Impact Factor ISSN: 1837-9664
J Cancer 2022; 13(6):1713-1724. doi:10.7150/jca.70550 This issue Cite
Review
Cytokeratin Expression Pattern in Human Endometrial Carcinomas and Lymph Nodes Micrometastasis: a Mini-review
1. Vilnius University Hospital “Santaros Clinics”, Department of Gynecology, Vilnius, Lithuania
2. Faculty of Medicine, Vilnius University, Vilnius, Lithuania
3. IIND Department of Gynecology, Lublin Medical University, Lublin, Poland
4. Department of Pathology of Pregnancy, Lublin Medical University, Lublin, Poland
5. Department of Clinical Pathology, Lublin Medical University, Lublin, Poland
Received 2021-12-28; Accepted 2022-2-24; Published 2022-3-14
Abstract
Cytokeratins (CKs) are the largest subgroup of intermediate filament proteins, preferentially expressed in epithelial tissues. CKs play a critical role in determining epithelial structural integrity under stressful conditions in addition to their various fundamental functions in cellular proliferation, apoptosis, migration, adherence and molecular signaling. Immunohistochemical CKs staining could be evaluated with a proper comprehension of their task limitations and their association with the normal morphology to avoid misdiagnosis. Herein, we critically review the CKs expression patterns in ECs in relation to clinicopathological features and patients' outcome. We also briefly discussed the recent advantage of CKs immunohistochemical staining in the detection of EC micrometastasis.
Keywords: Cytokeratin, Endometrial Carcinomas, Lymph Node, Metastasis, OSNA.
Introduction
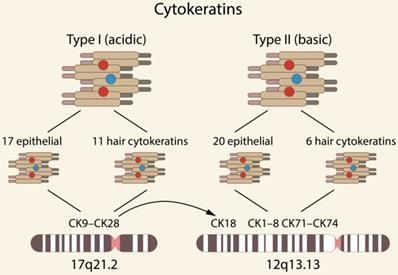
Cytokeratins (CKs) are the largest subgroup of intermediate filament (IF) proteins, preferentially expressed in epithelial tissues, coded by keratin genes [1-2]. Based on the 2D gel migrations, molecular weight and isoelectric point, they are subdivided into type I, i.e., acidic (CK9-CK28) and type II, i.e., basic (CK1-CK8 and CK71-CK74) [1, 3-4]. Type I comprises 17 epithelial and 11 hair keratins, and type II comprises 20 epithelial and 6 hair keratins. Genome analyses have demonstrated that humans possess a total of 54 functional CK genes, i.e., 28 type I and 26 type II CKs, forming two clusters of 27 genes each on chromosomes 17q21.2 and 12q13.13 (the gene for the type I CK18 is located in the type II CK gene domain) [3] [Fig. 1].
Chromosomal localization of selected types of human CKs [3].
CKs are resistant to degradation, show great fidelity of expression, and are very antigenic. All CKs share the common basic molecular structure of cytoplasmic IF proteins. They have a highly conserved central coil α-helical “rod” domain, which is crucial for proper filament assembly. The central domain is surrounded by non-α-helical, N-terminal “head” and C-terminal “tail” domains of various lengths [5-7]. Figure 2 presents the IF protein organization.
A-B. The IF protein organization.
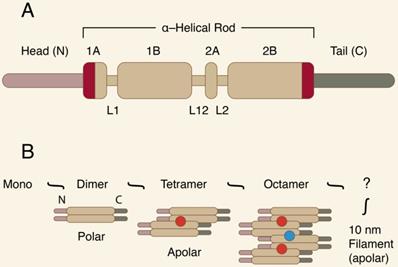
The “rod” domain's sequence homology is usually mutual for the entire protein family, while individual features of particular CK proteins are ensured by variations in the “head” and “tail” domains. CKs are obligatory hetero-polymers, and for proper filament formation, at least one type I and one type II CK must be expressed together. Their expression is differentiation-dependent and developmentally regulated, and they are specific for different types of human epithelia. While liver epithelial cells express only one pair of CKs (CK8 and CK18), all other types of epithelia produce approximately 4 to 8 CKs [7]. For example, stratified squamous epithelia express mostly CKs 1 to 6 and 9 to 17, while CKs 7, 8, 18, 19, and 20 are identified in simple squamous epithelia. Of the latter, CKs 8, 18, and 19 are the most abundant in human malignancies [8].
The point mutations in human CK genes (in the “rod”domain) are associated with different epithelial disorders in multiple tissue types [4, 9]. Some inherited dermal human diseases exhibit cytolysis of epithelial cells, resulting in blistering of the corresponding epithelial sheets [8-10]. These observations, along with several studies conducted on CK knockout mice and mice carrying dominant CK mutations, suggest that CK filaments provide mechanical support to tissue architecture and are critical for the maintenance of cell viability [9-11]. In addition to their cyto-protective function, they form complex signaling platforms and interact with various proteins, such as kinases, adaptors and receptors [12-14]. Moreover, they regulate different cellular processes, including protein synthesis, cell growth and differentiation [15-18].
CKs play a critical role in determining epithelial structural integrity under stressful conditions in addition to their fundamental functions in cellular proliferation, apoptosis, migration, adherence and molecular signaling [13, 18]. They have been conventionally applied as a diagnostic tool in cancer [17-21]. Recently, growing number of evidence suggests their importance, not only in diagnosis, but also in the regulation of the formation of epithelial tumors, as well as in the evaluation of the response to treatment and prognosis [22-27]. In the literature, there are reports investigating the expression patterns of various CKs in diagnosis and therapy of epithelial ovarian cancer [28-33], and in CIN/invasive cervical cancer [34-40]. It is highly debatable if assessment of CK expression pattern may serve as a potential tool for EC development and progression.
There is evidence about an active keratin role in cancer cell invasion and metastasis, highlighting their ability to transform cell shape and migration pattern through interactions with the extracellular environment [41]. Specifically, co-expression of CK8/CK18 in particular type of cells (i.e., fibroblasts) with vimentin in mouse models, as well as in human melanoma and breast cancers cells in vitro indicates enhanced deformability and invasiveness. Moreover, it appears that sphingosylphosphorylcholine, which is abundant in the blood and ascites of ovarian cancer patients, initiates CK8 and CK18 phosphorylation, inducing reorganization in the keratin network. This contributes to changes in the cell shape and better cell migration and permeation abilities. Especially keratin phosphorylation significance was noticed in colorectal cancer progression. As shown in hepatocellular and breast carcinoma cells, CK8 on the cell surface mediates plasmin production via urokinase-type plasminogen activator pathway, which in turn augments malignant cell potential to adhere to fibronectin in the extracellular matrix and promotes detachment from primary tumor [21, 27].
So far, EC has been classically sub-divided into two major categories, endometrioid and non-endometrioid, with different clinico-pathological and molecular features [42-44]. Differences between various EC histotypes have been analyzed based on profound IHC characterization with molecular genetic analysis as well [45-48].
However, recent molecular and histopathological findings recommended a more complex scenario, and new predictive tissue markers are needed to assess the risk stratification. In this context, a major finding has changed the landscape of how we approach EC today, namely, the molecular classification accomplished by the Cancer Genome Atlas (TCGA) in 2013, which subdivides EC in four distinctive subgroups [49-51]. The recent ESGO/ESTRO/ESP guidelines for the management of patients with EC incorporate the molecular classification into the definition of risk groups [52].
Unfortunately, the expression pattern of CKs was not provided as a real validated value in the classification of ECs, but still microscopic evaluation with immunohistochemical staining for CK in lymph node micrometastasis, to identify small clusters of cancer cells, is a “gold” standard [53-54]. In this procedure, they serve as prognostic or predictive biomarkers - even in patients with low-risk or intermediate-risk disease (except for cases showing the lack of myometrial infiltration). There is no review of CKs in EC up to now, suggesting the topic is innovative and of worth interest.
The relevant literature reviewed for this article was retrieved by searching for the terms “endometrial carcinoma”, “cytokeratins”, “micrometastasis” from 1983 to December 2021 in PubMed® database. Further references were identified by analyzing the retrieved publications as well as by the authors' personal knowledge.
Detection of CKs
CK profiling is especially valuable for poorly-differentiated carcinomas, carcinomas spreading over several organs, and, in particular, for distant metastases of an unknown primary origin [55-57].
