Orientin activates the intrinsic apoptotic pathway by promoting DNA damage and intracellular ROS generation, leading to mitochondrial dysfunction, cytochrome c release, and subsequent caspase the migration activity. (B) Orientin inhibits cancer cell cycle progression, thereby suppressing the proliferative and migratory activities of cancer cells. (C) Orientin Inhibits Cancer Cell Invasion and Metastasis. (D) Orientin potentiates anticancer activity in combined Therapy. (E) Orientin inhibits angiogenic processes and regulates vascular dynamics. ORI, orientin; ROS, reactive oxygen species; SMAC/DIABLO, second mitochondria-derived activator of caspases/direct IAP-binding protein with low PI; Casp, caspase; PARP, poly (ADP-ribose) polymerase; BAX, Bcl-2-associated X protein; Bcl-2, B-cell lymphoma 2; p53, tumor suppressor protein p53; γ-H2AX, phosphorylated histone H2A.X; p21, cyclin-dependent kinase inhibitor 1; CDK2/4, cyclin-dependent kinase 2/4; Cyclin D1/E, cyclin D1/E; PCNA, proliferating cell nuclear antigen; PTCH, patched receptor; SMO, smoothened receptor; NF-κB, nuclear factor kappa B; COX-2, cyclooxygenase-2; TNF-α, tumor necrosis factor alpha; MMP-9, matrix metalloproteinase-9; IL-8, interleukin-8; PKCα, protein kinase C alpha; ERK, extracellular signal-regulated kinase; AP-1, activator protein 1; STAT3, signal transducer and activator of transcription 3; VEGFA, vascular endothelial growth factor A; HIF-1α, hypoxia-inducible factor 1 alpha; LPO, lipid peroxidation; NO, nitric oxide.
Orientin inhibits cancer cell invasion and metastasis
Orientin has demonstrated significant anti-metastatic properties across various cancer types by targeting key molecular pathways involved in cell invasion and metastasis. In TPA-stimulated MCF-7 breast cancer cells, Kim et al. reported that orientin suppressed migration and invasion by downregulating MMP-9 and IL-8 expression through the PKCα/ERK/AP-1/STAT3 signaling pathway, which is critical for extracellular matrix remodeling and metastatic behavior8. Furthermore, Ghosh et al. demonstrated that orientin, when combined with 5-fluorouracil (5-FU), significantly reduced cancer stem cell (CSC)-mediated angiogenesis and metastasis in colorectal cancer by suppressing HIF1α and VEGFA expression6. Elumalai et al. reported that orientin inhibited proliferation and migration in human liver cancer cell lines, highlighting its potential as a therapeutic agent against cancer metastasis17. Collectively, these findings emphasize orientin's multifaceted ability to suppress cancer cell invasion and metastatic progression, supporting its promise as a candidate for developing anti-metastatic therapies.
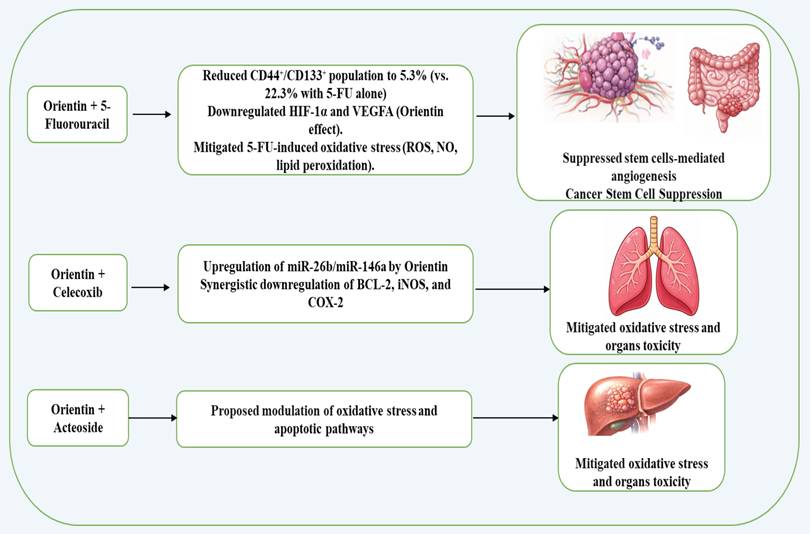
Synergistic anticancer effects of orientin in combined therapy
When being combined with 5-fluorouracil in colorectal cancer, orientin potentiated the 5-fluorouracil effect by suppressing cancer stem cell-mediated angiogenesis through downregulation of HIF-1α and VEGFA, while also mitigating 5-fluorouracil-induced oxidative stress and organ toxicities6. In this combination treatment, orientin markedly decreased the cancer stem cell (CSC) population (CD44⁺/CD133⁺) in HCT116-derived tumorospheres from 22.3% with 5-FU alone to 5.3% with the combination, indicating a strong synergistic effect. That combination also significantly suppressed reactive oxygen species (ROS), nitric oxide (NO), and lipid peroxidation, key mediators of oxidative stress and angiogenic signaling6. Additionally, Orientin has been reported to synergize with the natural compound, Acteoside, to enhance anticancer effects via reduction of oxidative stress and modulation of apoptotic pathways30. Moreover, Khalil et al. reported that orientin synergizes with the COX-2 inhibitor celecoxib to more effectively inhibit A549 lung cancer cell migration, invasion, and pro-survival signaling than either agent alone, indicating a potentiated anticancer effect of the combination treatment18. In all, these studies (summarized in Figure 2) indicate that orientin can act as a synergistic agent in cancer therapy, enhancing the efficacy of existing treatments while reducing toxicity and limiting tumor progression.
