Impact Factor ISSN: 1837-9664
J Cancer 2017; 8(2):227-239. doi:10.7150/jca.17093 This issue Cite
Research Paper
Aberrant Signaling through the HER2-ERK1/2 Pathway is Predictive of Reduced Disease-Free and Overall Survival in Early Stage Non-Small Cell Lung Cancer (NSCLC) Patients
1. Biogem scarl, Institute of Genetic Research, Ariano Irpino (AV), Italy.
2. Department of Experimental and Clinical Medicine, University “Magna Graecia”, Catanzaro, Italy.
3. Pathology Unit, Istituto Nazionale Tumori “Fondazione G. Pascale”, IRCCS, Naples, Italy.
4. Hospital “Monaldi-Cotugno-CTO”, Naples, Italy.
5. Department of Medical and Surgical Sciences, University “Magna Graecia” Medical School, Catanzaro, Italy
6. Pathology Unit, Second University of Naples, Italy.
Received 2016-8-3; Accepted 2016-10-17; Published 2017-1-15
Abstract
Background: Purpose of this study was to evaluate the contribution of the Extracellular-regulated protein kinase (ERK)-1/2 pathway to oncogenic signaling elicited by the tyrosine kinase receptor HER2 in Non-Small Cell Lung Cancer (NSCLC) and to assess the prognostic value of these oncoproteins in NSCLC patients.
Methods: Immunohistochemistry was performed to determine expression and activation of HER2 and ERK1/2 (detected by phosphorylation of Y1248 and T202/Y204, respectively) using Tissue Micro Arrays (TMA) containing matched normal and neoplastic tissues from 132 NSCLC patients. Survival analysis was carried out using the Kaplan-Meier method. Univariate and multivariate analysis were used to evaluate the prognostic value of pERK1/2, pHER2 and a combination thereof with clinical-pathological parameters such as age, lymph node status (N), size (T), stage (TNM) and grade.
Results: We found that HER2 was overexpressed in 33/120 (27%) and activated in 41/114 (36%) cases; ERK1/2 was activated in 44/102 (43%) cases. A direct association was found between pERK1/2 and pHER2 (23/41; p=0.038). In addition, patients positive for pERK1/2 and for both pHER2 and pERK1/2 showed significantly worse overall survival (OS) and disease-free survival (DFS) compared with negative patients. Univariate and multivariate analysis of patients' survival revealed that positivity for pHER2-pERK1/2 and for pERK1/2 alone were independent prognostic factors of poor survival in NSCLC patients. In particular, this association was significantly important for DFS in stage I+II patients.
Conclusion: This study provides evidence that activated ERK1/2 and/or the combined activation of HER2 and ERK1/2 are good indicators of poor prognosis in NSCLC patients, not only in unselected patients but also in early stage disease.
Keywords: NSCLC, HER2, ERK1/2, Tissue Micro Array, survival.
Introduction
Lung carcinoma is the most common form of cancer that accounts for approximately 18% of all cancer deaths. Non-small cell lung cancer (NSCLC) represents 80-85% of all cases [1]. Despite major diagnostic and therapeutic improvements most NSCLC patients die within few years from diagnosis [2] because of the lack of an effective screening strategy that helps to identify lung cancer at an early stage [3]. Patients with Stage I or II NSCLC have 70% five-year survival after surgery alone whereas advanced patients (stages IIIB and IV) typically show <5% five-year survival [3], though several patients with stage I and/or II disease eventually recur. The absence of reliable predictors of recurrence has hampered the identification of the patients with poor prognosis.
In recent years, several studies have demonstrated that NSCLC develops as the consequence of the accumulation of somatically acquired genetic defects [1, 2, 4-7]. These include mutations, amplifications and/or rearrangements in the genes encoding epidermal growth factor receptor (EGFR), HER2, KRAS, BRAF, PIK3CA, AKT1, DDR2, ALK and, more recently, ROS and RET [6-12]. These findings have allowed a better understanding of the molecular origins of the disease and have contributed to improve therapeutic treatment of NSCLC patients [5-7].
The HER2 proto-oncogene encodes a trans-membrane tyrosine kinase receptor with marked sequence homology with the Epidermal Growth Factor Receptor (EGFR). HER2 transduces signals leading to cell proliferation, motility and survival [6, 13]. HER2 is mutated, amplified and/or overexpressed in many epithelial cancers including NSCLC [13, 14]. Mutations in the HER2 kinase domain have been reported in lung adenocarcinomas at a relatively low frequency (2-4%) [15, 16]. The majority of the HER2 mutations identified in NSCLC samples are in-frame duplications or insertions in exon 20 [8, 9, 17]. On the other hand, HER2 overexpression, observed in 7-32% of cases, appears to be a poor prognostic factor for NSCLC patients [18, 19].
Constitutively activated HER2 signals in the absence of ligand through phosphorylation of Y1248/Y1253 residues, which are necessary for the activation of downstream RAF-MEK-ERK1/2 and PI3K-AKT pathways [20-27]. Once dysregulated, these cascades play a major role in cancer development [4, 28-32]. Direct evidence of the importance of simultaneous signaling from the PI3K-AKT and RAF-MEK-ERK1/2 pathways in cancer cells derives from the findings that both pathways are activated in NSCLC cells and that the combined use of MEK and PI3K inhibitors is apparently necessary for optimal anti-tumor activity [33, 34]. The combined use of MEK and PI3K inhibitors enhances anti-proliferative activity in vitro [35] and efficiently inhibits the growth of EGFR- or KRAS-induced lung tumors in mice [36, 37].
The purpose of this study was to evaluate the relative contribution of downstream pathways to the deregulated signaling elicited by oncogenic tyrosine kinase receptor HER2 in NSCLC and to evaluate the activation status of these pathways as predictive factors in NSCLC patients.
We provide evidence that activated HER2 is associated to increased signaling through the ERK1/2 pathway, and that the combined pHER2-pERK1/2 positivity predict poor prognosis in NSCLC patients.
Materials and Methods
Ethics Statement
Patient accrual was conducted according to internal Review Board of the INT Fondazione Pascale (Naples, Italy) (CEI 556/10 of 12/3/2010) and approved by the internal Review Board of AOU Mater Domini/University Magna Graecia (Catanzaro, Italy) on 16/3/2011. Written informed consent was obtained from all participants to the study.
Patients
Archive material from 132 patients diagnosed of NSCLC [38, 39] was obtained from INT Fondazione Pascale (Naples, Italy) and Azienda Ospedaliera “Monaldi-Cotugno-CTO” (Naples, Italy). Median age was 64 years (range 28-82). Women were 28 and males were 104. Stage was known for 130 patients: 73 patients had stage IA-IB disease, 34 patients had IIA-IIB and 23 patients had stage III-IV disease [40]. Grade was known for 125 patients: 72 cases were G1-G2, 53 cases were G3. See Supplemental Table 1 for more detailed clinical characteristics of patients. All cases were classified according to the new IASLC/ATS/ERS classification and graded based on the Sica scoring system [41]. The data for 3-year follow-up was available for 77 patients.
Tissue Microarray (TMA) and Immunohistochemistry
TMAs named TMA1 and TMA2 were constructed using Galileo TMA CK3500 Tissue Microarrayer (ISE TMA Software, Integrated System Engineering). Immunostaining was performed using Peroxidase Detection Systems (RE7110-K; Novocastra, United Kingdom).
Antibodies used for immunostaining were selected according to previously published work [42]. Anti-phospho-p44/42 ERK1/2 T202/Y204 (#4376, 1:100 dilution), anti-p44/42 ERK1/2 (#9102, 1:50 dilution), anti-HER2 (#4290, 1:100 dilution), anti-pAKT Ser473 (#3787, 1:100 dilution) were purchased from Cell Signaling Technology Inc. (Danvers, MA, USA). Anti-phospho HER2 Y1248 (#NB100-81960) was from Novus Biologicals [43].
For immunohistochemical evaluation of HER2, tumors were scored as previously described [44]. In brief, scores were assigned as follows: score 0, if staining was detected in <10% of tumor cells; score 1+, if tumors showed incomplete and faint staining in >10% of tumor cells; score 2+, if tumors showed weak-to-moderate membrane staining in >10% of tumor cells; score 3+, if tumors showed strong membrane immunoreactivity in >10% neoplastic cells. Tumors classified as 0 or 1+ were considered “negative” and those scored as 2+ or 3+ were classified as “positive”.
Phosphorylated HER2 (Tyr 1248) was scored as previously described [45]: 0, if tumor cells had complete absence or faint staining in <10%; 1+, if <10% of tumor cells had faint staining; 2+, if tumor cells had moderate staining in >10% of cells; 3+, if tumor cells had strong staining in >10% of cells.