CKs have also been recognized as prognostic indicators in a variety of epithelial malignancies. Immunohistochemical detection of CKs has become a widely established tool in clinical tumor pathology, where particularly CK5-CK8 and CK18-CK20 are routinely used [1, 31, 54].
Over the past 3 decades, a considerable amount of monoclonal antibodies, that can successfully be implemented on routinely processed tissue specimens, have been developed and are currently commercially available. These antibodies could be broadly categorized into 2 major groups. The term “broad-spectrum” often refers to the first group, which comprises antibodies that react with several CKs and usually stains nearly all types of epithelia and their derived tumors. Those individual clones or mixtures of clones (“cocktails”) in the aforementioned group are commonly used as screening reagents for the demonstration of epithelial differentiation, aiding in distinguishing a poorly differentiated carcinoma from a melanoma, lymphoma or sarcoma [52, 54]. The second group includes antibodies, which recognize only a single CK peptide and whose, therefore, have a more limited immuno-reactivity [54].
AE1/AE3 is an example of a broad-spectrum CK antibody cocktail, and is probably most commonly used in pathological assessment. It is composed of the mouse monoclonal antibody AE1 that recognizes the acidic (type I) CKs 10, 14, 15, 16, and 19, and AE3 that reacts with the basic (type II) CKs 1, 2, 3, 4, 5, 6, 7, and 8. Another broad-spectrum CK antibody cocktail is MNF116. It reacts with CKs 5, 6, 8, 17, and probably 19. Similar to AE1/AE3, this antibody has the drawback of not reacting with CK18.
A brief summary of the main anti-CK antibodies used in the experimental setting is presented in Table 1.
Basic information about the main anti-CK antibodies used in the experimental setting [53].
| Clone | Reactivity | Immunogen | Remarks | Expression in cancers |
|---|---|---|---|---|
| AE1/AE3 | CK1-6, CK8, CK10, CK14, CK15, CK16, CK19. | Epidermal keratin | Does not react with CK18 (some sources have added clone 5D3, which reacts with CK18 and CK8). Cross-reacts with glial fibrillary acidic protein. | Negative in hepatocellular, adrenal cortical, some renal cell carcinomas, renal oncocytomas. |
| KL1 | CK1, CK2, CK5-8, CK11, CK14, CK16-CK18. | Human keratin isolated from epidermal stratum corneum | One of the most sensitive anti-keratin antibodies. Cross-reacts with normal brain tissue and astrocytic tumors. | Stains large majority of hepatocellular carcinomas and renal epithelial tumors. |
| OSCAR | CK7, CK8, CK18, CK19. | Keratin extract from RT-4 and MCF-7 cell lines | Does not react with with normal brain tissue or gliomas. | Limited evidence. |
| MAK-6 (KA4 & UCD/PR10.11) | CK8, CK14, CK15, CK16, CK18, CK19. | KA4 - human sole epidermis UCD/PR10.11 - antigen purified from MCF-7 tissue culture media | Cross-reactivity with neural tissue. | Limited evidence. Indications exist, that this coctail stains all squamous cell carcinomas, majority of adenocarcinomas, transitional cell carcinomas, carcinoid tumors, undifferentiated carcinomas. |
| 34βE12 | CK1, CK5, CK10, CK14. | Human stratum corneum | Sometimes erroneously referred to as keratin 903. | Stains squamous cell carcinomas, nasopharyngeal carcinomas, thymomas. Usually does not stain prostatic and colorectal adenocarcinomas, follicular thyroid and hepatocellular carcinomas. |
| CAM 5.2 | CK7, CK8, CK18, CK19. | Human colorectal carcinoma cell line HT24 | Reactivity restricted primarily to CK8- screening marker for epithelial differentiation. May stain astrocytic tumors. | Reacts with hepatocellular carcinomas, lung and colorectal adenocarcinomas, ovary serous and endometrioid carcinomas, lobular breast carcinoma. Does not stain adrenal cortical carcinomas. |
Expression profiles of selected CKs in human endometrial carcinomas (ECs)
Endometrioid-type carcinoma is almost always positive for CK7, CK8, CK18, and CK19 [56-57], and the vast majority of the cases are CK7 positive and CK20 negative [58-62]. Recently, Miyamoto et al. [63] reported that sero-mucinous component of EC had positive reactivity for CK7, and negative reactivity for CK20, and could be a histologic predictor for prognosis. Several studies have reported different CKs expression patterns in primary human ECs.
In one study, normal endometrial glands were usually CK19-positive [64]. There was a more intense staining in the functional layer, while the basal zone epithelium generally showed weak or even focal immunoreactivity. The proliferative epithelium particularly showed a pattern of more intense staining in the basal and apical cytoplasmic segments. However, it was noticed, that only sizeable neoplastic glands in the middle part of the tumor stained strongly, while peripheral glands were characterized by weak-to-absent CKs' reactivity. In contrast, endometrioid carcinoma with areas of microcystic, elongated and fragmented glands (MELF-type) was homogeneously and strongly CK19 positive, even when the closest ''conventional''- type tumor glands revealed no reactivity [65]. In a previous study of the afomentioned researchers, endometrioid-type ECs with a MELF-pattern of myometrial infiltration tended to show higher expression of CK7 and CK19 [64].
In another report [66], normal endometrial glandular tissues immunostained completely with CK8 and CK18. The aforementioned CKs were mainly distributed around the nuclei, and there was no staining on the surface of the glandular epithelia or within the nucleus. In terms of EC samples, total CK8 scores ranged from 0 up to 6. At least weak and focal staining was reported in all samples, except one. CK8 was distributed mainly around the nucleus in strongly stained tissues. Basal or basal-apical staining in the cytoplasm was seen in weak or moderately stained neoplastic cells. In comparison, CK18 staining was generally more intense, with total CK18 scores ranging from 3 up to 6. Strong and complete cytoplasmic CK18 staining around the nucleus was observed in 61% of cases. Most samples showed evenly distributed cytoplasmic staining, while there was no reaction on the surface of the tumor cells or in the nucleus. Both CK8 and CK18 revealed LVSI in 82% of cases, whereas CK18 not only stained the neoplastic cells emboli and apoptotic cellular remnants in the vessels, but also the vessels themselves. CK18 also strongly stained micrometastasis of pelvic lymph nodes [66].
Interestingly, out of 10 ECs, six (60%) were positive for CK5/6 in a report of Baghla et al. [67]. Four of these cases - pure ECs - displayed only weak positivity for CK5/6. Comparatively, the poorly-differentiated neoplasms displayed increased CK5/6 immunostaining. Other researchers demonstrated that CK5/6 expression was focal, weak or negative, which correlates completely with the data published previously [58, 68]. Two out of six positive cases revealed squamous metaplasia, showing enhanced CK5/6 expression. Similarly, other authors noticed CK5/6 expression in cases of EC with squamous metaplasia [68].
In another report, Stefansson et al. [69] aimed at investigating the association between CK5/6 expression and specific EC phenotypes. CK5/6 expression was found in slightly less than a half of the cases. Endometrioid-type ECs with squamous differentiation (previously deemed “adenosquamous”) revealed a considerably more intense staining with CK5/6 than the pure endometrioid or serous/clear cell carcinomas. Expression of CK5/6 was mainly found in areas with squamous differentiation, but was also reported in non-squamous areas. Cases of normal endometrium, simple endometrial hyperplasia, and complex endometrial hyperplasia were also examined. In normal endometrium, staining of CK 5/6 was generally weak and focal. In comparison, nearly 30% of the dilated glands in simple endometrial hyperplasia revealed a strong cytoplasmic homogeneously distributed CK5/6 positivity. More intense positive staining was detected in areas of squamous metaplasia. In complex endometrial hyperplasia, CK5/6 expression was generally negative, apart from areas with squamous differentiation [69].
