Impact Factor ISSN: 1837-9664
J Cancer 2018; 9(1):13-20. doi:10.7150/jca.21460 This issue Cite
Research Paper
Inflammatory Response and Toxicity After Pressurized IntraPeritoneal Aerosol Chemotherapy
Department of Visceral Surgery, University Hospital of Lausanne (CHUV), Switzerland.
* equal contribution
Received 2017-6-4; Accepted 2017-10-8; Published 2018-1-1
Abstract
Background: Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) is a novel mode of intraperitoneal (IP) drug delivery claiming high IP tissue concentrations with low systemic uptake. The aim was to study inflammatory response and systemic toxicity after PIPAC.
Methods: Retrospective monocentric analysis of a consecutive cohort of PIPAC patients between January 2015 and April 2016. Detailed hematological and biochemical analysis was performed the day before surgery and once daily until discharge. Comparative statistics were performed using Mann-Whitney U test and Wilcoxon signed ranked test.
Results: Fourty-two consecutive patients underwent a total of 91 PIPAC procedures. Twenty patients received oxaliplatin and 22 cisplatin+doxorubicin (37 vs. 54 procedures). Creatinine, AST and ALT were not significantly altered after PIPAC (p=0.095, p= p=0.153 and p=0.351) and not different between oxaliplatin and cisplatin+doxorubicin regimens (p=0.371, p=0.251 and p=0.288). C-reactive protein (CRP) and procalcitonin (PCT) increased on post-operative day (POD) 2: ∆max 29±5 mg/L (p<0.001) and ∆max 0.05±0.01 μg/L (p=0.005), respectively. Leucocytes increased at POD 1: ∆max 2.2±0.3 G/L (p<0.001). Albumin decreased at POD 2: ∆max -6.0±0.5 g/L (p<0.001). CRP increase correlated positively with Peritoneal Cancer Index (tumor load) (ρ =0.521, p<0.001).
Conclusion: PIPAC was followed by a modest and transitory inflammatory response that was commensurate to the disease extent. No hematological, renal or hepatic toxicity was observed even after repetitive administration.
Keywords: PIPAC, Peritoneal Carcinomatosis, Toxicity, Intraperitoneal Chemotherapy
Introduction
Intraperitoneal (IP) chemotherapy is a treatment alternative for selected patients with peritoneal carcinomatosis (PC) [1, 2]. The rationale for IP treatment is to obtain higher intraperitoneal concentrations with low systemic uptake to reduce systemic toxicity [3, 4]. Intraabdominal complications, hematological but also renal toxicity, and a profound inflammatory response are typical adverse events of both types of conventional treatment, namely hyperthermic IP chemotherapy by lavage (HIPEC) and IP chemotherapy administered by an implantable catheter [5-7].
Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) is a novel minimal-invasive approach for intraperitoneal drug delivery. Administration as an aerosol allows for better distribution within the abdominal cavity, and tissue concentrations of the therapeutic agents are increased under pressure conditions (pneumoperitoneum) despite lower concentrations than for conventional intraperitoneal chemotherapy [1].
The aim of this study was to assess hematological, renal and hepatic toxicity in a consecutive cohort of PIPAC patients and to measure inflammatory response after PIPAC.
Methods
PIPAC was introduced at the University Hospital of Lausanne (CHUV) in January 2015. All patients entered a prospectively maintained coded online data base. Considered for PIPAC treatment were patients with isolated PC of various origins with persistence or progression after standard treatment including surgery and systemic chemotherapy (≥2 lines for most patients). All indications were confirmed during multidisciplinary tumor board. PIPAC treatment alone was preferred and combined intraperitoneal and systemic treatment was exceptional. The present analysis on toxicity and inflammatory response includes all consecutive patients operated until April 2016. Feasibility, postoperative outcomes and quality of life of this cohort were reported recently [8, 9]. The study was approved by the Institutional Review Board (N°2016/0274) and all patients provided written consent for utilization of their data. This study was registered online (www.researchregistry.com #UIN: 2197). STROBE criteria [10] were adhered to for conduction and reporting of the study. The study was conducted in accordance with the principles of the Declaration of Helsinki.
PIPAC procedure and treatment algorithm
Methodology and surgical approach of PIPAC have been reported elsewhere [8, 11]. Briefly, three PIPAC treatments were scheduled at 6-week intervals upon decision of multidisciplinary tumor board. Every patient was seen in outpatient consultation 4 weeks after PIPAC treatment for monitoring of complications and evaluation of contraindications to proceed with PIPAC procedures. Thoraco-abdominal computed tomography (CT) was performed 4 weeks prior to first PIPAC and between PIPAC#2 and PIPAC#3 to rule out extraperitoneal disease and a third CT was scheduled 2 months after completion of 3 PIPAC treatments [8]. Intraperitoneal chemotherapy regimens were defined on individual basis adhering closely to the empiric definitions of the German pioneer group suggesting oxaliplatin monotherapy (92 mg/m2 body surface) for colorectal primary and combined cisplatin (7.5 mg/m2 body surface) and doxorubicin (1.5 mg/m2 body surface) for ovarian and gastric origin [8, 11, 12]. IP treatment was administered for 30 minutes at 37°C and with a standard laparoscopic pressure of 12 mmHg. After treatment, pneumoperitoneum with potential remaining cytostatics was evacuated via a closed system including two microparticle filters [2].