For immunohistochemical evaluation of ERK1/2 (Thr202/Tyr204) phosphorylation the cut-off value was set at 15% as previously described [42]. On this basis tumors were scored as follows: pERK1/2-positive, when staining was observed in >15% of tumor cells; pERK1/2-negative, when staining observed in <15% of tumor cells.
Prior to each experiment we set up the experimental procedures for the use of the primary antibody by use of a control tissue as positive control, selected among those indicated in the datasheets provided by the furnisher. Once established the procedures, we proceeded to perform TMA staining for the specific antibody. For each round of immunostaining we performed positive and negative (primary antibody omission control) controls. All controls gave satisfactory results. Stained TMA sections were evaluated by two expert pathologists (RF, GB) using uniform criteria. Discrepancies between two cores from the same case were resolved through a joint analysis of the two cores.
Fluorescence In Situ Hybridization (FISH)
Interphase FISH analysis was performed on TMAs. TMA slides were deparaffinized after being pre-treated with Paraffin Pretreatment Kit (Vysis, Downers Grove, IL, USA) using protease to digest proteins for 10 minutes. Slides were then dehydrated in increasing ethanol concentrations and then air-dried. Denaturation of the probe mixture was performed at 95°C for 5 minutes in Thermobrite (Abbott Molecular Inc. Des Plaines, IL, USA) followed by overnight hybridization at 37°C. The following day, cover glass was removed and slides were washed in post-hybridization washing-buffer at 72°C for 2 minutes and then counterstained with DAPI counterstain and a coverslip was applied. Automated scanning and scoring of interphase FISH was performed by Cytovision System (CytoVision® Version 7.3, Leica Biosystems, Germany).
The copy number of the HER2 locus was determined by using two different probes: a red probe covering the gene and a green control probe covering the centromeric region of chromosome 17 (PathVysion HER-2 DNA Probe Kit, Abbott Molecular Inc. Des Plaines, IL, USA). Two different investigators (MS, RF) that had no previous knowledge of the genetic, clinical and IHC results evaluated FISH analysis.
According to the frequency of tumor cells with specific number of copies of the gene and the chromosome centromere, tumors were initially classified into six FISH categories (disomy, low and high trisomy, low and high polysomy, gene amplification) and finally divided into two groups: (a) FISH negative, including disomic, trisomic and low polysomic (≥4 copies of the gene in <40% of cells) tumors; and (b) FISH positive, which include tumors with high polysomic (≥4 copies in ≥40% of cells) and/or gene amplification (a ratio gene/chromosome/cell ≥2, presence of clusters of the HER2 gene signal in ≥10% of the analyzed cells) [46, 47].
Statistical analysis
The correlation between biomarkers was determined using Fisher's exact test. Relationships between clinical-pathological parameters were assessed using contingency tables with a Fisher's exact test. Survival curves were estimated using the Kaplan-Meier method and compared using the log rank test. Overall survival (OS) was calculated from the day of surgery to the day of death or to the end of follow-up. Disease-free survival (DFS) was calculated from the day of surgery to the documented day of disease progression (relapse or metastasis) or to the end of follow-up. Univariate and multivariate survival analyses with calculation of hazard ratios (HR) were performed using Cox's proportional-hazards model. All statistical analyses were performed using the SPSS software system (version 17.0; SPSS. Inc., Chicago, IL, USA). A p-value ≤ 0.05 was considered to be statistically significant.
Results
Activation of HER2 in NSCLC
We performed a comprehensive study aimed at determining the relative contribution of MAPK-dependent signaling to the activity of HER2 in NSCLC. To this aim, we determined the activation status of HER2, ERK1/2 and a combination thereof and correlated them with the clinical-pathological characteristics of patients. The TMAs used in this study (TMA1, TMA2) have been published in Scrima et al. and in Malanga et al. [48, 49]. Of the 132 cases present on the TMAs, 114 cases could be analyzed for pHER2 (among these, stage was known for 112 patients), 120 cases were analyzed for HER2 (among these, stage was known for 118 patients) and 102 cases were analyzed for pERK1/2 (among these, stage was known for 100 patients). Patients' clinical-pathological characteristics are described in detail in Supplemental Table 1 and summarized in Materials and Methods.
As a read-out of HER2 signaling in NSCLC we determined the phosphorylation status of residue Y1248 of HER2 (pHER2). PHER2 was evaluated on TMAs containing duplicated core biopsies of 114 NSCLCs. The results obtained from pHER2 staining in NSCLC are summarized in Table 1. HER2 activation was observed in 41 out of 114 of NSCLC analyzed (36%). Data were also analyzed by histological type and/or disease stage. As to the histological type, we found that pHER2 staining was observed at similar frequency in ADC and SCC, being detected in 15/47 SCC (32%) and 26/67 ADC (39%) (Figure 1A and B, respectively).
Association of pHER2, pERK1/2 and/or a combination thereof with clinicopathological parameters.
| Characteristics | pERK1/2 | p-value | pHER2 | p-value | pHER2-pERK1/2 | p-value | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | Low | High | N | Low | High | N | Low/Low | High/High | Low/High | ||||
| Age (years) | |||||||||||||
| <64 years | 37 | 22 | 15 | 0.836 | 41 | 28 | 13 | 0.545 | 32 | 13 | 7 | 12 | 0.889 |
| ≥64 years | 65 | 36 | 29 | 73 | 45 | 28 | 62 | 22 | 16 | 24 | |||
| Gender | |||||||||||||
| Male | 83 | 48 | 35 | 0.799 | 92 | 60 | 32 | 0.626 | 76 | 29 | 17 | 30 | 0.636 |
| Female | 19 | 10 | 9 | 22 | 13 | 9 | 18 | 6 | 6 | 6 | |||
| Histological type | |||||||||||||
| ADC | 57 | 33 | 24 | 0.843 | 67 | 41 | 26 | 0.553 | 54 | 19 | 14 | 21 | 0.897 |
| SSC | 45 | 25 | 20 | 47 | 32 | 15 | 40 | 16 | 9 | 15 | |||
| pT stage | |||||||||||||
| T1+T2 | 90 | 49 | 41 | 0.292 | 97 | 60 | 37 | 0.132 | 83 | 30 | 22 | 31 | 0.197 |
| T3+T4 | 9 | 7 | 2 | 14 | 12 | 2 | 9 | 5 | 0 | 4 | |||
| pN stage | |||||||||||||
| N0 | 73 | 45 | 28 | 0.057 | 82 | 53 | 29 | 1 | 67 | 29 | 16 | 22 | 0.140 |
| N1+N2 | 24 | 9 | 15 | 25 | 16 | 9 | 23 | 5 | 6 | 12 | |||
| Differentiation | |||||||||||||
| G1+G2 | 56 | 33 | 23 | 0.540 | 62 | 39 | 23 | 0.841 | 51 | 20 | 13 | 18 | 0.857 |
| G3+G4 | 40 | 21 | 19 | 47 | 31 | 16 | 38 | 13 | 9 | 16 | |||
| TNM Stage | |||||||||||||
| I+II | 78 | 49 | 29 | 0.031* | 89 | 57 | 32 | 0.811 | 71 | 28 | 16 | 27 | 0.517 |
| III+IV | 22 | 8 | 14 | 23 | 14 | 9 | 21 | 6 | 7 | 8 | |||
Association between biomarkers and clinicopathological parameters were assessed using contingency tables with a Fisher's exact test.
Immunostaining of pHER2 and pERK1/2 in NSCLC. A. Representative immunostaining of pHER2 (Y1248) in ADC. B. Representative immunostaining of pHER2 (Y1248) in SCC. C. Representative immunostaining of pERK1/2 (T202/Y204) in ADC. D. Representative immunostaining of pERK1/2 (T202/Y204) in SCC. Magnification: x40; x10 in the inset at bottom right.
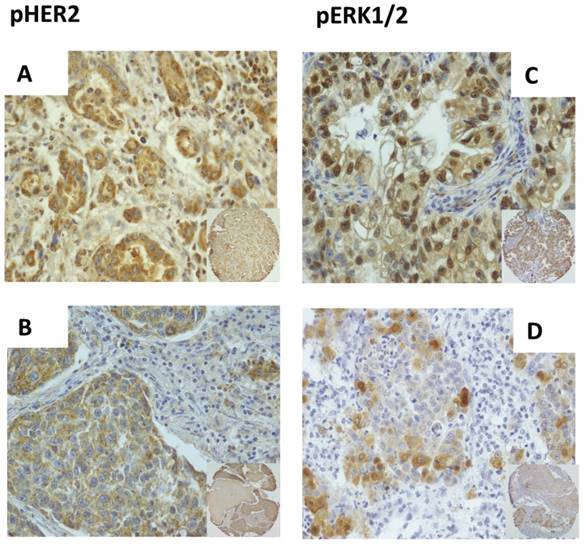
As to disease stage, pHER2 positivity was observed at similar frequency in patients with early stage cancer (stage I+II patients) or advanced disease (stage III+IV patients) being observed in 32/89 patients in stage I+II (36%) and 9/23 patients with cancer in stages III+IV (39%).