Undifferentiated carcinoma of the endometrium (UCAe) is an aggressive, under-recognized, high-grade tumor that occur either in its typical form or in conjunction with low-grade endometrioid carcinoma (i.e. dedifferentiated EC) [70]. UCAe is a high-grade carcinoma that must be distinguished from endometrioid-type EC, FIGO grades 2 and 3, because of its aggressive behavior. In addition, there are immunophenotypic differences between UCAe and other types of ECs that pose problems to the pathologist while evaluating biopsies of recurrent or metastatic disease. There is a considerable morphological overlap between the typical solid growth pattern of UCAe, the solid component of EC and sarcoma, leading to frequently inaccurate pathological assessment. In the literature, the role of a selected group of immunomarkers in the distinction of UCAe from other ECs has been evaluated [71]. Cases of UCAe were stained with antibodies against CK cocktail (AE1/AE3, CAM5.2, MNF116), CK8/18, CK5/6. Of these, 77% were positive for CK cocktail and CK8/18, whereas only 11% were CK 5/6 positive. Moreover, 10 out of 35 (33%) cases were diffusely positive for CK cocktail, 9 (25%) showed patchy staining, 8 (26%) showed focal staining, while 8 (26%) were negative. In addition, 14 out of 34 (41%) cases were diffusely positive for CK8/18, 7 (21%) showed patchy staining, 6 (17%) showed focal staining, and 7 (21%) were negative. CK5/6 was negative in 14 (78%) cases, 3 (17%) showed patchy staining, and only one (5%) was focally positive. The endometrioid component of all the dedifferentiated tumors was diffusely and strongly positive for all the aforementioned CK markers [71].
The use of keratin cocktail is not reliable in distinguishing UCAe from the solid component of endometrioid-type EC. Although most cases of UCAe tended to be just focally positive for CK cocktail (approximately 5%-10%), a new data published from the same Institute revealed that 54% of all cases have either patchy or diffuse expression [70]. This difference might be related to the antibody dilution and retrieval methods applied. In addition, some investigators reported that CK18 may be a new epithelial marker of choice in UCAe [72]. However, another study found that there was no distinct difference between the expression of CK cocktail and CK8/18 - 60% of their cases expressed the latter [71]. Therefore, pankeratin and CK8/18 are equally useful in establishing the epithelial component of UCAe, especially when lymphoma, melanoma or sarcoma is considered in the expanded differential diagnosis [71].
A recently published report on two cases of dedifferentiated EC showed that the undifferentiated component was only focally positive for cytokeratin staining, while the glandular component was diffusely positive [73].
The choice of the appropriate therapeutic plans for uterine endometrioid-type EC depends on the primer and proper diagnosis of the tumor's site of origin, distinguishing primary endocervical adenocarcinomas from uterine neoplasm [42, 74-75]. However, adenocarcinomas of the uterine cervix displayed a considerable overlap with EC in terms of resembling morphological features, making a precise pathological diagnosis challenging. The objective of the reported research was to compare the immunoprofiles of primary cervical adenocarcinoma and EC, using an extended panel of antibodies [76]. Obtained tissue samples were immunostained with pancytokeratin, CK5/6, CK7, CK8/18, CK19, CK20, CK22, and other commercially available antibodies. Only CK8/18 revealed a remarkably higher frequency of positivity in endometrioid-type ECs relative to cervical adenocarcinomas (p=0.002). There were positive 66% of cervical adenocarcinoma in situ cases and 63% of invasive cervical adenocarcinoma cases in comparison to 94% of ECs and 91% of serous carcinoma of the uterus [76].
Five commonly-used IHC markers, including CK7, CK2 and CK34βE12, were applied to analyze their potential use is distinguishing between primary endocervical and endometrial adenocarcinomas [77]. However, IHC expression pattern of antibodies did not differ in frequency from both the two primary adenocarcinomas originated from the female genital tract [77].
The following study aimed to make clear whether the immunohistochemistry of the CK 8/18 monoclonal antibody, instead of CAM 5.2, has potential use in distinguishing between endocervical adenocarcinomas and ECs [78]. The IHC expression of all 3 markers, CK8, 18, and 8/18, revealed no statistically significant (p > 0.05) differences between the immunostaining results (positive vs. negative) in tumors of both gynecologic malignancies. Although CAM 5.2, which was previously thought to react with both CK8 and CK18, has been reported to be helpful in distinguishing between primary endocervical adenocarcinomas and ECs [76], the above mentioned researchers could not confirm this observation (using the true CK8/18 monoclonal antibody). Finally, CAM 5.2 was mistakenly thought to react with both CK 8 and 18, as the latest revised data suggests that CAM 5.2 reacts with CK7 and CK18, rather than CK18. The results of this study support the idea that there is a misleading impression that CK8/18 is differentially expressed in the two aforementioned gynecologic neoplasms [78].
In a study of Chinese researchers [79], the expression of CK5 decreased significantly with malignant transformation of endometrial glands (p < 0.05). Expression of CK5 decreased when clinical stage, histologic grading, and MI increased. These Authors finally concluded that “…malignant transformation might be accompanied by a loss of CK5 expression…” [79].
Until now, knowledge about CKs expression in endometrial carcinoma is limited; hence their implementation in clinical practice faces a considerable challenge. To sum up, based on the evidence currently available, it appears that normal endometrial glands usually stain with CK19 (especially in the functional layer), with CK8 and CK18 (mainly around the nucleus). CK19 together with CK7 are also expressed in ECs with a MELF-pattern of myometrial infiltration. It appears, that CK8 and CK18 could serve not only as potential markers for detection of EC, but also reveal its invasiveness - LSVI and micrometastasis of pelvic lymph nodes. CK8/18 is statistically significantly more frequently expressed in endometrioid-type EC compared with cervical adenocarcinoma, making the challenging diagnostics of primary tumor's site of origin easier, although some authors showed no difference in expression of CK8/18 in these two cancers. In addition, CK8/18 is as useful as pankeratin in staining the epithelial component of UCAe. Loss of CK5/6 expression is frequent in ECs and is associated with aggressive tumor behavior (poorly-differentiated neoplasms) and decreased patients' survival as well. Endometrial hyperplasia CK5/6-negative may be more suspicious to EC progression. CK5/6 also stains more intensely in ECs with squamous metaplasia.
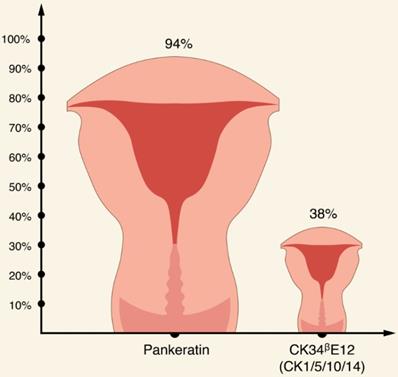
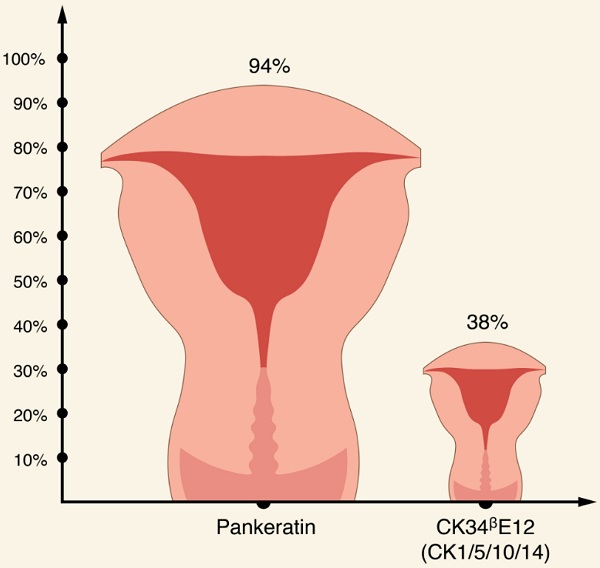
Apart from CKs discussed earlier, it appears that EC usually stains for CK7, while expression of CK20 is usually absent [56, 62, 75, 76, 77]. Interestingly, in one study, all EC specimens stained with CK22 [76]. In terms of broad spectrum CK antibody cocktails, there is an evidence that pankeratin is more specific for EC compared with other broad-spectrum, anti-keratin monoclonal antibody CK34ᵝE12 (CK1/5/10/14) (94% and 38.1%, respectively) [76,77].
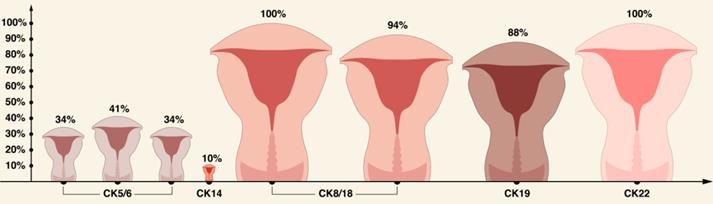
A brief summary of CKs expression pattern in ECs is shown in Table 2 and in Figures 3-5.