Assessment of postoperative toxicity and inflammation
Blood drawings were performed in a fasting state following standardized institutional guidelines, the day before surgery and once daily until discharge.
Toxicity was assessed by measuring creatinine (Crea) (μmol/L), aspartate aminotransferase (AST, institutional reference (IR): <50 U/L) and alanine aminotransferase (ALT, IR: <50 U/L). Acute renal dysfunction was defined as post-treatment creatinine increase >50% according to Risk, Injury, Failure, Loss of kidney function, and End stage kidney disease (RIFLE) and Acute Kidney Injury Network (AKIN) classification [13, 14].
Inflammatory response was assessed measuring leukocyte (Lc) counts (IR: 4.0-10.0 G/L), C-reactive protein (CRP, IR: <5 g/L), procalcitonin (PCT, IR: <0.06 μg/L) and albumin (Alb, IR: 32-46 g/L). Maximal values were considered for Lc, CRP and PCT. Relevant postoperative inflammation was defined according to literature as Lc >12 G/L, CRP >140 g/L and PCT >0.25 μg/L [15-17]. Maximal difference between the pre- and postoperative values was used for albumin (ΔAlb) and a cut-off value of >10 g/L was used to indicate important inflammatory response [18]. Temperature was measured 4 times per day during the hospitalization.
Predefined clinical questions
Several comparisons and statistical correlations were defined a priori: Evaluation of cumulative toxicity after repetitive PIPAC applications and comparison of systemic inflammation between PIPAC#1, PIPAC#2 and PIPAC#3. Comparison of inflammation and toxicity response between patients treated with oxaliplatin vs. cisplatin and doxorubicin. Correlation between tumor load as assessed by the Peritoneal Cancer Index (PCI) and post-inflammatory response [19].
Statistical analysis
Continuous variables were presented as mean with standard error of the mean (SEM) or median with range or interquartile range (IQR) for skewed data. Categorical variables were reported as frequencies (%) and compared with chi-square test. Wilcoxon signed ranked test, Mann-Whitney test and Kruskal-Wallis test were used for statistical comparisons. Statistical correlations were tested by use of Pearson's rank correlation. All statistical tests were two-sided and a level of 0.05 was used to indicate statistical significance. Statistical analyses were performed and figures were produced with SPSS v20 statistical software (Chicago, IL, USA) and GraphPad Prism 7 (GraphPad Software, Inc., La Jolla, CA, USA).
Results
Patients' characteristics and details of the procedures
The present cohort included 42 consecutive patients who underwent a total of 91 PIPAC procedures, distributed in 2 groups receiving oxaliplatin vs. cisplatin+doxorubicin, respectively. The oxaliplatin group was composed of 20 patients (37 procedures) with colorectal primay (15), small bowel cancer and pseudomyxoma (1 each), and 3 patients with gastric cancer. For the latter group, medical oncologists decided on individual basis to deviate from the empirical protocol (twice due to contraindications). The remaining 22 patients (21 ovarian, 1 mesothelioma; 54 procedures) received cisplatin+doxorubicin according to recommendations. Baseline characteristics are displayed in Table 1.
Baseline demographics of patients treated with Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC)
| Overall (n=42) | Oxaliplatin (n=20) | Cisplatin +Doxorubicin (n=22) | |
|---|---|---|---|
| Demographics | |||
| Age (years) | 66 (59-73) | 62 (53-73) | 68 (63-75) |
| Gender (male) (%) | 8 (19) | 8 (40) | - |
| BMI (kg/m2) | 22.5 (20-25) | 21.5 (19-25) | 22.7 (22-28) |
| Cancer origin | |||
| Colorectal (%) | 15 (36) | 15 (75) | - |
| Gastric (%) | 3 (7) | 3 (15) | - |
| Ovarian (%) | 21 (50) | - | 21 (95) |
| Other (%) | 3 (7) | 2 (10) | 1 (5) |
Median (IQR) for age and BMI. BMI: body mass index.
Surgery typically required 2 trocars for a median duration of 94 (89-108) minutes, whereas median PCI was 10 [8, 9].
Toxicity
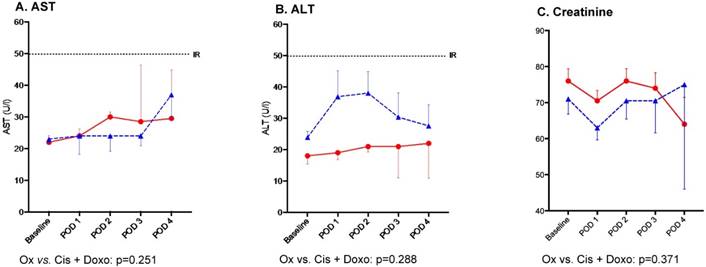
Creatinine level was not significantly altered after PIPAC procedures at post-operative day (POD) 2: ∆max -3±1μmol/L (p=0.095). Based on liver enzymes AST and ALT on POD 2, no hepatotoxicity was caused by PIPAC treatment: ∆max -13±4U/L (p=0.153) and ∆max 4±3U/L (p=0.351) respectively. Differences in hepato-renal impact comparing the two regimens were not significant as detailed in Figure 1. Likewise, no difference was noted when comparing consecutive PIPAC procedures for all three parameters (Figure 2).