Possible synergistic anticancer effects of orientin in combined therapy.
Orientin's anti-angiogenic and vascular-regulating potential
In colorectal cancer, orientin demonstrates strong anti-angiogenic activity by targeting CSC-driven and endothelial-mediated vascularization pathways (Ghosh et al., 2025). Interestingly, conditioned media from orientin-treated tumorospheres inhibited HUVEC tube formation and sprout elongation in chick chorioallantoic membrane (CAM) assays compared with 5-FU alone, confirming an anti-angiogenic effect. Furthermore, nano-formulated orientin (NF-O) enhanced these effects by improving bioavailability, reducing endothelial proliferation markers (CD31), and inhibiting VEGFR phosphorylation, underscoring its potential as a synergistic adjuvant to 5-FU in targeting CSCs and angiogenesis17. Beyond oncology, orientin modulated angiogenesis in vascular injury and reparative contexts where it reduced vascular smooth muscle cell proliferation, inflammatory infiltration, and vessel wall thickening in angiotensin II-induced injury31, but promoted angiogenesis in diabetic wound healing by enhancing tube formation and upregulating VEGF and FGF-2 via activation of the Nrf2/GPX4 pathway and reduction of lipid peroxidation32. Overall, orientin acts as a context-dependent vascular modulator—strongly inhibiting pathological angiogenesis in tumors while promoting functional neovascularization in tissue repair—highlighting its versatility as a therapeutic agent for oncology and regenerative medicine. Taken together, these findings indicate that orientin functions as a context-dependent vascular modulator, suppressing angiogenesis in tumors and pathological remodeling while promoting vascular regeneration during wound healing.
Orientin in medicinal plant extracts and its anticancer potential
In studies dedicated to investigating the anticancer effects of flavonoids in medicinal plant extracts, orientin has been identified among other bioactive compounds as contributing to cytotoxic effects against cancer cell lines. In Passiflora mucronata leaf extract, HPLC-MS analysis detected orientin alongside isoorientin, vitexin, and other flavones, and the hexane fraction exhibited notable cytotoxicity, suggesting that these flavonoids may contribute to apoptosis induction and inhibition of tumor cell proliferation33. Similarly, the extract of flax straw, a byproduct of Linum usitatissimum L., contained orientin, vitexin, and isoorientin, suppressed proliferation of MCF-7 breast cancer cells by inducing apoptosis, increasing Bax and caspase-7, -8, and -9 expression, and decreasing Bcl-2 levels26. In addition, a SIRT6 “fishing” approach using Trigonella foenum-graecum seed extract identified orientin as a binder of the histone deacetylase SIRT6, demonstrating its interaction with biologically relevant targets within complex plant matrices34. Together these findings highlight orientin as a bioactive phytochemical in medicinal plant extracts with promising anticancer effects, supporting its further investigation in natural product-based cancer research.