Pathways activated by HER2 in NSCLCs
Subsequently, we investigated the signaling pathways activated by deregulated activity of HER2 in NSCLC. To this aim we studied the activities of MAPK-dependent pathway and associated it with HER2 activation.
As a read-out of signaling downstream of HER2 in NSCLC we first determined the phosphorylation status of ERK1/2. PERK1/2 was evaluated on TMAs containing duplicated core biopsies of 102 NSCLCs. The results are summarized in Table 1. ERK1/2 activation was observed in 44/102 of NSCLC analyzed (43%). Notably, we found that pERK1/2 is associated with activated HER2 (n=94; p=0.038). See Table 2 for analysis of data. In particular, of the 41 pHER2-positive NSCLC samples 23 showed positivity for pERK1/2 (56%) whereas of the 53 pHER2-negative NSCLC samples only 18 showed positivity for pERK1/2 (34%), suggesting that tumors with phosphorylated HER2 preferentially presented activated ERK signaling. On the other hand, in the 18 HER2-negative, ERK1/2-positive NSCLC samples, other mechanisms such as mutations of EGFR or KRAS may account for the observed ERK1/2 activation.
Association of pHER2 with total HER2, pERK1/2, pAKT.
| HER2 | pERK1/2 | pAKT | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | Low | High | p-value | N | Low | High | p-value | N | Low | High | p-value | |||
| pHER2 | Low | 71 | 53 | 18 | 0.510 | Low | 53 | 35 | 18 | 0.038* | 62 | 24 | 38 | 0.015* |
| High | 40 | 27 | 13 | High | 41 | 18 | 23 | 40 | 26 | 14 | ||||
The association between biomarkers was determined using contingency tables with a Fisher's exact test.
As to the histological type, we found that pERK1/2 staining was observed at similar frequency in both major histological types of NSCLC (Table 1), being detected in 24/57 ADC (42%) and 20/45 SCC (44%) (Figure 1C and D, respectively). Data regarding ERK1/2 activation were also analyzed by disease stage. In our cohort of patients pERK1/2 positivity was observed more frequently in patients with advanced disease. In fact, when stratified for stage, NSCLC patients showed active ERK1/2 more frequently when in stages III+IV (14/22, 64%) than when in stages I+II (29/78, 37%).
The activation status of the PI3K/AKT pathway in the NSCLC samples under study here had already been analyzed [48]. Here, we only investigated the data for pAKT staining published in those previous works with the data of pHER2 staining of Table 2 of this manuscript, and found that the association between activated HER2 and pAKT was apparently inverse (n=102; p=0.015).
Mechanisms of activation of HER2
To investigate the molecular mechanisms leading to the activation of HER2 in the cohort of Italian NSCLC patients analyzed here, we investigated expression and genetic status of HER2. Analysis of TMAs showed that HER2 was overexpressed in 33/120 NSCLC cases (27%), showing 18% (13/71) of overexpression in ADCs and 41% (20/49) in SCCs (Figure 2A and B). Notably, only 32% of tumors (13/40) who were positive for pHER2 were also positive for increased expression of HER2, suggesting that mechanisms other than overexpression lead to HER2 activation in the remaining cases. The overexpression of HER2 oncoprotein was observed at similar frequency in patients with advanced disease [stage III+IV patients (6/22, 27%)] as patients with early stage cancer [stage I+II patients (27/96, 28%)].
Immunostaining and FISH analysis of HER2 in NSCLC. A. Representative immunostaining of HER2 in ADC. B. Representative immunostaining of HER2 in SCC. C. Gene copy number analysis of HER2 gene in a diploid sample by FISH. Blue DAPI scores nuclei. Red Cy5-labelled probe scores HER2. Green Cy3-labelled probe scores the centromere of chromosome 7. D. Gene copy number analysis of HER2 gene in a high polisomy sample by FISH. Blue DAPI scores nuclei. Red Cy5-labelled probe scores HER2. Green Cy3-labelled probe scores the centromere of chromosome 17. (A, B) Magnification: x40; x10 in the inset at bottom right. (C, D) Magnification, 60x.
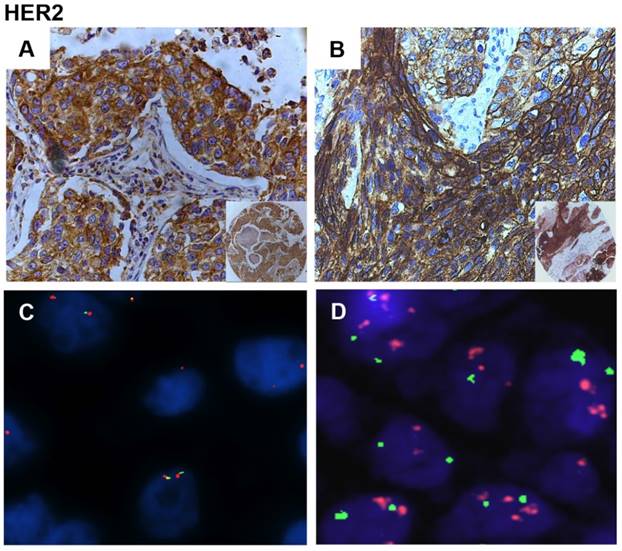
We then determined the molecular mechanisms of the overexpression of HER2 in NSCLC samples studied here by FISH analysis (Figure 2C and D). See Materials and Methods for classification of HER2-positive and negative tumors by FISH. We found that 27/115 NSCLCs (23%) presented copy number gain of HER2 at chromosome 17, all of which were high polysomic (Figure 2D) in agreement with literature data [47]. FISH positivity was observed in 13/71 ADC (18%) and 14/44 SCC (32%). See Supplemental Table 2 for a summary of the results.
High HER2 polysomy was detected in 7/28 (25%) of the samples that overexpressed HER2 and in 4/34 (12%) that presented HER2 activation, suggesting that HER2 over-expression in NSCLC may be accounted for by gene and/or chromosome copy number gain in approximately 25% of cases. On the other hand, 7 out of 22 NSCLC samples (32%) positive by FISH for HER2 showed moderate and/or high HER2 expression.
When the data regarding HER2 copy number were analyzed by disease stage we observed that in the cohort of patients under study, FISH positivity for HER2 was observed at similar frequency in patients with early stage cancer [stage I+II patients (22/92, 24%)] as in patients with advanced disease [stage III+IV patients (5/21, 24%)].
Association of the activation status of HER2, ERK1/2 and combined pHER2-pERK1/2 with clinical features of unselected NSCLC patients
Finally, we compared the activation status of HER2, ERK1/2 and/or a combination thereof with the clinical parameters of the patients under analysis. See Table 1 for a summary of the results. First we analyzed all patients and failed to find any significant association between pHER2, pERK1/2 or pHER2-pERK1/2 with clinical-pathological parameters except for patients' stage. As shown in Table 1, positivity for pERK1/2 was significantly enriched in NSCLC patients in advanced stage of the disease (stages III+IV) (n=100; p=0.031).
Subsequently, using Kaplan-Meier survival curve we investigated the relationship of pHER2 positivity, pERK1/2 positivity and a combination thereof with survival (OS, DFS) (Figure 3). We found that pERK1/2 positivity is significantly associated with poor prognosis (n=64; p=0.002 for OS; p=0.013 for DFS), as the combined positivity for pHER2 and pERK (pHER2-pERK1/2) that presented significantly shorter OS (n=64; p=0.024) and DFS (n=64; p=0.047). No statistical difference was detected in OS and DFS between pHER2 positive and negative patients (n=76).
Kaplan-Meier analysis of 3-year survival in NSCLC patients. A. Kaplan-Meier survival curve of OS in NSCLC patients that were stratified for being positive or negative for pHER2. B. Kaplan-Meier survival curve of DFS in NSCLC patients that were stratified for being positive or negative for pHER2. C. Kaplan-Meier survival curve of OS in NSCLC patients that were stratified for being positive or negative for pERK1/2. D. Kaplan-Meier survival curve of DFS in NSCLC patients that were stratified for being positive or negative for pERK1/2. E. Kaplan-Meier survival curve of OS in NSCLC patients that were stratified for being positive or negative for pHER2- pERK1/2. F. Kaplan-Meier survival curve of DFS in NSCLC patients that were stratified for being positive or negative for pHER2- pERK1/2.