Hepato-renal toxicity after Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) treatment by chemotherapy regimen (Oxaliplatin vs. Cisplatin and Doxorubicin) n Oxaliplatin, ▲ Cisplatin + Doxorubicin, Bars represent standard error of the mean (SEM), IR (Institutional Reference, U/L) Hepato-renal toxicity after PIPAC treatment by chemotherapy regimen until postoperative day (POD) 4. AST - aspartate aminotransferase, ALT - alanine aminotransferase.
Hepato-renal toxicity after repetitive Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) procedures. Hepato-renal toxicity under PIPAC treatment represented by AST, ALT and Creatinine levels. Δ represents Δmax. No significant difference (p<0.05) was found for repetitive PIPAC applications. AST - aspartate aminotransferase, ALT - alanine aminotransferase.
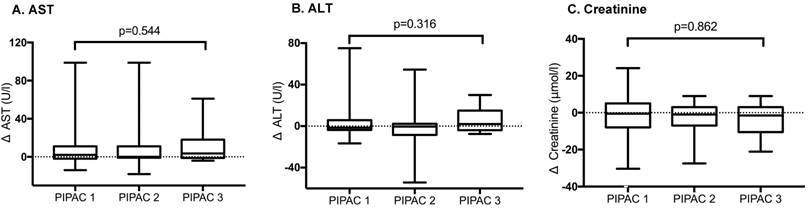
Seven (17%) and 6 (14%) patients surpassed institutional thresholds for AST and ALT, respectively. There was no lasting effect. No patient presented acute kidney injury according to RIFLE criteria or AKIN classification.
Inflammatory response
CRP and PCT increased on POD 2: ∆max 29±5 mg/L (p<0.001) and ∆max 0.05±0.01 μg/L (p=0.005), respectively. Leucocytes increased at POD 1: ∆max 2.2±0.3 G/L (p<0.001). Albumin decreased at POD 2: ∆max -6.0±0.5 g/L (p<0.001).
The profiles of inflammatory markers displayed by chemotherapy regimen are illustrated in Figure 3. For both regimens CRP and Lc count were characterized by an increase on POD 2 and POD 1 respectively (Figures 3A and 3C). Conversely, albumin level decreased by POD 2 (Figures 3D). Both regimens influenced markers' kinetics similarly except for PCT (where oxaliplatin induced a higher increase than cisplatin+doxorubicin) (Figure 3C).
Inflammatory response after Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) by chemotherapy regimen. n Oxaliplatin ▲Cisplatin +Doxorubicin. Inflammatory response under PIPAC treatment by chemotherapy regimen until postoperative day (POD) 4. Bars represent standard error of the mean (SEM).
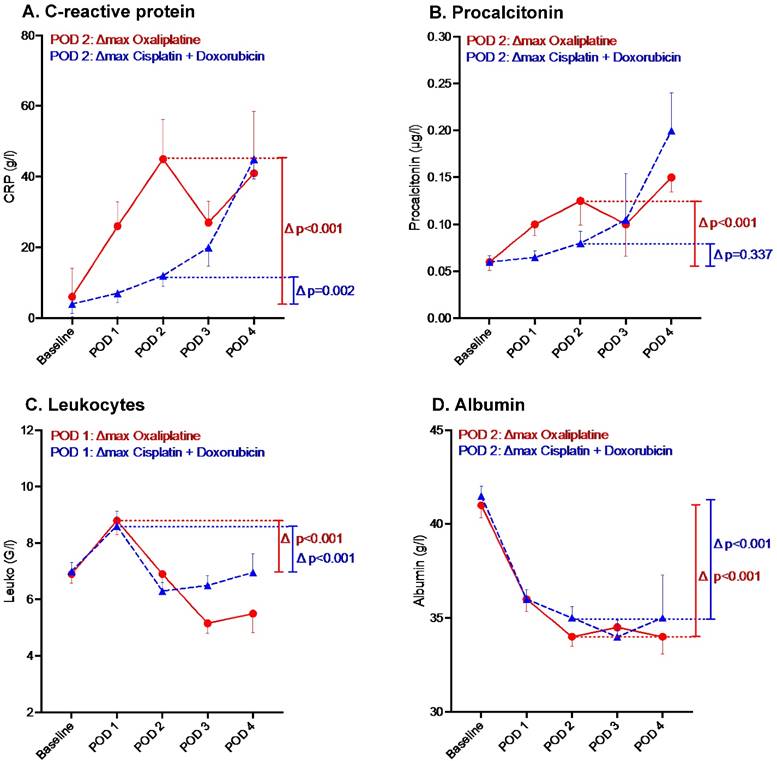
Of note, one patient (2%) presented leukopenia after PIPAC 1 with spontaneous resolution within 2 days. One patient had fever >38° after PIPAC #3 also with spontaneous resolution.