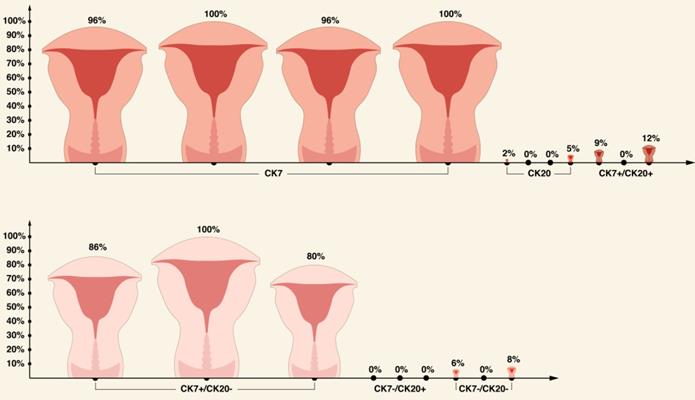
CK expression pattern in primary human ECs based on literature review.
| CKs | Total | Positive | n(%) | Reference |
|---|---|---|---|---|
| CK7 | 55 | 53 | 96 | [56] |
| 10 | 10 | 100 | [62] | |
| 53 | 51 | 96 | [76] | |
| 21 | 21 | 100 | [77] | |
| CK20 | 95 | 2 | 2 | [56] |
| 10 | 0 | 0 | [62] | |
| 52 | 0 | 0 | [76] | |
| 21 | 1 | 4.8 | [77] | |
| CK7+/CK20+ | 35 | 3 | 9 | [56] |
| 10 | 0 | 0 | [62] | |
| 25 | 3 | 12 | [75] | |
| CK7+/CK20- | 35 | 30 | 86 | [56] |
| 10 | 10 | 100 | [62] | |
| 25 | 20 | 80 | [75] | |
| CK7-/CK20+ | 35 | 0 | 0 | [56] |
| 10 | 0 | 0 | [62] | |
| 25 | 0 | 0 | [75] | |
| CK7-/CK20- | 35 | 2 | 6 | [56] |
| 10 | 0 | 0 | [62] | |
| 25 | 2 | 8 | [75] | |
| CK5/6 | 27 | 10 | 34 | [56] |
| 19 | 81 | 41 | [69] | |
| 53 | 18 | 34 | [76] | |
| CK14 | 10 | 1 | 10 | [56] |
| CK8/18 | 20 | 20 | 100 | [56] |
| 51 | 48 | 94 | [76] | |
| CK19 | 48 | 42 | 88 | [76] |
| CK22 | 49 | 49 | 100 | [76] |
| Pankeratin | 50 | 47 | 94 | [76] |
| CK34ᵝE12 (CK1/5/10/14) | 21 | 8 | 38.1 | [77] |
Comparison of CK5/6, CK14, CK8/18, CK19, and CK22 in ECs based on literature review [56, 69, 76].
Prognostic significance and relation with clinicopathological features of CK expression patterns in primary human ECs
It is highly debatable, if assessment of CK expression could serve as a potential tool for establishment of patients' outcome. One study showed that there were no significant differences between the favorable and unfavorable outcome of the early-stage EC compared to CK7 and AE1/AE immunoreactivity [80].
At a practical level, the fact that MELF-type invasion ECs stained strongly with CK19 could encourage the use of this staining as an additional immunohistochemical marker, which may be helpful in showing the extent of myometrial spread, including the unremarkable attenuated glands and single infiltrating cells that often extend beyond the promptly evident conventional tumor areas [81]. Additionally, as CK19 staining also accentuates intravascular neoplastic cells, foci of LVSI could be identified more accurately. Often subtle nodal metastases occurring in MELF-type ECs may also be precisely recognized with CK19 immunostaining [80, 82]. Recently, Rabe et al. [83] suggested the routine CKs staining on sentinel lymph nodes in MELF-pattern ECs to detect metastases and isolated tumor cells.
Similarly, another study demonstrated that staining with CK8 and CK18 may be useful in detecting LVSI, emphasizing that higher stages of ECs had statistically higher LVSI (p ≤ 0.005) [66]. Although EC samples stained variably with CK8 and CK18, there were no statistically significant differences between tumor grade and total CK8 and CK18 staining scores (p = 0.187 and p = 0.675, respectively). Comparably, the total CK8 and CK18 staining scores did not correlate significantly with the clinical stage of the disease (p = 0.412 and p = 0.129, respectively) [66].
As aforementioned, the presence of CK5/6 immunostaining was more frequent in endometrioid-type tumors with squamous differentiation [69]. The loss of CK5/6 expression in ECs (observed in slightly more than a half of the cases) was significantly associated with a higher FIGO stage, increased tumor cell proliferation assessed by Ki-67 expression, reduced β-catenin expression, MI as well as an unfavorable patient outcome (66% 5-year survival vs. 86% 5-year survival (preserved CK5/6 expression), p = 0.0001). For patients with the endometrioid-type EC subgroup, the corresponding figures were 71% and 89%, respectively (p = 0.0004). Comparably, patients with localized disease (characterized by FIGO stages I/II) and with the lack of CK5/6 expression showed significantly reduced survival (79% at 5 years) compared to patients with preserved expression (94%; p = 0.001). CK5/6 turned out to have an independent prognostic value in a multivariate model, including other well-known prognostic factors (histologic type, histologic grade, vascular invasion, myometrial invasion and clinical stage). By substituting histologic grade with nuclear grade, CK5/6 still had an independent prognostic impact (HR = 2.0; p = 0.02), in addition to the histologic type (HR = 2.7; p = 0.013), vascular invasion (HR = 2.9; p = 0.003), myometrial infiltration (HR = 2.6; p = 0.005) and the clinical stage (HR = 5.3; p < 0.0001). When cases of localized disease were studied separately, the variables: histologic type (HR = 3.9; p = 0.012), vascular invasion (HR = 17.3; p < 0.0001) and CK5/6 (HR = 3.9; p = 0.002), still had an independent prognostic impact. Interestingly, in a separate multivariate analysis of the endometrioid subgroup, the role of CK5/6 expression appeared to be of borderline significance (HR = 2.0; p = 0.09), while histologic grade (HR = 2.3; p = 0.03), vascular invasion (HR = 4.4; p = 0.001) and clinical stage (HR=6.1; p < 0.0001) obtained still an independent prognostic value. As a conclusion, that loss of CK5/6 expression is frequent in endometrioid-type ECs and is associated with aggressive tumor behavior and decreased patients' survival rates. In addition, it is an adverse prognostic marker in multivariate analysis [69].
In another report, the investigators assessed the value of the molecular biomarkers in endometrial hyperplasia progressed to uterine carcinoma [84]. In total, 142 cases of endometrial hyperplasia were available for analysis (134 non-endometrial intraepithelial neoplasia; 8 EIN). The median follow-up was 57 months (range, 12-283 months). Interestingly, CK5/6-positive endometrial hyperplasia progress to EC only in 1.2%, whereas the corresponding figure for CK5/6-negative cases was 13.8% (p = 0.001, HR 13.6) [84].
It is worth citing the results of the study by Bai et al. [85] who investigated CK17 IHC pattern in a cohort of 117 high-grade ECs. CK17 immunostaining correlated with decreased OS (HR, 1.8, p = 0.0488). Finally, they suggest that CK17 IHC test results could be clinically applied to make the final decision related to therapeutic intervention [85].
Cytokeratins and EC micrometastases
The presence of micrometastases in regional lymph nodes is one of the most core risk factor determining the widespread of EC corresponding with patients' outcome [86-88]. Novel insights in molecular biology have led to the development of more sensitive and sophisticated methods for detecting micrometastasis to lymph nodes in female genital tract carcinomas, including ECs. CKs staining of lymph nodes from EC patients has been proven to be more sensitive than traditional histopathologic evaluation for the detection of micrometastasis. In a study by Bosquet et al. [89] CK staining was performed with AE1/AE3 antibodies. All lymph nodes proved to have (micro)metastasis by examination of frozen sections during the initial surgery were also shown to be positive during secondary assessment with hematoxylin and eosin-staining. However, 12.5% of all lymph node-negative cases had micrometastasis revealed by CK staining. One of these patients developed recurrent disease in the para-aortic lymph nodes and, unfortunately, died from the disease after almost 3 years of follow-up. This study clearly highlights the clinical significance of micrometastasis investigation. Moreover, an approach incorporating CK staining during lymph nodes assessment could improve the risk evaluation, especially in the highly-risk EC women [89].