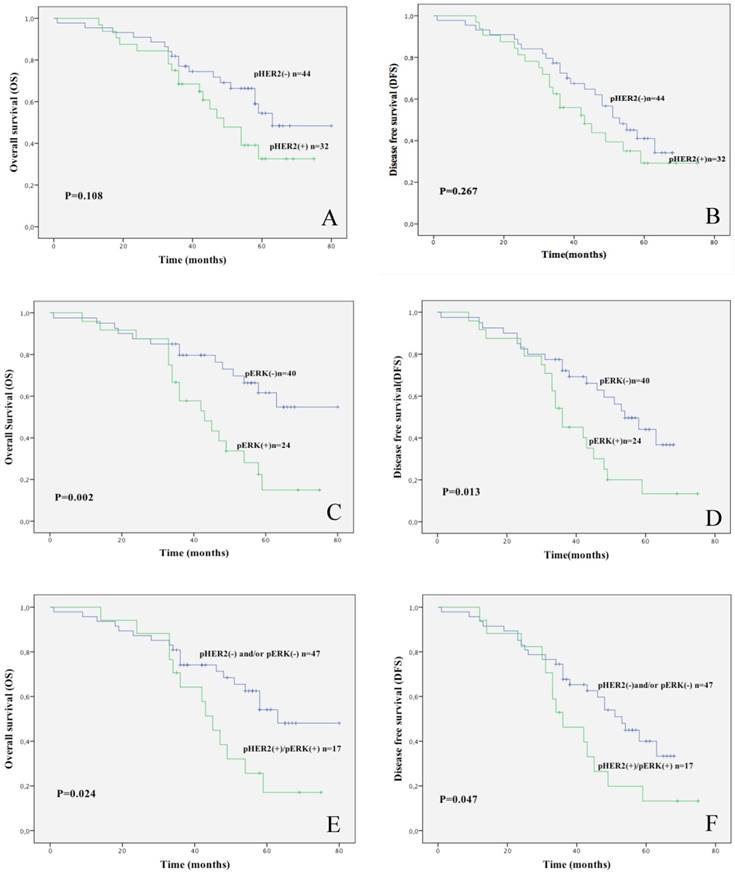
However, upon stratification for pERK1/2 status, the survival at 3 years of unselected pERK1/2 negative patients was 65% whereas survival of pERK1/2 positive patients was 25%. Similarly, survival of pHER2 negative patients was 59% whereas that of pHER2 positive was 44%. The best parameter that discriminated unselected NSCLC patients in our study was double positivity for pHER2-pERK1/2. Survival of pHER2-pERK1/2 positive patients was 60% whereas survival of all others was 23.5%. Similarly, relapse rates at 3 years of unselected NSCLC patients under analysis were 50% for pERK1/2 positive patients and 21% for pERK1/2 negative patients, 45.5% for pHER2 positive patients and 37.5% for pHER2 negative patients, 47% for pHER2-pERK1/2 positive patients and 18% for all other patients.
Univariate Cox Regression Analysis was concordant with results of the Kaplan-Meier curves. Patients with pERK1/2 positivity had significantly shorter OS (HR, 2.862-fold) and DFS (HR, 2.186-fold) as shown by the calculated value of the hazard ratio whereas patients positive for pHER2-pERK1/2 presented significantly shorter OS (HR, 2.195-fold) but not DFS (HR, 1.917-fold) (Table 3). As expected, we found that TNM stage was significantly related to shorter OS and DFS (see Table 3). Importantly, also when assessed by multivariate analysis, both pERK1/2 and the combined positivity for pHER2 and pERK1/2 turned out to be independent prognostic factors for unselected NSCLC patients (Table 4).
Univariate Cox regression analysis in unselected NSCLC patients.
| Variables | OS | DFS | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-value | HR | 95%CI | p-value | |
| Age (>64 vs<64) | 1.845 | 0.903-3.771 | 0.093 | 1.562 | 0.834-2.924 | 0.164 |
| Gender (male/female) | 0.714 | 0.325-1.567 | 0.401 | 0.665 | 0.328-1.348 | 0.258 |
| Histological type (ADC/SCC) | 1.607 | 0.834-3.097 | 0.157 | 1.525 | 0.844-2.757 | 0.162 |
| Lymph node metastasis (N0/N1+N2) | 2.019 | 0.980-4.157 | 0.057 | 1.669 | 0.851-3.372 | 0.136 |
| TNM stage (I+II/III+IV) | 2.530 | 1.248-5.125 | 0.010* | 2.129 | 1.103-4.108 | 0.024* |
| Differentiation Grade (G1+G2/G3+G4) | 1.389 | 0.719-2.682 | 0.328 | 1.699 | 0.936-3.084 | 0.081 |
| pHER2 (positive/negative) | 1.697 | 0.879-3.275 | 0.115 | 1.396 | 0.770-2.533 | 0.272 |
| pERK (positive/negative) | 2.862 | 1.412-5.798 | 0.004* | 2.186 | 1.156-4.134 | 0.016* |
| pHER2-pERK (positive/other1) | 2.195 | 1.081-4.457 | 0.030* | 1.917 | 0.992-3.706 | 0.053 |
* p-value < 0.05.
1 Others include either or neither negatives.
Multivariate analysis in unselected NSCLC patients.
| Variables | OS | DFS | ||||
|---|---|---|---|---|---|---|
| Model 1 | HR | 95% CI | p-value | HR | 95% CI | p-value |
| Age (>64 vs<64) | 1.918 | 0.770-4.777 | 0.162 | 2.168 | 1.010-4.655 | 0.047* |
| Gender (male/female) | 0.917 | 0.346-2.435 | 0.862 | 0.645 | 0.259-1.610 | 0.348 |
| Histological type (ADC/SCC) | 1.576 | 0.626-3.967 | 0.334 | 1.143 | 0.530-2.463 | 0.734 |
| Lymph node metastasis (N0. N1+N2) | 1.356 | 0.547-3.362 | 0.511 | 1.064 | 0.471-2.404 | 0.881 |
| TNM stage (I+II. III+IV) | 1.863 | 0.743-4.668 | 0.185 | 1.407 | 0.628-3.153 | 0.406 |
| Differentiation Grade (G1+G2. G3+G4) | 1.294 | 0.558-2.999 | 0.548 | 2.041 | 0.993-4.192 | 0.052 |
| pHER2 (positive/negative) | 1.282 | 0.528-3.113 | 0.584 | 1.064 | 0.491-2.305 | 0.875 |
| pERK1/2 (positive/negative) | 2.618 | 1.084-6.326 | 0.032* | 2.431 | 1.069-5.527 | 0.034* |
| Model 2 | HR | 95% CI | p-value | HR | 95% CI | p-value |
| Age (>64 vs<64) | 1.822 | 0.717-4.628 | 0.207 | 2.015 | 0.923-4.398 | 0.079 |
| Gender (male/female) | 0.857 | 0.310-2.367 | 0.765 | 0.631 | 0.250-1.592 | 0.329 |
| Histological type (ADC/SCC) | 1.449 | 0.592-3.546 | 0.417 | 1.155 | 0.558-2.393 | 0.697 |
| Lymph node metastasis (N0. N1+N2) | 1.630 | 0.667-3.983 | 0.284 | 1.332 | 0.601-2.952 | 0.480 |
| TNM stage (I+II. III+IV) | 2.024 | 0.819-5.001 | 0.126 | 1.508 | 0.688-3.306 | 0.304 |
| Differentiation Grade (G1+G2. G3+G4) | 1.359 | 0.594-3.108 | 0.468 | 2.173 | 1.054-4.482 | 0.036* |
| pHER2-pERK1/2 (positive/others1) | 2.686 | 1.168-6.174 | 0.020* | 2.443 | 1.148-5.200 | 0.021* |
*p-value < 0.05.
1 Others include either or neither negatives.
Association of the activation status of HER2, ERK1/2 and combined pHER2-pERK1/2 with clinical features of NSCLC patients in early stages
Given the importance to identify prognostic factors in early stage tumors, we also investigated the role of activated HER2 and ERK1/2 in patients with cancer in stages I+II. The number of patients with cancer in stages I+II in the cohort under analysis was 106. Among these patients, information about HER2 status was available for 89, information about pERK1/2 status was available for 78 and information for both proteins was available for 70.
By analysis of the clinical-pathological parameters of the patients with cancer in stages I+II, we found that pHER2 positivity (32/89, 36%), as well as the combined pHER2-pERK1/2 positivity (16/70, 23%) predicted significantly reduced OS and DFS (OS: p=0.009 and p=0.023, respectively; DFS: p=0.038 and p=0.038, respectively) whereas pERK1/2 positivity (29/78, 37%) predicted only reduced OS (p=0.044) as showed by Kaplan Meier curves (Fig.4).