In 17 patients (40%), relevant postoperative inflammation as defined under methods was observed. Ten patients (24%) showed increased Lc count above 12G/L, 2 patients (5%) increased CRP level above 140g/L and 3 patients (7%) PCT level above 0.25 μg/L. Only one patient (2%) had concomitant relevant increase of Lc and CRP levels and another patient (2%) of Lc and PCT levels.
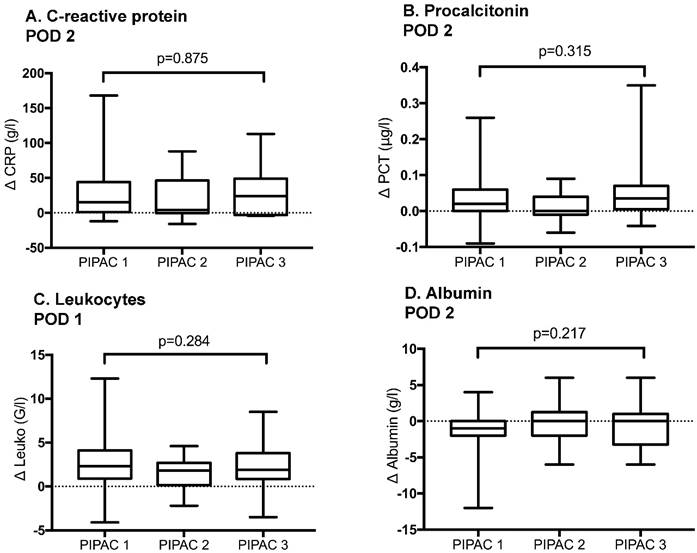
The extent of inflammatory response after consecutive PIPAC procedures was similar with no significant difference for any of the candidate markers (Figure 4).
Inflammatory response after repetitive Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) procedures. Inflammatory response under PIPAC treatment represented by C-reactive protein at post-operative day (POD) 2, Procalcitonin at POD 2, Leukocytes at POD 1 and Albumin at POD 2. Bars represent standard error of the mean (SEM). Δ represents Δmax. No significant difference was found when inflammatory response was compared before and after different PIPAC applications.
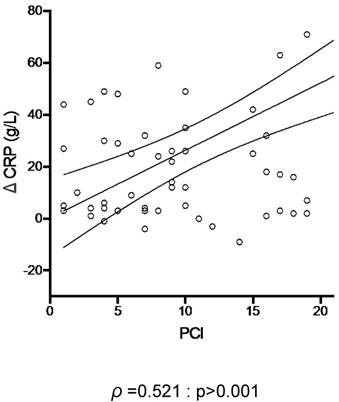
Using CRP and PCI as respective surrogate markers of inflammation and tumor burden, the inflammatory response induced by PIPAC correlated positively with tumor load (ρ =0.521, p>0.001) (Figure 5).
Tumor load and post- Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) inflammation. C-reactive protein (CRP) was plotted against the extent of peritoneal disease (measured by the Peritoneal Cancer Index (PCI)).
Discussion
In this cohort, PIPAC was followed by a modest and transitory inflammatory response that was commensurate to the disease extent. No hematological, renal or hepatic toxicity was observed even after repetitive administration.
The findings of the present study need to be discussed in the setting of four available treatment options for advanced peritoneal carcinomatosis: systemic chemotherapy, hyperthermic intraperitoneal chemotherapy (HIPEC) after cytoreductive surgery (CRS), intraperitoneal catheter chemotherapy and PIPAC (Table 2). Of note, it is not the intention to formally compare those treatment approaches due to their very different nature but to provide a comprehensive overview on what is known on toxicity and inflammatory response under the respective treatment.
Synopsis of systemic toxicity and inflammation after different treatments for peritoneal carcinomatosis
| Toxicity Renal | Hepatic | Hematologic | Other | Inflammatory Reaction | Drug Concentration | |
|---|---|---|---|---|---|---|
| Systemic chemo | Creatinin increase:5-10%* | AST/ALT increase: 36-54%* | Neutropenia: 42% [40] Thrombopenia: 3-4% [41] (III-IV) | Neurotoxicity up to 90% [42] (dose-dependent) | Fever 25% [40] | Oxaliplatin: 85 mg/m2 |
HIPEC | Creatinin increase: 18% [43] Nephro-toxicity: 6% [43] (III-V)* | Hepato-toxicity** 36% (III) 50% (II) [44] | Neutropenia: 27% [45] ** 40% [46] *** | Surgical morbidity: 34% (III+IV) 4% mortality [5] | CRP at 48h: 180mg/L [47] | *Cisplatin 75 mg/m2 +/-Mitomycin-C 10 mg/m2 **Oxaliplatin 460 mg/m2 ***Mitomycin-C 0.8 mg/kg |
IP catheter | Nephro-toxicity 7% [32] (III-IV) | Hepato-toxicity 3% [32] (III-IV) | Leukopenia: 40-76% [32, 33] Thrombopenia: 8-12% [32, 33] Anemia: 26% [33] | Neurologic events: 19% [32] (III-IV) | Infection: 16% [32] (III-IV) | Cisplatin 100 mg/m2 +/- Paclitaxel 60 mg/m2 |
PIPAC | None | TransitorygGT increase [36] AST/ALT increase: 14-16% | None | Surgical morbidity: 9% (mainly I+II) [8] | CRP at 48h: 45mg/L* CRP at 48h 15mg/L** | *Oxaliplatin 92 mg/m2 **Cisplatin 7.5 mg/m2 + Doxorubicin 1.5 mg/m2 |
Chemo - chemotherapy, HIPEC - Hyperthermic Intraperitoneal Chemotherapy, IP - intraperitoneal, PIPAC - Pressurized Intraperitoneal Aerosol Chemotherapy, AST - aspartate aminotransferase, ALT - alanine aminotransferase, CRP - C-Reactive Protein, gGT - gamma Glutamyltransferase Severity grades of complications are displayed in brackets.