In another report, investigators aimed to evaluate the clinic-pathological significance of CKs expression staining in lymph nodes with unconfirmed metastasis, which were assumed to be responsible for the recurrent disease of patients treated for early-stage EC [90]. They examined retrospectively 304 pelvic lymph nodes and 46 primary tumors resected from 46 patients with ECs. The study sample was comprised of 36 women with stage I disease and 10 patients with stage IIIC disease. In the subgroup of stage IIIc, pancytokeratin expression was detected in all 13 lymph nodes with metastasis and also in 20 out of 66 (30.3%) lymph nodes without previously detected metastasis. Furthermore, CK expression was demonstrated in 37 out of 225 (16.4%) lymph nodes with unconfirmed metastasis in the subgroup of stage I disease. In the latter subgroup, CK expression in lymph nodes was detected in 10 out of 14 (71.4%) patients with LVSI. This last was remarkably more frequent than the expression in 4 out of 22 (18.2%) patients without LVSI. More than a third of patients with lymph nodes expressing CK also had a recurrent tumor within the pelvic cavity, whereas all patients with CK-negative lymph nodes showed no recurrence of disease within 5 years after the primary surgery. A relationship between CK expression in the lymph nodes and LVSI of the primary tumor was detected, but there was no association between CK expression and the histologic grade or depth of myometrial invasion. In the multivariate analysis of the subgroup of stage I disease, CK-positive lymph nodes were identified as an independent risk factor for disease recurrence [90].
It was suggested that the detection of CK20 could substitute traditional histopathologic methods in the diagnosis of micrometastasis in lymph nodes of EC patients [91]. The presence of metastases in 10% of all patients was demonstrated by histopathologic examination. These patients were also CK20-positive. Of the remaining 90% patients with negative histopathologic results, 33% were CK20-positive. Results of the meta-analysis also suggest that CK 20 is more sensitive than traditional histopathologic method with H&E (sensitivity was 94.5 and 91%, respectively) [92]. These results encourage the consideration of the use of CK20 more frequently in order to precisely detect micrometastasis in lymph nodes of patients with EC and take the appropriate treatment steps in order to prevent disease recurrence [91].
Altogether, the immunohistochemical staining of CKs in lymph nodes with undetected metastasis by conventional methods could serve as a predictor of occult metastasis to these nodes and increases the likelihood of disease recurrence in early-stage EC [90-93].
There are manuscripts describing advanced molecular techniques (for example qRT-PCR) in detecting EC lymph node micrometastases [94-95]. These techniques are even more sensitive than IHC, but they also carry high false-positive results [94-95]. Interestingly, Pappa et al. [94] assumed that “..qRT-PCR exhibits a better diagnostic accuracy compared with IHC (for the detection of lymph node micrometastasis in cervical, endometrial and vulvar cancer), while CK19 displays a consistent pattern of detection compared to carbonic anhydrase 9…”.
Moreover, OSNA (One-Step Nucleic Acid Amplification) method, based on CK19 mRNA concentration for the detection of lymph-node metastases in EC, has been reported [96-98]. OSNA showed high sensitivity (87.5%-100%) and specificity (82%-100%), suggesting an efficient intraoperative tool for the molecular detection of LNM, especially in early-stage ECs [97]. Additionally, it may also serve as a useful alternative to conventional pathological diagnosis of EC lymph node metastasis. Finally, Diestro et al. [98] reported that “…OSNA has recently received EC marking for the detection of lymph node metastases in endometrial and cervical cancer, allowing its use in routine practice”.
There is one retrospective study from Germany, analyzing follow-up of 428 patients (302 patients with node-negative EC without adjuvant treatment, 95 with nodal micrometastases who received adjuvant treatment and 31 with nodal micrometastases who did not receive adjuvant treatment) [99]. They showed that without adjuvant therapy, the DFS in the cohort of patients with micrometastasis was significantly reduced and adjuvant therapy was associated with improved DFS comparable to the DFS of node-negative patients [99].
Despite the fact of introducing the molecular classification of EC accomplished by the Cancer Genome Atlas (TCGA) in 2013, the NCCN guidelines still recommend mapping of lymph nodes with ultrastaging techniques in the form of H&E and pankeratin staining [100].
Recently, Xu et al. [101] published an interesting study in which the serum CK19 value was analyzed in EC patients undergoing surgical intervention. Interestingly, CK19 level could predict the possibly risk of ovarian metastases, and therefore “…the necessity of incorporating serum CK19 measurement into the pre-operative evaluation of EC, especially as extension of current standard approach with ovarian preservation counselling” is advocated [101].
Conclusions
CKs play a major role in determining epithelial structural integrity under stressful conditions in addition to their fundamental functions in cellular proliferation, apoptosis, migration, adherence, and molecular signaling. They have been conventionally used as a diagnostic tool in cancer. Recently growing evidence suggests their importance not only in diagnosis but also in the regulation of the formation of epithelial tumors as well as in the evaluation of the response to treatment and the prognosis. A significant correlation is exhibited by the expression of particular CK types and tumor phenotypes, aiding in distinguishing between aggressive types of carcinoma, the specific localization of the tumor, the chance of invasion and metastasis, as well as the likelihood of recurrence. In terms of EC, CK5/6, CK7, CK8, CK17, CK18, CK19 appear to be the most promising diagnostic and prognostic markers. CK staining of lymph nodes from EC patients has been proven to be more sensitive than traditional histopathologic evaluation for the detection of micrometastasis. The immunohistochemical staining of selected CKs in lymph nodes with undetected micrometastasis by conventional methods could serve as a predictor of occult micrometastasis increasing the likelihood of disease recurrence. However, the underlying pathology diagnostic methods in gynecological tumors still depend on the histologic evaluation of the hematoxylin and eosin-stained slides. Immunohistochemical stains should be evaluated with a proper comprehension of their limitations (i.e. cross-reactivity) and their association with the normal morphology to avoid misdiagnosis. Extensive investigation into the multifunctional role of CKs in malignant tumors will probably result in the unfolding of upgraded diagnostic and prognostic markers with more potent therapeutic implications in EC.
Abbreviations
CIN: Cervical Intraepithelial Neoplasia; CK: Cytokeratin; DFS: Disease Free Survival; EC: Endometrial Cancer; EIN: Endometrial intraepithelial neoplasia; ESGO: European Society of Gynaecological Oncology; ESP: European Society of Pathology; ESTRO: European Society of Radiation Oncology; FIGO: Federation Internationale de Gynaecologie et d'Obstetrique; HPV: Human Papillomavirus; IHC: Immunohistochemistry; IF: Intermediate Filament; LNM: Lymph Node Metastasis; LVSI: Lymphovascular Space Invasion; MELF: Microcystic, Elongated and Fragmented; MI: Myometrial Invasion; NCCN: National Comprehensive Cancer Newtork; OSNA: One-Step Nucleic Acid Amplification; OS: Overall Survival; UCAe: Undifferentiated Carcinoma of the Endometrium; qRT-PCR: Quantitative Real Time-PCR.
Acknowledgements
We would like to acknowledge our Colleagues from the Departments for valuable comments and meaningful discussion. Our research is funded by grant from Lublin Medical University, Lublin, Poland (Dz. St. 326/21 to AS).