Based on the available data to date, orientin exhibits a broad spectrum of anticancer activities, including induction of apoptosis, inhibition of proliferation, migration, and cell cycle progression, and suppression of invasion, metastasis, and angiogenesis. Furthermore, its synergistic interactions with conventional chemotherapeutics highlight its potential to enhance treatment efficacy by modulating key oncogenic signaling pathways. A detailed mechanistic overview of orientin-mediated molecular pathways involved in cancer is illustrated in Table 2 and Figure 1. (A) Activate the intrinsic apoptotic pathway via inducing DNA damage and stimulating intracellular ROS production, causing mitochondrial dysfunction and subsequent release of cytochrome c. This cascade triggers caspase activation and promotes PARP cleavage, culminating in apoptosis, and involves BAX and Bcl-2 (pro- and anti-apoptotic regulators of mitochondrial integrity), p53 (DNA damage-responsive transcription factor), γ-H2AX (marker of DNA double-strand breaks), and SMAC/DIABLO (pro-apoptotic mitochondrial protein that neutralizes inhibitor of apoptosis proteins, IAPs). (B) Suppress cell cycle progression through disrupting of key checkpoints controlling the G1/S transition by downregulating CDK2/4 and Cyclin D1/E (cell cycle drivers) and upregulating p21 and PCNA (cell cycle inhibitors and repair proteins), thereby restricting uncontrolled proliferation and migration of cancer cells. (C) Inhibit cancer cell invasion and metastasis and thus attenuates the invasive and metastatic potential of cancer cells by modulating pathways that regulate extracellular matrix degradation and cytoskeletal remodeling, limiting cell motility and metastatic spread. (D) Potentiate anticancer activity in combination therapy by enhancing the efficacy of co-administered chemotherapeutic agents, such as 5-FU and acteoside, through promoting anti-angiogenic effects, sensitizing tumor cells to apoptosis, and reducing drug resistance, thereby indicating synergistic potential. (E) Inhibit angiogenesis and regulate vascular dynamics via disrupting angiogenic signaling by downregulating VEGFA and HIF-1α (key mediators of vascular growth under hypoxia) and modulates inflammatory and oxidative cascades involving NF-κB (master regulator of cytokine expression), COX-2 (prostaglandin biosynthesis enzyme), TNF-α and IL-8 (proinflammatory cytokines), MMP-9 (extracellular matrix-degrading protease), PKCα and ERK (kinases that drive proliferation and survival), AP-1 and STAT3 (transcription factors regulating invasion and angiogenesis), as well as oxidative mediators such as LPO and NO. Additionally, orientin interferes with Hedgehog signaling through PTCH and SMO, thereby inhibiting tumor-associated angiogenic and proliferative responses.
Acknowledgements
Funding
This work was funded by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia [Grant No. KFU260159].
Author contributions
Conceptualization, EAA; methodology, validation, EAA, PR; formal analysis, EAA.; investigation, writing—original draft preparation, EAA.; writing—review and editing, EAA.; visualization, EAA, PR; project administration, EAA; funding acquisition, EAA, PR. All authors have read and agreed to the published version of the manuscript.
Data availability statement
Data of the present study will be provided on request.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Lam KY, Ling AP, Koh RY, Wong YP, Say YH. A Review on Medicinal Properties of Orientin. Advances in pharmacological sciences. 2016;2016:4104595
2. Girme A, Bhoj P, Saste G, Pawar S, Mirgal A, Raut D, Chavan M, Hingorani L. Development and Validation of RP-HPLC Method for Vicenin-2, Orientin, Cynaroside, Betulinic Acid, Genistein, and Major Eight Bioactive Constituents with LC-ESI-MS/MS Profiling in Ocimum Genus. Journal of AOAC International. 2021;104:1634-1651
3. Gu LL, Yao X, An RM, Guo XF. [Determination and antioxidant analysis of seven flavonoids in bamboo-leaf extracts]. Se pu = Chinese journal of chromatography. 2024;42:972-978