Kaplan-Meier analysis of 3-year survival in stage I+II NSCLC patients. A. Kaplan-Meier survival curve of OS in stage I+II NSCLC patients that were stratified for being positive or negative for pHER2. B. Kaplan-Meier survival curve of DFS in NSCLC patients that were stratified for being positive or negative for pHER2. C. Kaplan-Meier survival curve of OS in NSCLC patients that were stratified for being positive or negative for pERK1/2. D. Kaplan-Meier survival curve of DFS in NSCLC patients that were stratified for being positive or negative for pERK1/2. E. Kaplan-Meier survival curve of OS in NSCLC patients that were stratified for being positive or other combinations for pHER2-pERK1/2. F. Kaplan-Meier survival curve of DFS in NSCLC patients that were stratified for being positive or other combinations for pHER2-pERK1/2.
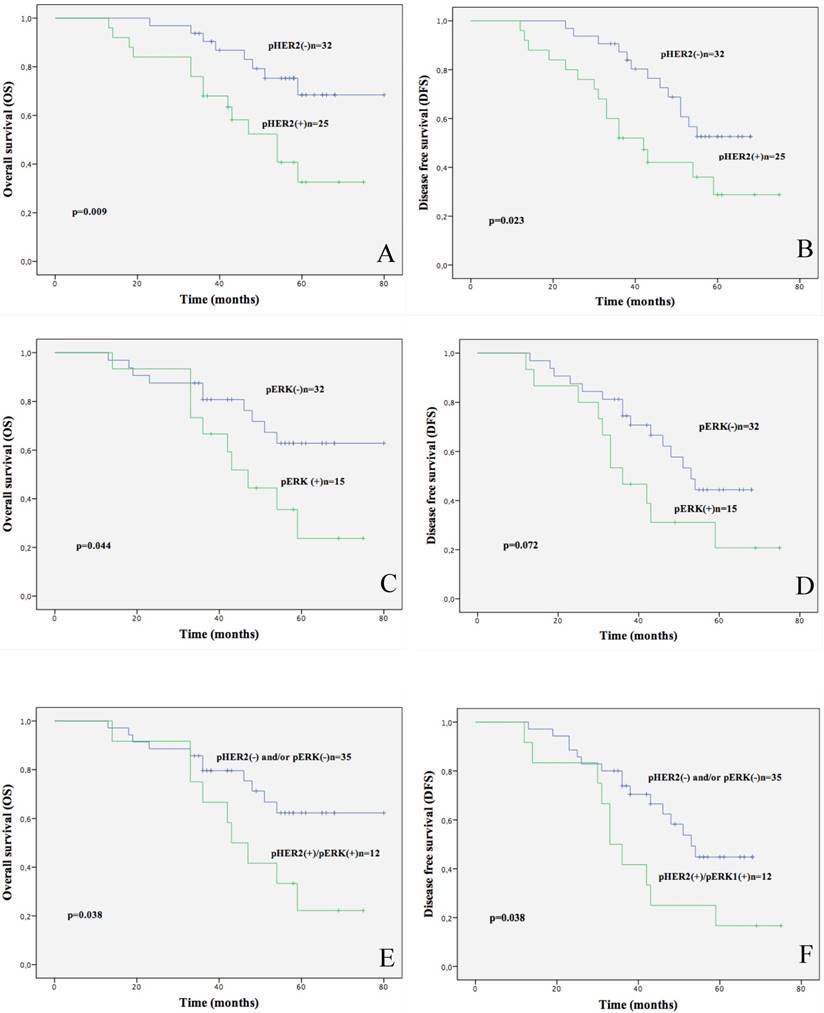
The average survival rate at 3-year of patients in stage I+II NSCLC patients under analysis was 58%. Upon stratification of patients, we found that positivity for pHER2 predicted poor survival. In fact, 3 year-survival of pHER2 negative patients was 75% whereas survival of pHER2 positive patients was 44%; 3 year-survival of pERK1/2 negative patients was 69% whereas survival of pERK1/2 positive patients was 33%; survival of pHER2-pERK1/2 positive patients was 25% whereas survival of all other patients was 69%.
Similarly, 3 year-relapse rate of stage I+II NSCLC patients under analysis was 59% for pHER2 positive patients, 36% for pHER2 negative patients, 53% for pERK1/2 positive patients, 27% for pERK1/2 negative patients, 54% for pHER2-pERK1/2 positive patients and 17% for all other patients.
In agreement with results from the Kaplan-Meier curves, stage I+II patients with pHER2 and/or pHER2-pERK1/2 positivity presented significantly shorter OS (HR, 3.016-fold and HR, 2.440-fold, respectively) and DFS (HR, 2.280-fold and HR, 2.250-fold, respectively) as determined by hazard ratio values in univariate Cox Regression analysis (Table 5). However, when assessed by multivariate analysis, only the combined positivity for pHER2 and pERK1/2 (but not the single pHER2 or pERK1/2 factors) turned out to be an independent prognostic factor for stage I+II NSCLC patients for DFS (p=0.015; HR 2.843-fold) but not for OS (Table 6).
Univariate Cox regression analysis of pERK, pHER2 and a combination thereof with OS and DFS in stage I and II NSCLC patients.
| Variables | OS | DFS | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-value | HR | 95%CI | p-value | |
| pHER2 (positive/negative) | 3.016 | 1.260- 7.221 | 0.013* | 2.280 | 1.090- 4.760 | 0.028* |
| pERK 1/2(positive/negative) | 2.381 | 0.989-5.734 | 0.053 | 2.011 | 0.920- 4.397 | 0.080 |
| pHER2-pERK1/2 (positive/others1) | 2.440 | 1.010- 5.890 | 0.047* | 2.250 | 1.017-4.979 | 0.045* |
*p-value < 0.05.
1 Others include either or neither negatives.
Multivariate analysis in stage I+II NSCLC patients.
| Variables | OS | DFS | ||||
|---|---|---|---|---|---|---|
| Model 1 | HR | 95% CI | p-value | HR | 95%CI | p-value |
| Age (>64 vs<64) | 1.280 | 0.436-3.758 | 0.654 | 2.451 | 0.920-6.524 | 0.073 |
| Gender (male/female) | 0.991 | 0.270-3.635 | 0.989 | 0.550 | 0.160-1.884 | 0.341 |
| Histological type (ADC/SCC) | 2.782 | 0.754-10.270 | 0.125 | 1.246 | 0.433-3.588 | 0.684 |
| Differentiation Grade (G1+G2/G3+G4) | 1.094 | 0.386-3.102 | 0.867 | 2.390 | 0.991-5.761 | 0.052 |
| pHER2 (positive/negative) | 3.456 | 0.874-13.669 | 0.077 | 1.681 | 0.600-4.712 | 0.323 |
| pERK1/2 (positive/negative) | 1.117 | 0.340-3.674 | 0.855 | 2.098 | 0.734-5.999 | 0.167 |
| Model 2 | HR | 95% CI | p-value | HR | 95%CI | p-value |
| Age (>64 vs<64) | 1.401 | 0.478-4.106 | 0.539 | 2.252 | 0.873-5.806 | 0.093 |
| Gender (male/female) | 0.880 | 0.240-3.229 | 0.847 | 0.550 | 0.170-1.784 | 0.320 |
| Histological type (ADC/SCC) | 1.663 | 0.537-5.148 | 0.377 | 1.074 | 0.421-2.738 | 0.882 |
| Differentiation Grade (G1+G2/G3+G4) | 1.350 | 0.510-3.571 | 0.546 | 2.475 | 1.041-5.884 | 0.040* |
| pHER2-pERK1/2 (positive/others1) | 2.339 | 0.956-5.724 | 0.063 | 2.843 | 1.229-6.578 | 0.015* |
*p-value < 0.05.
1 Others include either or neither negatives.
Discussion
The tyrosine kinase receptors EGFR and HER2 are frequently activated in NSCLC [17, 29, 50-52], and for this reason have been the focus of several studies. EGFR is activated both by gain-of-function mutations [53] and by protein overexpression in NSCLC [54-56].
Similarly, both mutations and overexpression of the related HER2 tyrosine kinase receptor contribute to the development of lung cancer. However, HER2 alterations are less frequent than those reported for EGFR, being found in about 7-35% of patients with NSCLC [15], [16], [18, 19, 57-61].