Systemic chemotherapy represents a first line therapy in patients with peritoneal carcinomatosis, especially with extraperitoneal spread of tumor [20]. New drugs such as antibodies or irinotecan allowed for better survival in recent years, but pharmacokinetic limitations such as peritoneum-plasma barrier impede proper penetration of cytostatics in peritoneal tumor nodes [21, 22]. Parenteral drug concentrations need thus to be high to achieve a therapeutic effect. Up to tenfold higher concentrations were described for systemic chemotherapy as compared to intraperitoneal use, and side effects for most therapies are common [4]. Chemotherapy induced peripheral neuropathy represents a well-known side effect of platin-based chemotherapy regimens [23]. Up to 90 % of patients are affected after systemic oxaliplatin treatment, and treatment options remain scarce [24, 25]. Cisplatin is eliminated by the kidney, and dose-dependent renal toxicity of cisplatin and its metabolites has been described [26]. Standard systemic doses of 75-100 mg/m2 lead to tubular necrosis in up to 30% of patients, and renal failure might be the ultimate consequence [26]. Taken together and in the light of pharmacokinetic limitations of systemic therapy for the treatment of peritoneal carcinomatosis, alternative locoregional treatments were considered.
Cytoreductive surgery combined with HIPEC represents the only potentially curative treatment option in few selected patients who are fit enough to tolerate this complex treatment option with important morbi-mortality [5, 27]. Intraperitoneal administration permits higher drug concentration with better tumor penetration as compared to systemic chemotherapy [28, 29]. Even if no formal consensus exists, common drugs for HIPEC are cisplatin, mitomycin C and oxaliplatin in different combinations and doses, depending on the primary tumor [27]. In high volume centers, mortality rates of up to 5.8%, major complication rates between 12-52% and hematological toxicity of up to 28% of patients were described using these regimens [27]. Irinotecan was added in more recent years; neutropenia and thrombopenia at the 7th postoperative day occurred in 11% of patients in a cohort treated with a combination of Irinotecan and Oxaliplatin [30]. In a recent study using the same regimen, hematologic toxicity was even higher with 41% neutropenic (<500/μl) patients and 26% with grade 3 thrombopenia (<50000/μl) [31]. PIPAC as a minimally invasive treatment option might thus represent an alternative for frail patients who are not eligible for this curative approach given the multitude of surgery- and drug-related side effects.
Repetitive administration through intraperitoneal catheters in an adjuvant setting after optimally debulked stage III ovarian cancer represents an alternative to HIPEC [32]. Intraperitoneal administration of paclitaxel and cisplatin combined with intravenous paclitaxel significantly improved survival compared to patients receiving the same drugs exclusively intravenously [32]. However, patients in the intraperitoneal group experienced more pain and presented more hematologic, gastrointestinal and neurologic toxic effects (Table 2). A former study compared intraperitoneal cisplatin to intravenous cisplatin to demonstrate improved survival and fewer toxic effects in the intraperitoneal group [33].
PIPAC combines the advantages of intraperitoneal administration, allowing for higher tumor concentrations [28] and administration under pressure, which permits higher intra-tumoral concentrations despite lower drug doses [3]. In a pilot study of the pioneer group from Herne, Germany, peripheral drug concentrations of doxorubicin were very low with 4.0-6.2 ng/ml after PIPAC treatment within standard conditions [1]. As a consequence, only modest and transitory inflammatory response was observed in former studies and in the present cohort [34-36]. This response might be a consequence of drug-induced chemical peritonitis after intraperitoneal vaporisation, which provides an explanation for abdominal pain as main postoperative complaint after PIPAC [34]. Two centers reported on toxicological aspects of PIPAC treatment [35, 36]. Similar to the present findings, Blanco described liver and renal parameters within the normal range with no cumulative toxicity [35]. Similarly, an Italian group described no clinically relevant liver cytolysis without metabolic nor synthetic hepatic and renal dysfunction [36]. Both groups concluded that PIPAC caused less hepatic and renal toxicity than other chemotherapy delivery routes due to lower therapeutic doses and favorable kinetics [35, 36]. The present findings confirm the previously reported results. As a consequence of low toxicity, encouraging results regarding quality of life and tolerance of the procedure have been described by our group and by others [8, 9, 37].