4. Thangaraj K, Vaiyapuri M. Orientin, a C-glycosyl dietary flavone, suppresses colonic cell proliferation and mitigates NF-κB mediated inflammatory response in 1,2-dimethylhydrazine induced colorectal carcinogenesis. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie. 2017;96:1253-1266
5. Tao JY, Li J, Wan L, Dong BZ, Yu YJ, Liu YM, Yi ML, Wan LP. Orientin regulates the proliferation and migration of hepatocellular carcinoma cells. Naunyn-Schmiedeberg's archives of pharmacology. 2023;396:2519-2528
6. Ghosh R, Bhowmik A, Biswas S, Samanta P, Sarkar R, Pakhira S, Mondal M, Hajra S, Saha P. Natural flavonoid Orientin restricts 5-Fluorouracil induced cancer stem cells mediated angiogenesis by regulating HIF1α and VEGFA in colorectal cancer. Molecular medicine (Cambridge, Mass). 2025;31:85
7. Tian F, Tong M, Li Z, Huang W, Jin Y, Cao Q, Zhou X, Tong G. The Effects of Orientin on Proliferation and Apoptosis of T24 Human Bladder Carcinoma Cells Occurs Through the Inhibition of Nuclear Factor-kappaB and the Hedgehog Signaling Pathway. Medical science monitor: international medical journal of experimental and clinical research. 2019;25:9547-9554
8. Kim SJ, Pham TH, Bak Y, Ryu HW, Oh SR, Yoon DY. Orientin inhibits invasion by suppressing MMP-9 and IL-8 expression via the PKCα/ ERK/AP-1/STAT3-mediated signaling pathways in TPA-treated MCF-7 breast cancer cells. Phytomedicine: international journal of phytotherapy and phytopharmacology. 2018;50:35-42
9. Laurindo LF, Pomini KT, de Lima EP, Laurindo LF, Rodrigues VD, da Silva Camarinha Oliveira J, Araújo AC, Guiguer EL, Rici REG, Maria DA, de Alvares Goulart R, Direito R, Barbalho SM. Isoorientin: Unveiling the hidden flavonoid's promise in combating cancer development and progression - A comprehensive review. Life sciences. 2025;360:123280
10. Silori Y, Chawla S, De AK, Shirke RP, Grover J, Ramasastry SSV, Sathyamurthy N. Spectral characteristics of the flavones and anthocyanins present in passionflower (Passiflora incarnata). Photochemistry and photobiology. 2024;100:923-935
11. Taherkhani A, Moradkhani S, Orangi A, Jalalvand A, Khamverdi Z. Molecular docking study of flavonoid compounds for possible matrix metalloproteinase-13 inhibition. Journal of basic and clinical physiology and pharmacology. 2020;32:1105-1119
12. Pirvu LC, Pintilie L, Albulescu A, Stefaniu A, Neagu G. Anti-Proliferative Potential of Cynaroside and Orientin-In Silico (DYRK2) and In Vitro (U87 and Caco-2) Studies. International journal of molecular sciences. 2023 24
13. Rajendran P, Ong TH, Chen L, Li F, Shanmugam MK, Vali S, Abbasi T, Kapoor S, Sharma A, Kumar AP, Hui KM, Sethi G. Suppression of signal transducer and activator of transcription 3 activation by butein inhibits growth of human hepatocellular carcinoma in vivo. Clinical cancer research: an official journal of the American Association for Cancer Research. 2011;17:1425-39
14. Rajendran P, Li F, Manu KA, Shanmugam MK, Loo SY, Kumar AP, Sethi G. γ-Tocotrienol is a novel inhibitor of constitutive and inducible STAT3 signalling pathway in human hepatocellular carcinoma: potential role as an antiproliferative, pro-apoptotic and chemosensitizing agent. British journal of pharmacology. 2011;163:283-98
15. Sethi G, Chatterjee S, Rajendran P, Li F, Shanmugam MK, Wong KF, Kumar AP, Senapati P, Behera AK, Hui KM, Basha J, Natesh N, Luk JM, Kundu TK. Inhibition of STAT3 dimerization and acetylation by garcinol suppresses the growth of human hepatocellular carcinoma in vitro and in vivo. Molecular cancer. 2014;13:66
16. Sharma P, Prakash O, Shukla A, Rajpurohit CS, Vasudev PG, Luqman S, Srivastava SK, Pant AB, Khan F. Structure-Activity Relationship Studies on Holy Basil (Ocimum sanctum L.) Based Flavonoid Orientin and its Analogue for Cytotoxic Activity in Liver Cancer Cell Line HepG2. Combinatorial chemistry & high throughput screening. 2016;19:656-666
17. Elumalai Y, Nachammai K, Chandrasekaran K, Kulanthaivel L, Stephen SB, Shil K, Anandan RK, Bima AI, Khan Z, Burzangi AS, Shaik NA, Al-Rayes N, Subbaraj GK. Unleashing the anti-tumor angiogenic potential of nano-formulated orientin: In Silico, In Vitro, and In Ovo studies. PloS one. 2025;20:e0322564