The purpose of this study was to evaluate the contribution of pathways signaling downstream HER2 to the development of lung cancer. The most relevant findings reported herein are that: i) in NSCLC, the activation of ERK1/2- but not AKT-dependent pathway, is directly associated with activated HER2, ii) in unselected NSCLC patients, pERK1/2 positivity is predictive of reduced survival (both DFS and OS) in univariate analysis and, when combined with pHER2, represent statistically significant indicators of poor prognosis independent of the most important clinical-pathological prognostic factors in multivariate analysis, iii) in early stages (I+II) NSCLC patients, the most relevant factors that predict reduced survival (both DFS and OS) in univariate analysis are pHER2 and pHER2-pERK1/2 whereas, in multivariate analysis, only the combined pHER2-pERK1/2 positivity represents a statistically significant indicator of poor prognosis (DFS) independent of the most relevant clinical-pathological prognostic factors.
The first conclusion derives from the observation that in immunostaining experiments performed on TMAs containing matched tumor and normal lung tissues the activation of ERK1/2 (using pERK1/2 as read-out) is significantly associated with high level of phosphorylated HER2. HER2 was overexpressed in 27% and activated (using phosphorylated Y1248-pHER2 as read-out) in 36% of tumors whereas ERK1/2 activation was observed in approximately 43% of tumors. These results obtained in the cohort of Italian patients under study here are in line with previous studies showing that the ERK1/2 pathway plays an important role in lung cancerogenesis, being over-activated in at least 30-50% NSCLC (particularly in those carrying EGFR and K-RAS mutations) [42, 62], and that the ERK1/2-dependent pathway represents a key downstream effector of HER2 [63].
Also the results of HER2 staining are in agreement with previous studies showing that approximately 6-35% of NSCLC tumors show positivity for HER2 [57, 64]. In addition, the frequency of HER2 positivity detected in the cohort of patients under analysis in this manuscript was within the reported range in ADC (17-42%) and SCC (∼40%) [47, 65].
As to the mechanisms whereby HER2 is overexpressed in NSCLC, results from FISH analysis indicated that an increase of copy number of the HER2 gene at 17q12 is responsible for ∼25% of cases with enhanced HER2 expression and in ∼12% of tumors that presented HER2 activation. These results are in agreement with previous reports indicating that gains of part or of the entire long arm of chromosome 17, where the HER2 gene maps, are recurrent in NSCLC, ranging from 2% to 23%, depending on the study [65, 66]. In a recent meta-analysis that confirmed the unfavorable prognosis associated with HER2 overexpression in NSCLC, the frequency of HER2 positivity was significantly increased in patients with ADC histology (38% versus 16% in SCC, respectively) [18]. Yet, since several NSCLCs overexpress HER2 in the absence of gene copy number gains, other mechanisms must be involved in the deregulation of the expression of this oncogene in Italian NSCLC patients.
The second relevant point that we make in this manuscript is that activated HER2 combined with activated ERK1/2 (pHER2-pERK1/2) in univariate and multivariate analysis predict poor outcome in unselected and in early stage NSCLC patients.
Currently, disease stage is the main prognostic factor for survival in patients with NSCLC, with a median survival of >8 years for stage I patients and <1 year for stage IV patients. Although surgery can be curative for a subset of early stage I+II patients, a significant number of early stage NSCLC patients recurs [67]. Demographic characteristics of patients such as gender, age, smoking habit or histopathological features of tumors that include histology, tumour size, local extension and infiltration or nodal status have been suggested as prognostic factors for NSCLC patients [68]. In addition, several single (i.e. K-RAS, ErbB receptors, bcl-2, Sox2) or multiple (gene or miRNA signatures) molecular determinants have been proposed as predictive factors for survival of NSCLC patients [60, 69-72]. However, none of these approaches has turned out to be completely satisfactory in predicting disease progression and clinical outcome, especially in early stage patients [73-75].
The conclusions derived from this study indicate that positivity for the combination pHER2-pERK1/2 predicts significantly shorter OS and DFS in NSCLC patients with stages I+II tumors (48.4 vs. 63.7 months, respectively) (40.2 vs. 51 months, respectively) and shorter OS in unselected patients (46.3 vs. 59.1 months, respectively). In addition, multivariate analysis indicated that the combined pHER2-pERK1/2 positivity (but not HER2 or ERK1/2) represents a statistically significant indicator of poor prognosis independent of the most important clinical-pathological prognostic factors for both unselected and early stage NSCLC patients.
On the other hand, shorter OS and DFS in unselected patients is also significantly associated with pERK1/2 positivity (44.9 vs. 61.9 months; 40.2 versus 49.6 months, respectively) whereas shorter OS and DFS in early stages patients is also significantly associated with pHER2 positivity (50.5 vs. 68.7 months; 45.3 vs. 55.8 months, respectively), though subsequent multivariate analysis failed to confirm pHER2 and pERK1/2 as independent prognostic factors.
These results are in agreement with prior studies showing association of HER2 overexpression with poor prognosis, shorter survival and drug resistance in NSCLC patients [47, 57, 76] as well as association of pERK staining with higher T stages, lymph node involvement, more aggressive tumours and lower survival [42]. However, at difference with the present report, multivariate analysis performed in those previous studies failed to show significant survival difference.
Strikingly, early stage NSCLC patients double positive for pHER2-pERK1/2 showed 3-year survival of 25%, which was much lower in comparison with all other combination (69%). Similarly, 3-year relapse rate observed in pHER2-pERK1/2 double positive patients was higher (54%) than the value observed in all other early stage patients (17%).
It is of note that pERK1/2 positivity reached statistically significance as independent indicator of poor prognosis in unselected NSCLC patients. This observation is in agreement with the finding that in our cohort of patients, positivity for pERK1/2 was significantly enriched in patients in stages III+IV of the disease. In addition, the finding that pERK1/2 as a single factor was not apparently a significant prognostic marker in early stage NSCLC patients, is also in line with a recent report in which single biomarkers within the mTOR/AKT and/or the MAPK pathway were not predictive of survival in NSCLC patients when taken individually [77].
In conclusion, this study provides evidence that the activation of HER2-ERK1/2 pathways represents an early critical event in NSCLC development and that activation of this pathway is predictive of poor prognosis in NSCLC patients (in particular, in patients with early stage cancer). Above all, these findings suggest that the evaluation of the activation status of HER2, ERK1/2 and a combination thereof is a new tool for ameliorating the characterization of NSCLC patients, although further studies with larger cohort of patients are needed to confirm the results reported here.
Supplementary Material
Supplementary tables.
Acknowledgements
This work was supported by Associazione Italiana Ricerca sul Cancro (AIRC, IG_12969) and MIUR (PRIN, 20087FSFFP_001; PON01_02782) to GV and by Regione Campania grant Laboratori pubblici progetto “Hautville” to RF and GB. MS was recipient of a fellowship from Federazione Italiana Ricerca sul Cancro (FIRC).
Conflicts of Interest
The authors have disclosed that they have no significant relationships with, or financial interest in, any commercial companies pertaining to this article.