Several limitations of the present study need to be discussed beyond retrospective study design and small patient cohort. Given the very short median hospital stay, only few patients were available for blood drawings after POD 3 and inflammatory parameters beyond POD 3 should therefore not be considered representative for the entire PIPAC cohort. Systemic toxicity was low in the present study, which might be due to low administered drug concentrations. The proposed regimens did not derive from a dose escalation protocol and hence, drug choices and doses rely on empirical protocols. Three dose escalation studies however have been initiated in the meantime (NCT02475772, NCT03172416 and Eudra-CT 2016-003666-49). Since PIPAC is a very new technique, no long-term results are available to date. Despite favorable short-term results regarding clinical and histological response rates [34, 38, 39], sustained long-term impact of PIPAC under presently applied conditions needs yet to be proven.
In conclusion, there was no relevant systemic toxicity after PIPAC with the current treatment standard even when repeatedly applied. Inflammatory response was modest and transitory. It is important to repeat this type of studies for new treatment standards, especially when using higher drug concentrations.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Solass W, Kerb R, Murdter T, Giger-Pabst U, Strumberg D, Tempfer C, Zieren J, Schwab M, Reymond MA. Intraperitoneal chemotherapy of peritoneal carcinomatosis using pressurized aerosol as an alternative to liquid solution: first evidence for efficacy. Annals of surgical oncology. 2014;21(2):553-559
2. Solass W, Hetzel A, Nadiradze G, Sagynaliev E, Reymond MA. Description of a novel approach for intraperitoneal drug delivery and the related device. Surgical endoscopy. 2012;26(7):1849-1855
3. Facy O, Al Samman S, Magnin G, Ghiringhelli F, Ladoire S, Chauffert B, Rat P, Ortega-Deballon P. High pressure enhances the effect of hyperthermia in intraperitoneal chemotherapy with oxaliplatin: an experimental study. Annals of surgery. 2012;256(6):1084-1088
4. Esquis P, Consolo D, Magnin G, Pointaire P, Moretto P, Ynsa MD, Beltramo JL, Drogoul C, Simonet M, Benoit L. et al. High intra-abdominal pressure enhances the penetration and antitumor effect of intraperitoneal cisplatin on experimental peritoneal carcinomatosis. Annals of surgery. 2006;244(1):106-112
5. Glehen O, Gilly FN, Boutitie F, Bereder JM, Quenet F, Sideris L, Mansvelt B, Lorimier G, Msika S, Elias D. et al. Toward curative treatment of peritoneal carcinomatosis from nonovarian origin by cytoreductive surgery combined with perioperative intraperitoneal chemotherapy: a multi-institutional study of 1,290 patients. Cancer. 2010;116(24):5608-5618
6. Verwaal VJ. Long-term results of cytoreduction and HIPEC followed by systemic chemotherapy. Cancer journal. 2009;15(3):212-215
7. Ceelen WP, Pahlman L, Mahteme H. Pharmacodynamic aspects of intraperitoneal cytotoxic therapy. Cancer Treat Res. 2007;134:195-214
8. Hubner M, Teixeira Farinha H, Grass F, Wolfer A, Mathevet P, Hahnloser D, Demartines N. Feasibility and Safety of Pressurized Intraperitoneal Aerosol Chemotherapy for Peritoneal Carcinomatosis: A Retrospective Cohort Study. Gastroenterol Res Pract. 2017;2017:6852749
9. Teixeira Farinha H, Grass F, Kefleyesus A, Achtari C, Romain B, Montemurro M, Demartines N, Hubner M. Impact of Pressurized Intraperitoneal Aerosol Chemotherapy on Quality of Life and Symptoms in Patients with Peritoneal Carcinomatosis: A Retrospective Cohort Study. Gastroenterol Res Pract. 2017;2017:4596176
10. Vandenbroucke JP, von Elm E, Altman DG, Gotzsche PC, Mulrow CD, Pocock SJ, Poole C, Schlesselman JJ, Egger M, Initiative S. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): explanation and elaboration. Epidemiology. 2007;18(6):805-835
11. Solass W, Giger-Pabst U, Zieren J, Reymond MA. Pressurized intraperitoneal aerosol chemotherapy (PIPAC): occupational health and safety aspects. Annals of surgical oncology. 2013;20(11):3504-3511
12. Hubner M, Grass F, Teixeira-Farinha H, Pache B, Mathevet P, Demartines N. Pressurized IntraPeritoneal Aerosol Chemotherapy - Practical aspects. European journal of surgical oncology: the journal of the European Society of Surgical Oncology and the British Association of Surgical Oncology. 2017;43(6):1102-1109