Author Contributions
DV1 contributed to the planning of the manuscript, performed the literature review, wrote the primary draft of the manuscript; VR planned of the manuscript and provided the critical review of the manuscript, AA-G and AS-S contributed to the literature review and provided the critical review of the paper; DL performed the literature review; DV2 planned of the manuscript, wrote the primary draft of the manuscript; AS conceived and designed of the manuscript. All Authors contributed to the article and approved the submitted version.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Moll R. Cytokeratins as markers of differentiation in the diagnosis of epithelial tumors. Subcell Biochem. 1998;31:205-62
2. Coulombe PA, Omary MB. "Hard" and "soft" principles defining the structure, function and regulation of keratin intermediate filaments. Curr Opin Cell Biol. 2002;14:110-22
3. Schweizer J, Bowden PE, Coulombe PA, Langbein L, Lane BE, Magin TM. et al. New consensus nomenclature for mammalian keratins. J Cell Biol. 2006;174:169-74
4. Hesse M, Zimek A, Weber K, Magin TM. Comprehensive analysis of keratin gene clusters in humans and rodents. Eur J Cell Biol. 2004;83:19-26
5. Fuchs E, Weber K. Intermediate filaments: structure, dynamics, function, and disease. Annu Rev Biochem. 1994;63:345-82
6. Herrmann H, Aebi U. Intermediate filaments: molecular structure, assembly mechanism, and integration into functionally distinct intracellular Scaffolds. Annu Rev Biochem. 2004;73:749-89
7. Albers KM. Keratin biochemistry. Clin Dermatol. 1996;14:309-20
8. Moll R, Divo M, Langbein L. The human keratins: biology and pathology. Histochem Cell Biol. 2008;129:705-33
9. Fuchs E, Cleveland DW. A structural scaffolding of intermediate filaments in health and disease. Science. 1998;279:514-9
10. Coulombe PA, Tong X, Mazzalupo S, Wang Z, Wong P. Great promises yet to be fulfilled: Defining keratin intermediate filament function in vivo. Eur J Cell Biol. 2004;83:735-46
11. Gu LH, Coulombe PA. Keratin function in skin epithelia: a broadening palette with surprising shades. Curr Opin Cell Biol. 2007;19:13-23
12. Paramio JM, Jorcano JL. Beyond structure: do intermediate filaments modulate cell signaling? BioEssays. 2002;24:836-44
13. Magin TM, Vijayaraj P, Leube RE. Structural and regulatory functions of keratins. Exp Cell Res. 2007;313:2021-32
14. Pallari HM, Eriksson JE. Intermediate filaments as signaling platforms. Sci STKE. 2006;366:pe53
15. Ku NO, Michie S, Resurreccion EZ, Broome RL, Omary MB. Keratin binding to 14-3-3 proteins modulates keratin filaments and hepatocyte mitotic progression. Proc Natl Acad Sci USA. 2002;99:4373-8
16. Oshima RG. Apoptosis and keratin intermediate filaments. Cell Death Differ. 2002;9:486-92
17. Koch PJ, Roop DR. The role of keratins in epidermal development and homeostasis-going beyond the obvious. J Invest Dermatol. 2004;123:x-xi
18. Kim S, Wong P, Coulombe PA. A keratin cytoskeletal protein regulates protein synthesis and epithelial cell growth. Nature. 2006: 441:362-5.
19. Abdelrahman AE, Rashed HE, Abdelgawad M, Abdelhamid MI. Prognostic impact of EGFR and cytokeratin 5/6 immunohistochemical expression in triple-negative breast cancer. Ann Diagn Pathol. 2017;28:43-53
20. Yang J, Gao S, Xu J, Zhu J. Prognostic value and clinicopathological significance of serum- and tissue-based cytokeratin 18 express level in breast cancer: a meta-analysis. Biosci Rep. 2018;38:BSR20171145
21. Karantza V. Keratins in health and cancer: more than mere epithelial cell markers. Oncogene. 2011;30:127-38
22. Valentin MD, da Silva SD, Privat M, Alaoui-Jamali M, Bignon YJ. Molecular insights on basal-like breast cancer. Breast Cancer Res Treat. 2012;134:21-30
23. Yu KD, Zhu R, Zhan M, Rodriguez AA, Yang W, Wong S. et al. Identification of prognosis-relevant subgroups in patients with chemoresistant triple-negative breast cancer. Clin Cancer Res. 2013;19:2723-33
24. Cen D, Chen J, Li Z, Zhao J, Cai X. Prognostic significance of cytokeratin 19 expression in pancreatic neuroendocrine tumor: A meta-analysis. PLoS One. 2017;12:e0187588
25. Sun D, Zhang Y, Sun X, Chen Y, Qiu W, Ji M, Lv G. Prognostic value of cytokeratin 19 in hepatocellular carcinoma: A meta-analysis. Clin Chim Acta. 2015;448:161-9
26. Volkmer JP, Sahoo D, Chin RK, Ho PL, Tang C, Kurtova AV. et al. Three differentiation states risk-stratify bladder cancer into distinct subtypes. Proc Nat Acad Sci. USA. 2012;109:2078-83
27. Toivola DM, Boor P, Alam C, Strnad P. Keratins in health and disease. Curr Opin Cell Biol. 2015;32:73-81
28. Taube ET, Denkert C, Sehouli J, Unger U, Kunze CA, Budczies J. et al. Cytokeratin 5/6 expression, prognosis, and association with estrogen receptor α in high-grade serous ovarian carcinoma. Hum Pathol. 2017;67:30-6
29. Ordonez NG. Value of immunohistochemistry in distinguishing peritoneal mesothelioma from serous carcinoma of the ovary and peritoneum: a review and update. Adv Anat Pathol. 2006;13:16-25
30. Corr BR, Finlay-Schultz J, Rosen RB, Qamar L, Post MD, Behbakht K. et al. Cytokeratin 5-positive cells represent a therapy resistant subpopulation in epithelial ovarian cancer. Int J Gynecol Cancer. 2015;25:1565-73
31. Chu PG, Weiss LM. Expression of cytokeratin 5/6 in epithelial neoplasms: an immunohistochemical study of 509 cases. Mod Pathol. 2002;15:6-10
32. Nofech-Mozes S, Khalifa MA, Ismiil N, Saad RS, Hanna WM, Covens A. et al. Immunophenotyping of serous carcinoma of the female genital tract. Mod Pathol. 2008;21:1147-55
33. Ricciardelli C, Lokman NA, Pyragius CE, Ween MP, Macpherson AM, Ruszkiewicz A. et al. Keratin 5 overexpression is associated with serous ovarian cancer recurrence and chemotherapy resistance. Oncotarget. 2017;8:17819-32
34. Mills AM, Paquette C, Terzic T, Castle PE, Stoler MH. CK7 immunohistochemistry as a predictor of CIN1 progression: A retrospective study of patients from the quadrivalent HPV vaccine trials. Am J Surg Pathol. 2017;41:143-52
35. Cao L, Sun PL, Yao M, Chen S, Gao H. Clinical significance of CK7, HPV-L1, and koilocytosis for patients with cervical low-grade squamous intraepithelial lesions: a retrospective analysis. Hum Pathol. 2017;65:194-200
36. Huang EC, Tomic MM, Hanamornroongruang S, Meserve EE, Herfs M, Crum CP. p16ink4 and cytokeratin 7 immunostaining in predicting HSIL outcome for low-grade squamous intraepithelial lesions: a case series, literature review and commentary. Mod Pathol. 2016;29:1501-10
37. Paquette C, Mills AM, Stoler MH. Predictive value of cytokeratin 7 immunohistochemistry in cervical low-grade squamous intraepithelial lesion as a marker for risk of progression to a high-grade lesion. Am J Surg Pathol. 2016;40:236-43
38. da Costa LBE, Triglia RDM, Andrade LALDA. p16INK4a, cytokeratin 7, and Ki-67 as potential markers for low-grade cervical intraepithelial neoplasia progression. J Low Genit Tract Dis. 2017;21:171-6
39. Lee H, Lee H, Cho YK. Cytokeratin7 and cytokeratin19 expression in high grade cervical intraepithelial neoplasm and squamous cell carcinoma and their possible association in cervical carcinogenesis. Diagn Pathol. 2017;12:18
40. Mockler D, Escobar-Hoyos LF, Akalin A, Romeiser J, Shroyer AL, Shroyer KR. Keratin 17 is a prognostic biomarker in endocervical glandular neoplasia. Am J Clin Pathol. 2017;148:264-73