18. Khalil HE, Ibrahim HM, Ahmed EA, Emeka PM, Alhaider IA. Orientin, a Bio-Flavonoid from Trigonella hamosa L, Regulates COX-2/PGE-2 in A549 Cell Lines via miR-26b and miR-146a. Pharmaceuticals (Basel, Switzerland). 2022 15
19. Lee W, Ku SK, Bae JS. Vascular barrier protective effects of orientin and isoorientin in LPS-induced inflammation in vitro and in vivo. Vascular pharmacology. 2014;62:3-14
20. Ma T, He J, Long Q, Wang Y, Chen F, Chen S, Xu K, Cao Y. Orientin attenuates UVB-induced skin photodamage by inhibiting ROS generation via the AMPK/Nrf2 axis. International immunopharmacology. 2025;155:114655
21. Orzoł D, Kępska M, Zyzak M. Orientin Reverses Premature Senescence in Equine Adipose Stromal Cells Affected by Equine Metabolic Syndrome Through Oxidative Stress Modulation. International journal of molecular sciences. 2025 26
22. Law BN, Ling AP, Koh RY, Chye SM, Wong YP. Neuroprotective effects of orientin on hydrogen peroxide-induced apoptosis in SH-SY5Y cells. Molecular medicine reports. 2014;9:947-54
23. Fahmy MI, Sadek MA, Abdou K, El-Dessouki AM, El-Shiekh RA, Khalaf SS. Orientin: a comprehensive review of a promising bioactive flavonoid. Inflammopharmacology. 2025;33:1713-1728
24. Thangaraj K, Natesan K, Settu K, Palani M, Govindarasu M, Subborayan V, Vaiyapuri M. Orientin mitigates 1, 2-dimethylhydrazine induced lipid peroxidation, antioxidant and biotransforming bacterial enzyme alterations in experimental rats. Journal of cancer research and therapeutics. 2018;14:1379-1388
25. Thangaraj K, Balasubramanian B, Park S, Natesan K, Liu W, Manju V. Orientin Induces G0/G1 Cell Cycle Arrest and Mitochondria Mediated Intrinsic Apoptosis in Human Colorectal Carcinoma HT29 Cells. Biomolecules. 2019 9
26. Czemplik M, Mierziak J, Szopa J, Kulma A. Flavonoid C-glucosides Derived from Flax Straw Extracts Reduce Human Breast Cancer Cell Growth In vitro and Induce Apoptosis. Frontiers in pharmacology. 2016;7:282
27. Yuan L, Wang J, Xiao H, Xiao C, Wang Y, Liu X. Isoorientin induces apoptosis through mitochondrial dysfunction and inhibition of PI3K/Akt signaling pathway in HepG2 cancer cells. Toxicology and applied pharmacology. 2012;265:83-92
28. Raza Ishaq A, H ASE-N, A MA-Q, Asfandyar, Bashir A, Younis T. Orientin: a natural glycoside with versatile pharmacological activities. Natural product research. 2025;39:7180-7202
29. An F, Wang S, Tian Q, Zhu D. Effects of orientin and vitexin from Trollius chinensis on the growth and apoptosis of esophageal cancer EC-109 cells. Oncology letters. 2015;10:2627-2633
30. Szkudlarek J, Piwowarczyk L, Jelińska A. Antioxidants Acteoside and Orientin as Emerging Agents in Synergistic Cancer Therapy: A Focus on Innovative Applications. Antioxidants. 2025;14:855
31. Wang S, Yu Y, Feng Y, Zou F, Zhang X, Huang J, Zhang Y, Zheng X, Huang XF, Zhu Y, Liu Y. Protective effect of the orientin on noise-induced cognitive impairments in mice. Behavioural brain research. 2016;296:290-300
32. Zheng Y, Wang X, Pan YJ, Shi XF, Yang L, Lou YL. Orientin suppresses osteoclastogenesis and ameliorates ovariectomy-induced osteoporosis via suppressing ROS production. Food science & nutrition. 2023;11:5582-5595
33. da Silva ICV, Kaluđerović GN, de Oliveira PF, Guimarães DO, Quaresma CH, Porzel A, Muzitano MF, Wessjohann LA, Leal ICR. Apoptosis Caused by Triterpenes and Phytosterols and Antioxidant Activity of an Enriched Flavonoid Extract from Passiflora mucronata. Anti-cancer agents in medicinal chemistry. 2018;18:1405-1416
34. Singh N, Ravichandran S, Spelman K, Fugmann SD, Moaddel R. The identification of a novel SIRT6 modulator from Trigonella foenum-graecum using ligand fishing with protein coated magnetic beads. Journal of chromatography B, Analytical technologies in the biomedical and life sciences. 2014;968:105-11
Author contact
Corresponding author: Emad A Ahmed (PhD), Department of Biological Sciences, College of Science, King Faisal University, Al-Ahsa, 31982, Saudi Arabia; +966568331887; Email: eaahmededu.sa.

 Global reach, higher impact
Global reach, higher impact