References
1. Weir BA, Woo MS, Getz G, Perner S, Ding L, Beroukhim R. et al. Characterizing the cancer genome in lung adenocarcinoma. Nature. 2007;450:893-8
2. Greenman C, Stephens P, Smith R, Dalgliesh GL, Hunter C, Bignell G. et al. Patterns of somatic mutation in human cancer genomes. Nature. 2007;446:153-8
3. Lu Y, Lemon W, Liu PY, Yi Y, Morrison C, Yang P. et al. A gene expression signature predicts survival of patients with stage I non-small cell lung cancer. PLoS medicine. 2006;3:e467
4. Bild AH, Yao G, Chang JT, Wang Q, Potti A, Chasse D. et al. Oncogenic pathway signatures in human cancers as a guide to targeted therapies. Nature. 2006;439:353-7
5. Rikova K, Guo A, Zeng Q, Possemato A, Yu J, Haack H. et al. Global survey of phosphotyrosine signaling identifies oncogenic kinases in lung cancer. Cell. 2007;131:1190-203
6. Lipson D, Capelletti M, Yelensky R, Otto G, Parker A, Jarosz M. et al. Identification of new ALK and RET gene fusions from colorectal and lung cancer biopsies. Nature medicine. 2012;18:382-4
7. Kohno T, Ichikawa H, Totoki Y, Yasuda K, Hiramoto M, Nammo T. et al. KIF5B-RET fusions in lung adenocarcinoma. Nature medicine. 2012;18:375-7
8. Sekido Y, Fong KM, Minna JD. Molecular genetics of lung cancer. Annual review of medicine. 2003;54:73-87
9. Takeuchi K, Soda M, Togashi Y, Suzuki R, Sakata S, Hatano S. et al. RET, ROS1 and ALK fusions in lung cancer. Nature medicine. 2012;18:378-81
10. Malanga D, Scrima M, De Marco C, Fabiani F, De Rosa N, De Gisi S. et al. Activating E17K mutation in the gene encoding the protein kinase AKT1 in a subset of squamous cell carcinoma of the lung. Cell cycle. 2008;7:665-9
11. Amodio N, Scrima M, Palaia L, Salman AN, Quintiero A, Franco R. et al. Oncogenic role of the E3 ubiquitin ligase NEDD4-1, a PTEN negative regulator, in non-small-cell lung carcinomas. The American journal of pathology. 2010;177:2622-34
12. Malanga D, Belmonte S, Colelli F, Scarfo M, De Marco C, Oliveira DM. et al. AKT1E(1)(7)K Is Oncogenic in Mouse Lung and Cooperates with Chemical Carcinogens in Inducing Lung Cancer. PloS one. 2016;11:e0147334
13. Hynes NE, Lane HA. ERBB receptors and cancer: the complexity of targeted inhibitors. Nature reviews Cancer. 2005;5:341-54
14. Jackman DM, Chirieac LR, Janne PA. Bronchioloalveolar carcinoma: a review of the epidemiology, pathology, and treatment. Seminars in respiratory and critical care medicine. 2005;26:342-52
15. Stephens P, Hunter C, Bignell G, Edkins S, Davies H, Teague J. et al. Lung cancer: intragenic ERBB2 kinase mutations in tumours. Nature. 2004;431:525-6
16. Shigematsu H, Takahashi T, Nomura M, Majmudar K, Suzuki M, Lee H. et al. Somatic mutations of the HER2 kinase domain in lung adenocarcinomas. Cancer research. 2005;65:1642-6
17. Pao W, Girard N. New driver mutations in non-small-cell lung cancer. The lancet oncology. 2011;12:175-80
18. Swanton C, Futreal A, Eisen T. Her2-targeted therapies in non-small cell lung cancer. Clinical cancer research: an official journal of the American Association for Cancer Research. 2006;12:4377s-83s
19. Timotheadou E, Skarlos DV, Samantas E, Papadopoulos S, Murray S, Skrickova J. et al. Evaluation of the prognostic role of a panel of biomarkers in stage IB-IIIA non-small cell lung cancer patients. Anticancer research. 2007;27:4481-9
20. Ben-Levy R, Paterson HF, Marshall CJ, Yarden Y. A single autophosphorylation site confers oncogenicity to the Neu/ErbB-2 receptor and enables coupling to the MAP kinase pathway. The EMBO journal. 1994;13:3302-11
21. Dankort D, Jeyabalan N, Jones N, Dumont DJ, Muller WJ. Multiple ErbB-2/Neu Phosphorylation Sites Mediate Transformation through Distinct Effector Proteins. The Journal of biological chemistry. 2001;276:38921-8
22. Klapper LN, Glathe S, Vaisman N, Hynes NE, Andrews GC, Sela M. et al. The ErbB-2/HER2 oncoprotein of human carcinomas may function solely as a shared coreceptor for multiple stroma-derived growth factors. Proceedings of the National Academy of Sciences of the United States of America. 1999;96:4995-5000
23. Klapper LN, Waterman H, Sela M, Yarden Y. Tumor-inhibitory antibodies to HER-2/ErbB-2 may act by recruiting c-Cbl and enhancing ubiquitination of HER-2. Cancer research. 2000;60:3384-8
24. Santen RJ, Song RX, McPherson R, Kumar R, Adam L, Jeng MH. et al. The role of mitogen-activated protein (MAP) kinase in breast cancer. The Journal of steroid biochemistry and molecular biology. 2002;80:239-56
25. Wetzker R, Bohmer FD. Transactivation joins multiple tracks to the ERK/MAPK cascade. Nature reviews Molecular cell biology. 2003;4:651-7
26. Akiyama T, Matsuda S, Namba Y, Saito T, Toyoshima K, Yamamoto T. The transforming potential of the c-erbB-2 protein is regulated by its autophosphorylation at the carboxyl-terminal domain. Molecular and cellular biology. 1991;11:833-42
27. DiGiovanna MP, Lerman MA, Coffey RJ, Muller WJ, Cardiff RD, Stern DF. Active signaling by Neu in transgenic mice. Oncogene. 1998;17:1877-84
28. Pearson G, Robinson F, Beers Gibson T, Xu BE, Karandikar M, Berman K. et al. Mitogen-activated protein (MAP) kinase pathways: regulation and physiological functions. Endocrine reviews. 2001;22:153-83
29. Davies H, Bignell GR, Cox C, Stephens P, Edkins S, Clegg S. et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949-54
30. Malumbres M, Barbacid M. RAS oncogenes: the first 30 years. Nature reviews Cancer. 2003;3:459-65
31. Sarbassov DD, Guertin DA, Ali SM, Sabatini DM. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science. 2005;307:1098-101
32. Montgomery RB, Makary E, Schiffman K, Goodell V, Disis ML. Endogenous anti-HER2 antibodies block HER2 phosphorylation and signaling through extracellular signal-regulated kinase. Cancer research. 2005;65:650-6
33. Kohno M, Pouyssegur J. Targeting the ERK signaling pathway in cancer therapy. Annals of medicine. 2006;38:200-11
34. Ma L, Chen Z, Erdjument-Bromage H, Tempst P, Pandolfi PP. Phosphorylation and functional inactivation of TSC2 by Erk implications for tuberous sclerosis and cancer pathogenesis. Cell. 2005;121:179-93
35. Liu D, Xing M. Potent inhibition of thyroid cancer cells by the MEK inhibitor PD0325901 and its potentiation by suppression of the PI3K and NF-kappaB pathways. Thyroid: official journal of the American Thyroid Association. 2008;18:853-64
36. Engelman JA, Chen L, Tan X, Crosby K, Guimaraes AR, Upadhyay R. et al. Effective use of PI3K and MEK inhibitors to treat mutant Kras G12D and PIK3CA H1047R murine lung cancers. Nature medicine. 2008;14:1351-6
37. Faber AC, Li D, Song Y, Liang MC, Yeap BY, Bronson RT. et al. Differential induction of apoptosis in HER2 and EGFR addicted cancers following PI3K inhibition. Proceedings of the National Academy of Sciences of the United States of America. 2009;106:19503-8
38. Hirsch FR CB, Colby TV. In: Travis WD BE, Muller-Hermelink HK, Harris CC editor. Tumours of the lung Clinical features and staging In: WHO Classification, Tumours of the Lung, Pleura, Thymus and Heart. Lyon: France: IARC Press. 2004:16-9
39. Travis WD, Brambilla E, Riely GJ. New pathologic classification of lung cancer: relevance for clinical practice and clinical trials. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2013;31:992-1001
40. Mountain CF. The international system for staging lung cancer. Seminars in surgical oncology. 2000;18:106-15
41. Sica G, Yoshizawa A, Sima CS, Azzoli CG, Downey RJ, Rusch VW. et al. A grading system of lung adenocarcinomas based on histologic pattern is predictive of disease recurrence in stage I tumors. The American journal of surgical pathology. 2010;34:1155-62
42. Vicent S, Lopez-Picazo JM, Toledo G, Lozano MD, Torre W, Garcia-Corchon C. et al. ERK1/2 is activated in non-small-cell lung cancer and associated with advanced tumours. British journal of cancer. 2004;90:1047-52
43. Scrima M, De Marco C, De Vita F, Fabiani F, Franco R, Pirozzi G. et al. The nonreceptor-type tyrosine phosphatase PTPN13 is a tumor suppressor gene in non-small cell lung cancer. The American journal of pathology. 2012;180:1202-14
44. Pellegrini C, Falleni M, Marchetti A, Cassani B, Miozzo M, Buttitta F. et al. HER-2/Neu alterations in non-small cell lung cancer: a comprehensive evaluation by real time reverse transcription-PCR, fluorescence in situ hybridization, and immunohistochemistry. Clinical cancer research: an official journal of the American Association for Cancer Research. 2003;9:3645-52