41. Loschke F, Seltmann K, Bouameur JE. Regulation of keratin network organization. Curr Opin Cell Biol. 2015;32:56-64
42. Ronnett BM, Zaino RJ, Ellenson LH, Kurman RJ. Endometrial carcinoma. [in] Blaustein's Pathology of the Female Genital Tract, Kurman RJ [Editor], Springer. 2002:501-59
43. Lu KH, Broaddus RR. Endometrial cancer. N Engl J Med. 2020;383:2053-64
44. Huvila J, Pors J, Thompson EF, Gilks CB. Endometrial carcinoma: molecular subtypes, precursors and the role of pathology in early diagnosis. J Pathol. 2021;253:355-65
45. Reid-Nicholson M, Iyengar P, Hummer AJ, Linkov I, Asher M, Soslow RA. Immunophenotypic diversity of endometrial adenocarcinomas: implications for differential diagnosis. Mod Pathol. 2006;19:1091-100
46. McConechy MK, Ding J, Cheang MC, Wiegand K, Senz J, Tone A. et al. Use of mutation profiles to refine the classification of endometrial carcinomas. J Pathol. 2012;228:20-30
47. Yasuda M. Immunohistochemical characterization of endometrial carcinomas: Endometrioid, serous, and clear cel adenocarcinomas in association with genetic analysis. J Obstet Gynaecol Res. 2014;40:2167-76
48. Szewczuk W, Szewczuk O, Czajkowski K, Gornicka B, Ilczuk T, Semczuk A. ARID1A and GRP30 expression patterns characterize the different histological subtypes of endometrial carcinoma. Eur J Gynaecol Oncol. 2021;42:1138-45
49. Winterhoff B, Hamidi H, Wang C, Kalli KR, Fridley BL, Dering J. et al. Molecular classification of high grade endometrioid and clear cell ovarian cancer using TCGA gene expression signatures. Gynecol Oncol. 2016;141:95-100
50. De Leo A, de Biase D, Lenzi J, Barbero G, Turchetti D, Grillini M. et al. ARID1A and CTNNB1/beta-Catenin molecular status affects the clinicopathologic features and prognosis of endometrial carcinoma: Implications for an improved surrogate molecular classification. Cancers. 2021;13:950
51. Kasius JC, Pijnenborg JMA, Lindemann K, Forsse D, van Zwol J, Kristensen GB. et al. Risk stratification of endometrial cancer patients: FIGO stage, biomarkers and molecular classification. Cancers. 2021;13:5848
52. Concin N, Matias-Guiu X, Vergote I, Cibula D, Mirza MR, Marnitz S. et al. ESGO/ESTRO/ESP guidelines for the management of patients with endometrial carcinoma. Int J Gynecol Cancer. 2021;31:12-39
53. Ordonez NG. Broad-spectrum immunohistochemical epithelial markers: a review. Hum Pathol. 2013;44:1195-215
54. Murali R, Davidson B, Fadare O, Carlson JA, Crum CP, Gilks CB. et al. High-grade endometrial carcinomas: Morphologic and immunohistochemical features, diagnostic challenges and recommendations. Int J Gynecol Pathol. 2019;38(Iss 1 Suppl 1):40-63
55. Moll R, Levy R, Czernobilsky B, Hohlweg-Majert P, Dallenbach-Hellweg G, Franke WW. et al. Cytokeratins of normal epithelia and some neoplasms of the female genital tract. Lab Invest. 1983;49:599-610
56. Chu PG, Weiss LM. Keratin expression in human tissues and neoplasms. Histopathology. 2002;40:403-39
57. Vasilevska D, Semczuk-Sikora A, Lewkowicz D, Jóźwik M, Bogusiewicz M, Semczuk A. Expression of selected cytokeratins in neoplastic endometrium. The 24th World Congress on Advances in Oncology and 24th International Symposium on Molecular Medicine, Sparta, Greece, 10-12 October 2019. Int J Mol Med. 2019;44:S47
58. Zemer R, Fishman A, Bernheim J, Zimlichman S, Markowicz O, Altaras M. et al. Expression of cytokeratin-20 in endometrial carcinoma. Gynecol Oncol. 1998;70:410-13
59. Hubers JA, Soni A. A rare case of endometrial cancer metastatic to the sigmoid colon and small bowel. Case Rep Gastr Med. 2017 ID 9382486
60. El M'rabet FZ, Hottinger A, George AC, Tille JC, El Mesbahi O, Castiglione-Gertsch M. et al. Cutaneous metastasis of endometrial carcinoma: a case report and literature review. J Clin Gynecol Obstet. 2012;1:19-23
61. Blazer III DG, Ramirez PT, Wang H, Fleming JB. Distal pancreatectomy for isolated metastasis of endometrial carcinoma to the pancreas. J Pancreas. 2008;9:56-60
62. Chu P, Wu E, Weiss LM. Cytokeratin 7 and cytokeratin 20 expression in epithelial neoplasms: a survey of 435 cases. Mod Pathol. 2000;13:962-72
63. Miyamoto M, Takano M, Aoyama T, Soyama H, Yoshikawa T, Tsuda H. et al. Seromucinous component in endometrioid endometrial carcinoma as a histological predictor of prognosis. J Gynecol Oncol. 2018;29:e20
64. Stewart CJR, Crook ML, Lacey J, Louwen K. Cytokeratin 19 expression in normal endometrium and in low-grade endometrioid adenocarcinoma of the endometrium. Int J Gynecol Pathol. 2011;30:484-91
65. Stewart CJR, Crook ML, Doherty DA. Micro-anatomical variation in cellular proliferation in endometrial adenocarcinoma, and inverse correlation between Ki67 and cytokeratin 7 expression. Histopathology. 2010;57:46-54
66. Kabukcuoglu S, Ozalp SS, Yalcin OT, Colak E, Abubakar AA. Comparison of histopathologic classification and surgical stage by cytokeratin 8 and cytokeratin 18 in endometrial cancer. Eur J Gynaecol Oncol. 2010;31:641-54
67. Baghla A, Choudhry S, Kataria A. Immunohistochemical expression of cytokeratin 5/6 in gynaecological tumors. Internet J Pathol. 2012;13:2
68. Moll R, Pitz S, Levy R, Weikel W, Franke WW, Czernobilsky B. Complexity of expression of intermediate filament proteins, including glial filament protein, in endometrial and ovarian adenocarcinomas. Hum Pathol. 1991;22:989-1001
69. Stefansson IM, Salvesen HB, Akslen LA. Loss of p63 and cytokeratin 5/6 expression is associated with more aggressive tumors in endometrial carcinoma patients. Int J Cancer. 2006;118:1227-33
70. Altrabulsi B, Malpica A, Deavers MT, Bodurka DC, Broaddus R, Silva EG. Undifferentiated carcinoma of the endometrium. Am J Surg Pathol. 2005;29:1316-21
71. Ramalingam P, Masand RP, Euscher ED, Malpica A. Undifferentiated carcinoma of the endometrium: An expanded immunohistochemical analysis including PAX-8 and basal-like carcinoma surrogate markers. Int J Gynecol Pathol. 2016;35:410-8
72. Tafe LJ, Garg K, Chew I, Tornos C, Soslow RA. Endometrial and ovarian carcinomas with undifferentiated components: clinically aggressive and frequently underrecognized neoplasms. Mod Pathol. 2010;23:781-9
73. Boler AK, Akhtar S, Bandyopadhyay A, Roy S. A report of two cases of dedifferentiated endometrioid carcinoma: A newly described underrecognized tumor of poor prognosis. Indian J Pathol Microbiol. 2020;63:91-3
74. Mittal K, Soslow R, McCluggage WG. Application of immunohistochemistry to gynecologic pathology. Arch Pathol Lab Med. 2008;132:402-23
75. Wang NP, Zee S, Zarbo RJ, Bacchi C, Gown A. Coordinate expression of cytokeratins 7 and 20 defines unique subsets of carcinomas. Appl Immunohistochem. 1995;3:99-107
76. Alkushi A, Irving J, Hsu F, Dupuis B, Liu CL, Rijn M. et al. Immunoprofile of cervical and endometrial adenocarcinomas using a tissue microarray. Virchows Arch. 2003;442:271-7
77. Han CP, Kok LF, Lee MY, Wu TS, Ruan A, Cheng YW. et al. Five commonly used markers (p53, TTF1, CK7, CK2 and CK34betaE12) are of no use in distinguishing between primary endocervical and endometrial adenocarcinomas in a tissue microarray extension study. Arch Gynecol Obstet. 2010;281:317-23