13. Kellum JA, Levin N, Bouman C, Lameire N. Developing a consensus classification system for acute renal failure. Curr Opin Crit Care. 2002;8(6):509-514
14. Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P, Acute Dialysis Quality Initiative w. Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Critical care. 2004;8(4):R204-212
15. Meisner M. Update on procalcitonin measurements. Ann Lab Med. 2014;34(4):263-273
16. Almeida AB, Faria G, Moreira H, Pinto-de-Sousa J, Correia-da-Silva P, Maia JC. Elevated serum C-reactive protein as a predictive factor for anastomotic leakage in colorectal surgery. Int J Surg. 2012;10(2):87-91
17. Pedersen T, Roikjaer O, Jess P. Increased levels of C-reactive protein and leukocyte count are poor predictors of anastomotic leakage following laparoscopic colorectal resection. Dan Med J. 2012;59(12):A4552
18. Hubner M, Mantziari S, Demartines N, Pralong F, Coti-Bertrand P, Schafer M. Postoperative Albumin Drop Is a Marker for Surgical Stress and a Predictor for Clinical Outcome: A Pilot Study. Gastroenterol Res Pract. 2016;2016:8743187
19. Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, Carbone PP. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol. 1982;5(6):649-655
20. Franko J, Shi Q, Goldman CD, Pockaj BA, Nelson GD, Goldberg RM, Pitot HC, Grothey A, Alberts SR, Sargent DJ. Treatment of colorectal peritoneal carcinomatosis with systemic chemotherapy: a pooled analysis of north central cancer treatment group phase III trials N9741 and N9841. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2012;30(3):263-267
21. Franko J, Ibrahim Z, Gusani NJ, Holtzman MP, Bartlett DL, Zeh HJ 3rd. Cytoreductive surgery and hyperthermic intraperitoneal chemoperfusion versus systemic chemotherapy alone for colorectal peritoneal carcinomatosis. Cancer. 2010;116(16):3756-3762
22. Flessner MF. The transport barrier in intraperitoneal therapy. Am J Physiol Renal Physiol. 2005;288(3):F433-442
23. Poupon L, Kerckhove N, Vein J, Lamoine S, Authier N, Busserolles J, Balayssac D. Minimizing chemotherapy-induced peripheral neuropathy: preclinical and clinical development of new perspectives. Expert Opin Drug Saf. 2015;14(8):1269-1282
24. Hershman DL, Lacchetti C, Dworkin RH, Lavoie Smith EM, Bleeker J, Cavaletti G, Chauhan C, Gavin P, Lavino A, Lustberg MB. et al. Prevention and management of chemotherapy-induced peripheral neuropathy in survivors of adult cancers: American Society of Clinical Oncology clinical practice guideline. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2014;32(18):1941-1967
25. Balayssac D, Ferrier J, Descoeur J, Ling B, Pezet D, Eschalier A, Authier N. Chemotherapy-induced peripheral neuropathies: from clinical relevance to preclinical evidence. Expert Opin Drug Saf. 2011;10(3):407-417
26. dos Santos NA, Carvalho Rodrigues MA, Martins NM, dos Santos AC. Cisplatin-induced nephrotoxicity and targets of nephroprotection: an update. Arch Toxicol. 2012;86(8):1233-1250
27. Chua TC, Yan TD, Saxena A, Morris DL. Should the treatment of peritoneal carcinomatosis by cytoreductive surgery and hyperthermic intraperitoneal chemotherapy still be regarded as a highly morbid procedure? a systematic review of morbidity and mortality. Annals of surgery. 2009;249(6):900-907
28. Solass W, Herbette A, Schwarz T, Hetzel A, Sun JS, Dutreix M, Reymond MA. Therapeutic approach of human peritoneal carcinomatosis with Dbait in combination with capnoperitoneum: proof of concept. Surgical endoscopy. 2012;26(3):847-852
29. Ceelen WP, Hesse U, de Hemptinne B, Pattyn P. Hyperthermic intraperitoneal chemoperfusion in the treatment of locally advanced intra-abdominal cancer. Br J Surg. 2000;87(8):1006-1015
30. Elias D, Goere D, Blot F, Billard V, Pocard M, Kohneh-Shahri N, Raynard B. Optimization of hyperthermic intraperitoneal chemotherapy with oxaliplatin plus irinotecan at 43 degrees C after compete cytoreductive surgery: mortality and morbidity in 106 consecutive patients. Annals of surgical oncology. 2007;14(6):1818-1824
31. Quenet F, Goere D, Mehta SS, Roca L, Dumont F, Hessissen M, Saint-Aubert B, Elias D. Results of two bi-institutional prospective studies using intraperitoneal oxaliplatin with or without irinotecan during HIPEC after cytoreductive surgery for colorectal carcinomatosis. Annals of surgery. 2011;254(2):294-301
32. Armstrong DK, Bundy B, Wenzel L, Huang HQ, Baergen R, Lele S, Copeland LJ, Walker JL, Burger RA, Gynecologic Oncology G. Intraperitoneal cisplatin and paclitaxel in ovarian cancer. N Engl J Med. 2006;354(1):34-43