45. Miller TW, Forbes JT, Shah C, Wyatt SK, Manning HC, Olivares MG. et al. Inhibition of mammalian target of rapamycin is required for optimal antitumor effect of HER2 inhibitors against HER2-overexpressing cancer cells. Clinical cancer research: an official journal of the American Association for Cancer Research. 2009;15:7266-76
46. Varella-Garcia M, Mitsudomi T, Yatabe Y, Kosaka T, Nakajima E, Xavier AC. et al. EGFR and HER2 genomic gain in recurrent non-small cell lung cancer after surgery: impact on outcome to treatment with gefitinib and association with EGFR and KRAS mutations in a Japanese cohort. Journal of thoracic oncology: official publication of the International Association for the Study of Lung Cancer. 2009;4:318-25
47. Cappuzzo F, Varella-Garcia M, Shigematsu H, Domenichini I, Bartolini S, Ceresoli GL. et al. Increased HER2 gene copy number is associated with response to gefitinib therapy in epidermal growth factor receptor-positive non-small-cell lung cancer patients. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2005;23:5007-18
48. Scrima M, De Marco C, Fabiani F, Franco R, Pirozzi G, Rocco G. et al. Signaling networks associated with AKT activation in non-small cell lung cancer (NSCLC): new insights on the role of phosphatydil-inositol-3 kinase. PloS one. 2012;7:e30427
49. Malanga D, De Marco C, Guerriero I, Colelli F, Rinaldo N, Scrima M. et al. The Akt1/IL-6/STAT3 pathway regulates growth of lung tumor initiating cells. Oncotarget. 2015;6:42667-86
50. Scagliotti GV, Selvaggi G, Novello S, Hirsch FR. The biology of epidermal growth factor receptor in lung cancer. Clinical cancer research: an official journal of the American Association for Cancer Research. 2004;10:4227s-32s
51. Hirsch FR, Varella-Garcia M, Bunn PA Jr, Di Maria MV, Veve R, Bremmes RM. et al. Epidermal growth factor receptor in non-small-cell lung carcinomas: correlation between gene copy number and protein expression and impact on prognosis. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2003;21:3798-807
52. Shigematsu H, Lin L, Takahashi T, Nomura M, Suzuki M, Wistuba II. et al. Clinical and biological features associated with epidermal growth factor receptor gene mutations in lung cancers. Journal of the National Cancer Institute. 2005;97:339-46
53. Hirsch FR, Bunn PA Jr. EGFR testing in lung cancer is ready for prime time. The lancet oncology. 2009;10:432-3
54. Salomon DS, Brandt R, Ciardiello F, Normanno N. Epidermal growth factor-related peptides and their receptors in human malignancies. Critical reviews in oncology/hematology. 1995;19:183-232
55. Merrick DT, Kittelson J, Winterhalder R, Kotantoulas G, Ingeberg S, Keith RL. et al. Analysis of c-ErbB1/epidermal growth factor receptor and c-ErbB2/HER-2 expression in bronchial dysplasia: evaluation of potential targets for chemoprevention of lung cancer. Clinical cancer research: an official journal of the American Association for Cancer Research. 2006;12:2281-8
56. Grandis JR, Sok JC. Signaling through the epidermal growth factor receptor during the development of malignancy. Pharmacology & therapeutics. 2004;102:37-46
57. Hirsch FR, Franklin WA, Veve R, Varella-Garcia M, Bunn PA Jr. HER2/neu expression in malignant lung tumors. Seminars in oncology. 2002;29:51-8
58. Tan D, Deeb G, Wang J, Slocum HK, Winston J, Wiseman S. et al. HER-2/neu protein expression and gene alteration in stage I-IIIA non-small-cell lung cancer: a study of 140 cases using a combination of high throughput tissue microarray, immunohistochemistry, and fluorescent in situ hybridization. Diagnostic molecular pathology: the American journal of surgical pathology, part B. 2003;12:201-11
59. Au NH, Cheang M, Huntsman DG, Yorida E, Coldman A, Elliott WM. et al. Evaluation of immunohistochemical markers in non-small cell lung cancer by unsupervised hierarchical clustering analysis: a tissue microarray study of 284 cases and 18 markers. The Journal of pathology. 2004;204:101-9
60. Liu L, Shao X, Gao W, Bai J, Wang R, Huang P. et al. The role of human epidermal growth factor receptor 2 as a prognostic factor in lung cancer: a meta-analysis of published data. Journal of thoracic oncology: official publication of the International Association for the Study of Lung Cancer. 2010;5:1922-32
61. Takenaka M, Hanagiri T, Shinohara S, Kuwata T, Chikaishi Y, Oka S. et al. The prognostic significance of HER2 overexpression in non-small cell lung cancer. Anticancer research. 2011;31:4631-6
62. Dhillon AS, Hagan S, Rath O, Kolch W. MAP kinase signalling pathways in cancer. Oncogene. 2007;26:3279-90
63. Roberts PJ, Der CJ. Targeting the Raf-MEK-ERK mitogen-activated protein kinase cascade for the treatment of cancer. Oncogene. 2007;26:3291-310
64. Garrido-Castro AC, Felip E. HER2 driven non-small cell lung cancer (NSCLC): potential therapeutic approaches. Translational lung cancer research. 2013;2:122-7
65. Nakamura H, Saji H, Ogata A, Hosaka M, Hagiwara M, Kawasaki N. et al. Correlation between encoded protein overexpression and copy number of the HER2 gene with survival in non-small cell lung cancer. International journal of cancer Journal international du cancer. 2003;103:61-6
66. Hirsch FR, Varella-Garcia M, Franklin WA, Veve R, Chen L, Helfrich B. et al. Evaluation of HER-2/neu gene amplification and protein expression in non-small cell lung carcinomas. British journal of cancer. 2002;86:1449-56
67. Chang MY, Sugarbaker DJ. Surgery for early stage non-small cell lung cancer. Seminars in surgical oncology. 2003;21:74-84
68. Brundage MD, Davies D, Mackillop WJ. Prognostic factors in non-small cell lung cancer: a decade of progress. Chest. 2002;122:1037-57
69. Sholl LM, Xiao Y, Joshi V, Yeap BY, Cioffredi LA, Jackman DM. et al. EGFR mutation is a better predictor of response to tyrosine kinase inhibitors in non-small cell lung carcinoma than FISH, CISH, and immunohistochemistry. American journal of clinical pathology. 2010;133:922-34
70. Sonobe M, Nakagawa M, Takenaka K, Katakura H, Adachi M, Yanagihara K. et al. Influence of epidermal growth factor receptor (EGFR) gene mutations on the expression of EGFR, phosphoryl-Akt, and phosphoryl-MAPK, and on the prognosis of patients with non-small cell lung cancer. Journal of surgical oncology. 2007;95:63-9
71. Tantraworasin A, Saeteng S, Lertprasertsuke N, Arayawudhikule N, Kasemsarn C, Patumanond J. Completely resected n0 non-small cell lung cancer: prognostic factors affecting long-term survival. ISRN surgery. 2013;2013:175304
72. Korrapati V, Gaffney M, Larsson LG, Di Nunno L, Riggs M, Beissner RS. et al. Effect of HER2/neu expression on survival in non-small-cell lung cancer. Clinical lung cancer. 2001;2:216-9
73. Traynor AM, Weigel TL, Oettel KR, Yang DT, Zhang C, Kim K. et al. Nuclear EGFR protein expression predicts poor survival in early stage non-small cell lung cancer. Lung cancer. 2013;81:138-41
74. Xia Q, Zhu Z, Wang J, Situ D, Zhou N, Jang W. Expression and association of HER2 with prognosis in early-stage (T1-T2N0M0) non-small cell lung cancer. Tumour biology: the journal of the International Society for Oncodevelopmental Biology and Medicine. 2012;33:1719-25
75. Onn A, Correa AM, Gilcrease M, Isobe T, Massarelli E, Bucana CD. et al. Synchronous overexpression of epidermal growth factor receptor and HER2-neu protein is a predictor of poor outcome in patients with stage I non-small cell lung cancer. Clinical cancer research: an official journal of the American Association for Cancer Research. 2004;10:136-43
76. Tsai CM, Chang KT, Perng RP, Mitsudomi T, Chen MH, Kadoyama C. et al. Correlation of intrinsic chemoresistance of non-small-cell lung cancer cell lines with HER-2/neu gene expression but not with ras gene mutations. Journal of the National Cancer Institute. 1993;85:897-901
77. Kitano H, Chung JY, Ylaya K, Conway C, Takikita M, Fukuoka J. et al. Profiling of phospho-AKT, phospho-mTOR, phospho-MAPK and EGFR in non-small cell lung cancer. The journal of histochemistry and cytochemistry: official journal of the Histochemistry Society. 2014;62:335-46
Author contact
Corresponding author: Marianna Scrima, Biogem scarl, Institute of Genetic Research, Ariano Irpino (AV), Italy. email: marianna.scrimait. Pietro Zoppoli, Department of Experimental and Clinical Medicine, University Magna, Graecia, Catanzaro, Italy. email:zoppoli.pietrocom. Giuseppe Viglietto, Dipartimento di Medicina Sperimentale e Clinica, Scuola di Medicina e Chirurgia, Università Magna Graecia. Campus Universitario Germaneto 88100, Catanzaro, Italy. Tel: +390961/3694215 Fax: +390961/3694090 Email: vigliettoit.

 Global reach, higher impact
Global reach, higher impact