Impact Factor
ISSN: 1837-9664
J Cancer 2021; 12(8):2371-2384. doi:10.7150/jca.51173 This issue Cite
Research Paper
Identification of a Prognostic Signature Model with Tumor Microenvironment for predicting Disease-free Survival after Radical Prostatectomy
1. Department of Epidemiology and Biostatistics, College of Public Health, Zhengzhou University, Zhengzhou 450001, China.
2. Department of Medicine, Zhengzhou First People's Hospital, Zhengzhou 450004, China.
3. Department of Urology, Henan Province Hospital of Traditional Chinese Medicine, Zhengzhou 450002, China.
* ?
Abstract
Background: The tumor microenvironment (TME) and immune checkpoint inhibitors have been shown to promote active immune responses through different mechanisms. We attempted to identify the important prognostic genes and prognostic characteristics related to TME in prostate cancer (PCa).
Methods: The gene transcriptome profiles and clinical information of PCa patients were obtained from The Cancer Genome Atlas (TCGA) database, and the immune and stromal scores were calculated by the ESTIMATE algorithm. We evaluated the prognostic value of the risk score (RS) model based on univariate Cox analysis and least absolute shrinkage and selection operation (LASSO) Cox regression analysis and established a nomogram to predict disease-free survival (DFS) in PCa patients. The GSE70768 dataset was utilized for external validation. Twenty-two subsets of tumor-infiltrating immune cells were analyzed using the CIBERSORT algorithm.
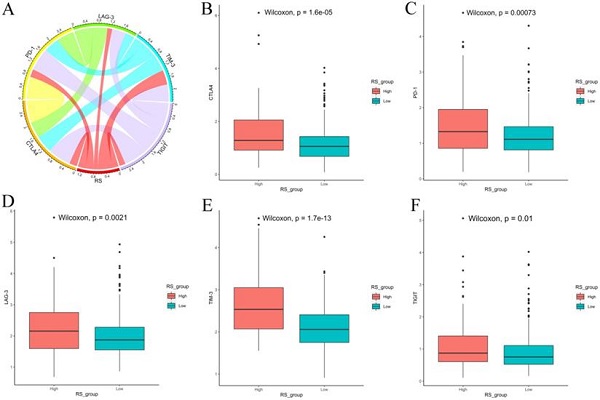
Results: In this study, the patients with higher immune/stromal scores were associated with a worse DFS, higher Gleason score, and higher pathological T stage. Based on the immune and stromal scores, 515 differentially expressed genes (DEGs) were identified. The univariate Cox and LASSO Cox regression models were employed to select 18 DEGs from 515 DEGs and construct an RS model. The DFS of the high-RS group was significantly lower than that of the low-RS group (P<0.001). The AUCs for the 1-year, 3-year and 5-year DFS rates in the RS model were 0.890, 0.877 and 0.841, respectively. A nomogram of DFS was established based on the RS and Gleason score, and the AUCs for the 1-year, 3-year and 5-year DFS rates in the nomogram were 0.907, 0.893, and 0.872, respectively. These results were further validated in the GSE70768 dataset. In addition, the proportion of Tregs was determined to be higher in high-RS patients (P<0.05), and the expression levels of five immune checkpoints (CTLA-4, PD-1, LAG-3, TIM-3 and TIGIT) were observed to be higher in high-RS patients (P<0.05).
Conclusions: Our study established and validated an 18-gene prognostic signature model associated with TME, which might serve as a prognosis stratification tool to predict DFS in PCa patients after radical prostatectomy.
Keywords: prostate cancer, radical prostatectomy, microenvironment, stromal, immune, prognosis, prediction

 Global reach, higher impact
Global reach, higher impact