33. Alberts DS, Liu PY, Hannigan EV, O'Toole R, Williams SD, Young JA, Franklin EW, Clarke-Pearson DL, Malviya VK, DuBeshter B. Intraperitoneal cisplatin plus intravenous cyclophosphamide versus intravenous cisplatin plus intravenous cyclophosphamide for stage III ovarian cancer. N Engl J Med. 1996;335(26):1950-1955
34. Tempfer CB, Winnekendonk G, Solass W, Horvat R, Giger-Pabst U, Zieren J, Rezniczek GA, Reymond MA. Pressurized intraperitoneal aerosol chemotherapy in women with recurrent ovarian cancer: A phase 2 study. Gynecologic oncology. 2015;137(2):223-228
35. Blanco A, Giger-Pabst U, Solass W, Zieren J, Reymond MA. Renal and hepatic toxicities after pressurized intraperitoneal aerosol chemotherapy (PIPAC). Annals of surgical oncology. 2013;20(7):2311-2316
36. Robella M, Vaira M, De Simone M. Safety and feasibility of pressurized intraperitoneal aerosol chemotherapy (PIPAC) associated with systemic chemotherapy: an innovative approach to treat peritoneal carcinomatosis. World journal of surgical oncology. 2016;14:128
37. Odendahl K, Solass W, Demtroder C, Giger-Pabst U, Zieren J, Tempfer C, Reymond MA. Quality of life of patients with end-stage peritoneal metastasis treated with Pressurized IntraPeritoneal Aerosol Chemotherapy (PIPAC). European journal of surgical oncology: the journal of the European Society of Surgical Oncology and the British Association of Surgical Oncology. 2015;41(10):1379-1385
38. Nadiradze G, Giger-Pabst U, Zieren J, Strumberg D, Solass W, Reymond MA. Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) with Low-Dose Cisplatin and Doxorubicin in Gastric Peritoneal Metastasis. Journal of gastrointestinal surgery: official journal of the Society for Surgery of the Alimentary Tract. 2016;20(2):367-373
39. Demtroder C, Solass W, Zieren J, Strumberg D, Giger-Pabst U, Reymond MA. Pressurized intraperitoneal aerosol chemotherapy with oxaliplatin in colorectal peritoneal metastasis. Colorectal disease: the official journal of the Association of Coloproctology of Great Britain and Ireland. 2016;18(4):364-371
40. Haller DG. Safety of oxaliplatin in the treatment of colorectal cancer. Oncology (Williston Park). 2000;14(12 Suppl 11):15-20
41. Jardim DL, Rodrigues CA, Novis YA, Rocha VG, Hoff PM. Oxaliplatin-related thrombocytopenia. Annals of oncology: official journal of the European Society for Medical Oncology. 2012;23(8):1937-1942
42. Pachman DR, Qin R, Seisler DK, Smith EM, Beutler AS, Ta LE, Lafky JM, Wagner-Johnston ND, Ruddy KJ, Dakhil S. et al. Clinical Course of Oxaliplatin-Induced Neuropathy: Results From the Randomized Phase III Trial N08CB (Alliance). Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2015;33(30):3416-3422
43. Canda AE, Sokmen S, Terzi C, Arslan C, Oztop I, Karabulut B, Ozzeybek D, Sarioglu S, Fuzun M. Complications and toxicities after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Annals of surgical oncology. 2013;20(4):1082-1087
44. Ceelen WP, Peeters M, Houtmeyers P, Breusegem C, De Somer F, Pattyn P. Safety and efficacy of hyperthermic intraperitoneal chemoperfusion with high-dose oxaliplatin in patients with peritoneal carcinomatosis. Annals of surgical oncology. 2008;15(2):535-541
45. Hompes D, D'Hoore A, Wolthuis A, Fieuws S, Mirck B, Bruin S, Verwaal V. The use of Oxaliplatin or Mitomycin C in HIPEC treatment for peritoneal carcinomatosis from colorectal cancer: a comparative study. Journal of surgical oncology. 2014;109(6):527-532
46. Kemmel V, Mercoli HA, Meyer N, Brumaru D, Romain B, Lessinger JM, Brigand C. Mitomycin C Pharmacokinetics as Predictor of Severe Neutropenia in Hyperthermic Intraperitoneal Therapy. Annals of surgical oncology. 2015;22(Suppl 3):S873-879
47. Medina Fernandez FJ, Munoz-Casares FC, Arjona-Sanchez A, Casado-Adam A, Gomez-Luque I, Garcilazo Arismendi DJ, Thoelecke H, Rufian Pena S, Briceno Delgado J. Postoperative time course and utility of inflammatory markers in patients with ovarian peritoneal carcinomatosis treated with neoadjuvant chemotherapy, cytoreductive surgery, and HIPEC. Annals of surgical oncology. 2015;22(4):1332-1340
Author contact
Corresponding author: Nicolas Demartines, Department of Visceral Surgery, Lausanne University Hospital CHUV, 1011 Lausanne, Switzerland. Phone : +41 21 314 24 00; Fax : +41 21 314 24 11; E-mail : martin.hubnerch

 Global reach, higher impact
Global reach, higher impact