Impact Factor ISSN: 1837-9664
J Cancer 2021; 12(9):2643-2653. doi:10.7150/jca.53303 This issue Cite
Research Paper
Bevacizumab Plays a double-edged role in Neoadjuvant Therapy for Non-metastatic Breast Cancer: A Systemic Review and Meta-Analysis
1. Beijing University of Chinese Medicine, No. 11, Bei San Huan Dong Lu, Chaoyang District, Beijing, 100029, China.
2. Oncology Department, Beijing Hospital of Traditional Chinese Medicine, Capital Medical University, No.23 Back Road of Art Gallery, Dongcheng District, Beijing, 100010, China.
Abstract
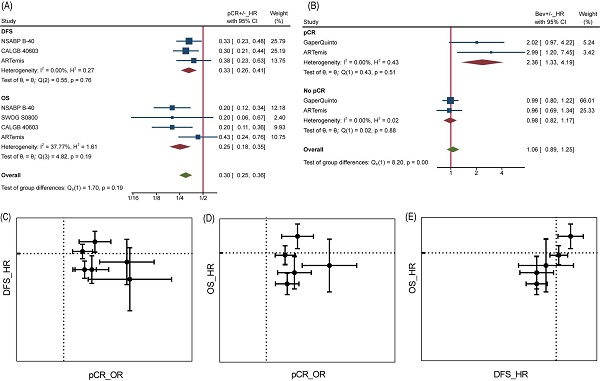
The anti-angiogenic drug Bevacizumab (Bev) is engaged in neoadjuvant therapy for non-metastatic breast cancer (NMBC). However, whether neoadjuvant Bev providing a greater benefit to patients is debatable. Our study aimed to review Bev's role in Neoadjuvant therapy (NAT) in NMBC and identify predictive markers associated with its efficacy by systemic review and meta-analysis. Eligible trials were retrieved from the Pubmed, Embase, and Cochrane Library, and random or fixed effects models were applied to synthesize data. Power of pCR to predict DFS or OS was evaluated by nonlinear mixed effect model. In NMBC, Bev significantly improved the rate of patients achieving pCR, but this benefit discontinued in DFS or OS. Biomarkers such as PAM50 intrinsic subtype, VEGF overexpression, regulation of VEGF signaling pathway, hypoxia-related genes, BRCA1/2 mutation, P53 mutation and immune phenotype can be used to predict Bev-inducing pCR and/or DFS/OS. Unfortunately, although patients with pCR survived longer than those without pCR when ignoring the use of Bev, but patients achieving pCR with Bev might survive shorter than those achieving pCR without Bev. Subgroup analyses found Bev prolonged patients' OS when given pre- and post-surgery. Lastly, adding Bev increased adverse effects. Overall, Bev offered limited effect for patients with NMBC in an unscreened population. However, in biomarkers - identified subgroup, Bev could be promising to ameliorate the prognosis of specific patients with NMBC.
Keywords: Bevacizumab, neoadjuvant therapy, surrogate endpoint, prognostic biomarker, non-metastatic breast cancer.

 Global reach, higher impact
Global reach, higher impact