78. Hsu JD, Yao CC, Lee MY, Kok LF, Wang PH, Tyan YS. et al. True cytokeratin 8/18 immunohistochemistry is of no use in distinguishing between primary endocervical and endometrial adenocarcinomas in a tissue microarray study. Int J Gynecol Pathol. 2010;29:282-9
79. Zhao LJ, Li XP, Qi WJ, Wang JL, Wei LH. Value of T cell receptor gamma alternate reading frame protein and keratin 5 in endometrial carcinoma. Chin Med J. 2013;126:4260-4
80. Zou S, Sun H, Fan L, Xiao X, Gong L, Zhu J. et al. Prognostic indicators in patients with early stage endometrioid adenocarcinoma: a retrospective case-control study of 523 patients. Int J Clin Exp Med. 2017;10:3692-8
81. Stewart CJR, Brennan BA, Leung YC, Little L. MELF pattern invasion in endometrial carcinoma: association with low grade, myoinvasive endometrioid tumours, focal mucinous differentiation and vascular invasion. Pathology. 2009;41:454-9
82. Pavlakis K, Messini I, Vrekoussis T, Panoskaltsis T, Chrysanthakis D, Yiannou P. et al. MELF invasion in endometrial cancer as a risk factor for lymph node metastasis. Histopathology. 2011;58:966-73
83. Rabe KM, Klein ME, Ghatak S, Stout I, Schefter A, Erickson BK. et al. Sentinel nodal metastasis detection in endometrial carcinoma with Microcystic, Elongated, and Fragmented (MELF) pattern by cytokeratin immunostaining. Am J Surg Pathol. 2021;156:846-52
84. Steinbakk A, Gudlaugsson E, Aasprong OG, Skaland I, Malpica A, Feng W. et al. Molecular biomarkers in endometrial hyperplasias predict cancer progression. Am J Obstet Gynecol. 2011;204:357.e1-e12
85. Bai JDK, Babu S, Roa-Pena L, Hou W, Akalin A, Eskobar-Hoyos LF. et al. Keratin 17 is a negative prognostic biomarker in high-grade endometrial carcinomas. Hum Pathol. 2019;94:40-50
86. Bogani G, Mariani A, Paolini B, Ditto A, Raspagliesi F. Low-volume disease in endometrial cancer: The role of micrometastasis and isolated tumor cells. Gynecol Oncol. 2019;153:670-5
87. Gomez-Hidalgo NR, Ramirez PT, Ngo B, Perez-Hoyos S, Coreas N, Sanchez-Iglesias JL. et al. Oncologic impact of micrometastases or isolated tumor cells in sentinel lymph nodes of patients with endometrial cancer: a meta-analysis. Clin Transl Oncol. 2020;22:1272-9
88. Yoshioka T, Suzuki Y, Imai Y, Ruiz-Yokota N, Yamanaka S, Furuya M. et al. Inspection for micrometastasis is essential for predicting the prognosis of serous endometrial intraepithelial carcinoma: Case report and literature review. J Obstet Gynaecol Res. 2021;47:4484-9
89. Bosquet GJ, Keeney GL, Mariani A, Webb MJ, Cliby WA. Cytokeratin staining of resected lymph nodes may improve the sensitivity of surgical staging for endometrial cancer. Gynecol Oncol. 2003;91:518-25
90. Yabushita H, Shimazu M, Yamada H, Sawaguchi K, Noguchi M, Nakanishi M. et al. Occult lymph node metastases detected by cytokeratin immunohistochemistry predict recurrence in node-negative endometrial cancer. Gynecol Oncol. 2001;80:139-44
91. Fishman A, Klein A, Zemer R, Zimlichman S, Bernheim J, Cohen I. et al. Detection of micrometastasis by Cytokeratin-20 (Reverse Transcription Polymerase Chain Reaction) in lymph nodes of patients with endometrial cancer. Gynecol Oncol. 2000;77:399-404
92. Gomez-Hidalgo NR, Ramirez PT, Ngo B, Perez-Hoyos S, Coreas N, Sanchez-Iglesias JL. et al. Oncologic impact of micrometastases or isolated tumor cells in sentinel lymph nodes of patients with endometrial cancer: a meta-analysis. Clin Transl Oncol. 2020;22:1272-9
93. Pineda VG, Gutierrez AH, Segovia MG, Ridruejo JS, Diestro Tejada MD, Zapardiel I. Low-volume nodal metastasis in endometrial cancer: Risk factors and prognostic significance. J Clin Med. 2020;9:1999
94. Pappa KI, Rodolakis A, Chrostodoulou J, Gazouli M, Markari S, Antsaklis A. et al. Comparative assessment of lymph node micrometastasis in cervical, endometrial and vulvar cancer: insights on the real time qRT-PCR approach versus immunohistochemistry, employing dual molecular markers. Biomed Res Int. 2014. 2014 187684
95. Togami S, Kawamura T, Fukuda M, Yanazume S, Kamio M, Kobayashi H. Quantitative RT-PCR assay for detecting lymph node metastasis in endometrial cancer: A preliminary study. Oncology. 2019;96:179-82
96. Lopez-Ruiz ME, Diestro MD, Yebenes L, Berjon A, Diaz de la Naval B, Mendiola M. et al. One-step Nucleic Acid Amplification (OSNA) for the detection of sentinel lymph node metastasis in endometrial cancer. Gynecol Oncol. 2016;143:54-9
97. Fanfani F, Monterossi G, Ghizzoni V, Rossi ED, Dinoi G, Inzani F. et al. One-step nucleic acid amplification (OSNA): A fast molecular test based on CK19 mRNA concentration for assessment of lymph-nodes metastases in early stage endometrial cancer. PLoS One. 2018;13(e):0195877
98. Diestro MD, Berjon A, Zapardiel I, Yebenes L, Ruiz I, Lekuona A. et al. One-Step nucleic acid amplification (OSNA) of sentinel lymph node in early-stage endometrial cancer: Spanish multicenter study (ENDO-OSNA). Cancers. 2021;13:4465
99. Ignatov A, Lebius C, Ignatov T, Ivros S, Knueppel R, Papathemelis T. et al. Lymph node micrometastases and outcome of endometrial cancer. Gynecol Oncol. 2019;154:475-9
100. Abu-Rustum NR, Yashar CM, Bean S, Bradley K, Campos SM, Chon HS. et al. NCCN guidelines, version 4,2019. uterine neoplasms/endometrial carcinoma. Plymouth Meeting PA: National Comprehensive Cancer Network (NCCN). 2019
101. Xu J, Chen C, Xiong J, Wang H, Linghu H. Predictive value of serum cytokeratin 19 level for the feasibility of conserving ovaries in endometrial cancer. Front Med. 2021;8:670109
Author Biography
 Andrzej Semczuk is a professor of obstetrics and gynecology at the IInd Department of Gynecology, Lublin Medical University, Lublin, Poland. He has coauthored over 120 peer-reviewed publications including J Cancer, Cancer Biol Ther, J Clin Med, Histol Histopathol, Cancer Invest, BMC Cancer, Oncology, J Ovarian Res, Breast Cancer Res Treat, Gynecol Oncol, and many others. He is a Member of the EU Bioendocar grant entitled “Targeted proteomics, targeted and non-targeted metabolomics for development of diagnostic and prognostic algorithms of endometrial carcinoma”. The current research interest is focused on study the molecular and immunohistochemical alterations associated with the development and dissemination of endometrial cancer and female genital tract carcinosarcoma. He is a Member of the Editorial Board of the J Cancer.
Andrzej Semczuk is a professor of obstetrics and gynecology at the IInd Department of Gynecology, Lublin Medical University, Lublin, Poland. He has coauthored over 120 peer-reviewed publications including J Cancer, Cancer Biol Ther, J Clin Med, Histol Histopathol, Cancer Invest, BMC Cancer, Oncology, J Ovarian Res, Breast Cancer Res Treat, Gynecol Oncol, and many others. He is a Member of the EU Bioendocar grant entitled “Targeted proteomics, targeted and non-targeted metabolomics for development of diagnostic and prognostic algorithms of endometrial carcinoma”. The current research interest is focused on study the molecular and immunohistochemical alterations associated with the development and dissemination of endometrial cancer and female genital tract carcinosarcoma. He is a Member of the Editorial Board of the J Cancer.
Corresponding author: Prof. Andrzej Semczuk Email: andrzej.semczukpl

 Global reach, higher impact
Global reach, higher impact