Impact Factor
ISSN: 1837-9664
J Cancer 2023; 14(9):1623-1634. doi:10.7150/jca.83719 This issue Cite
Research Paper
Efficacy and Safety Analysis of Immune Checkpoint Inhibitors plus Angiogenesis Inhibitors for the Treatment of Advanced Driver-negative NSCLC in Elderly Patients: A Retrospective Study
1. Department of Radiation Oncology, The Affiliated Suzhou Hospital of Nanjing Medical University, Suzhou Municipal Hospital, Gusu School, Nanjing Medical University, Suzhou 215001, China.
2. Department of Medical Oncology, The Affiliated Suzhou Hospital of Nanjing Medical University, Suzhou Municipal Hospital, Gusu School, Nanjing Medical University, Suzhou 215001, China.
3. Department of Radiation Oncology, The First Affiliated Hospital of Anhui Medical University, Hefei 230031, China.
*Jian Zhang and Zhonghua Zou contributed equally to this work.
Abstract
Background and Objective: Immune checkpoint inhibitors (ICIs) combined with angiogenesis inhibitors may have synergistic effects in elderly patients with advanced driver-negative NSCLC, but its true efficacy remains unclear. In addition, chemotherapy tolerance in elderly NSCLC patients is poor, and the precise identification of the population that may benefit from ICIs combined with angiogenesis inhibitors is also the focus of current research.
Methods: We retrospectively compared the efficacy and safety of ICIs combined with or without antiangiogenic agents in elderly patients with advanced driver-gene negative NSCLC ≥65 years of age in the Cancer Center of Suzhou Hospital Affiliated to Nanjing Medical University. The primary endpoint was PFS. Secondary endpoints were OS, ORR, and immune-related adverse events (irAEs).
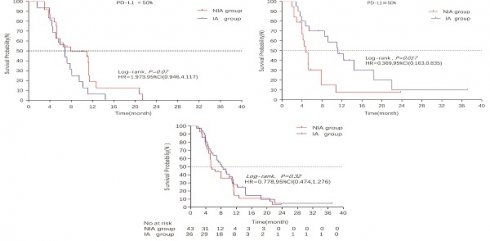
Results: A total of 36 patients in the IA group (immune checkpoint inhibitors plus angiogenesis inhibitors group) and 43 patients in the NIA group (immune checkpoint inhibitors without angiogenesis inhibitors group) were enrolled in the study between January 1, 2019 and December 31, 2021. The median follow-up time for patients in the IA group and NIA group was 18.2 months (95%CI: 14 - 22.5 months) and 21.4 months (95%CI: 16.7 -26.1 months), respectively. The median PFS and median OS were longer in the IA group compared to the NIA group (8.1 months vs 5.3 months; HR for PFS: 0.778, 95%CI: 0.474-1.276, P=0.32; NA vs 30.9 months; HR for OS: 0.795, 95%CI: 0.396-1.595, P=0.519). There were no significant differences in median PFS and median OS between the two groups. Subgroup analysis showed that patients in the IA group had significantly longer PFS in the subgroup with PD-L1 expression ≥50% (P=0.017), and the association between different groups and disease progression was still different in the two subgroups (P for interaction = 0.002). There was no significant difference in ORR between the two groups (23.3% vs 30.5%, P=0.465). The incidence of irAEs in the IA group was lower than that in the NIA group (39.5% vs 19.4%, P=0.05), and the cumulative incidence of treatment interruptions due to irAEs was significantly reduced (P=0.045).
Conclusion: In elderly patients with advanced driver-negative NSCLC, the addition of antiangiogenic agents to ICIs therapy did not provide significant clinical benefit, but the incidence of irAEs and treatment interruptions due to irAEs was significantly reduced. In the subgroup analysis, we found that the clinical benefit of this combination therapy was observed in patients with PD-L1 expression ≥50%, which warrants further exploration.
Keywords: immune checkpoint inhibitors, angiogenesis inhibitors, non-small cell lung cancer, elderly patient, retrospective

 Global reach, higher impact
Global reach, higher impact